[English] 日本語
 Yorodumi
Yorodumi- PDB-3vaw: Crystal structure of a smt fusion peptidyl-prolyl cis-trans isome... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 3vaw | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of a smt fusion peptidyl-prolyl cis-trans isomerase with surface mutation v3i from burkholderia pseudomallei complexed with fk506 | ||||||
 Components Components | Ubiquitin-like protein SMT3,Peptidyl-prolyl cis-trans isomerase | ||||||
 Keywords Keywords | Isomerase / PROTEIN BINDING/INHIBITOR / SSGCID / Structural Genomics / Seattle Structural Genomics Center for Infectious Disease / PROTEIN BINDING / PROTEIN BINDING-INHIBITOR complex | ||||||
| Function / homology |  Function and homology information Function and homology informationSUMO is conjugated to E1 (UBA2:SAE1) / SUMOylation of nuclear envelope proteins / SUMO is transferred from E1 to E2 (UBE2I, UBC9) / SUMO is proteolytically processed / SUMOylation of transcription factors / Postmitotic nuclear pore complex (NPC) reformation / SUMOylation of transcription cofactors / septin ring / SUMOylation of DNA damage response and repair proteins / Transcriptional and post-translational regulation of MITF-M expression and activity ...SUMO is conjugated to E1 (UBA2:SAE1) / SUMOylation of nuclear envelope proteins / SUMO is transferred from E1 to E2 (UBE2I, UBC9) / SUMO is proteolytically processed / SUMOylation of transcription factors / Postmitotic nuclear pore complex (NPC) reformation / SUMOylation of transcription cofactors / septin ring / SUMOylation of DNA damage response and repair proteins / Transcriptional and post-translational regulation of MITF-M expression and activity / SUMOylation of DNA replication proteins / SUMOylation of SUMOylation proteins / Recruitment and ATM-mediated phosphorylation of repair and signaling proteins at DNA double strand breaks / SUMOylation of RNA binding proteins / SUMOylation of chromatin organization proteins / ubiquitin-like protein ligase binding / protein sumoylation / condensed nuclear chromosome / peptidylprolyl isomerase / peptidyl-prolyl cis-trans isomerase activity / protein tag activity / metal ion binding / identical protein binding / nucleus Similarity search - Function | ||||||
| Biological species |   Burkholderia pseudomallei (bacteria) Burkholderia pseudomallei (bacteria) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  molecular replacement / Resolution: 1.55 Å molecular replacement / Resolution: 1.55 Å | ||||||
 Authors Authors | Seattle Structural Genomics Center for Infectious Disease (SSGCID) | ||||||
 Citation Citation |  Journal: Antimicrob.Agents Chemother. / Year: 2014 Journal: Antimicrob.Agents Chemother. / Year: 2014Title: A structural biology approach enables the development of antimicrobials targeting bacterial immunophilins. Authors: Begley, D.W. / Fox, D. / Jenner, D. / Juli, C. / Pierce, P.G. / Abendroth, J. / Muruthi, M. / Safford, K. / Anderson, V. / Atkins, K. / Barnes, S.R. / Moen, S.O. / Raymond, A.C. / Stacy, R. ...Authors: Begley, D.W. / Fox, D. / Jenner, D. / Juli, C. / Pierce, P.G. / Abendroth, J. / Muruthi, M. / Safford, K. / Anderson, V. / Atkins, K. / Barnes, S.R. / Moen, S.O. / Raymond, A.C. / Stacy, R. / Myler, P.J. / Staker, B.L. / Harmer, N.J. / Norville, I.H. / Holzgrabe, U. / Sarkar-Tyson, M. / Edwards, T.E. / Lorimer, D.D. | ||||||
| History |
|
- Structure visualization
Structure visualization
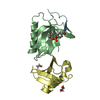
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  3vaw.cif.gz 3vaw.cif.gz | 89.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb3vaw.ent.gz pdb3vaw.ent.gz | 65.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  3vaw.json.gz 3vaw.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/va/3vaw https://data.pdbj.org/pub/pdb/validation_reports/va/3vaw ftp://data.pdbj.org/pub/pdb/validation_reports/va/3vaw ftp://data.pdbj.org/pub/pdb/validation_reports/va/3vaw | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  3uf8C  3uqaSC  3uqbC  4dz2C  4dz3C  4fn2C  4g50C  4ggqC  4givC S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data | |
| Other databases |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 23015.857 Da / Num. of mol.: 1 / Fragment: Q12306 RESIDUES 13-99, Q3JK38 RESIDUES 2-113 / Mutation: V3I,V3I Source method: isolated from a genetically manipulated source Details: FUSION PROTEIN Source: (gene. exp.)   Burkholderia pseudomallei (bacteria) Burkholderia pseudomallei (bacteria)Strain: ATCC 204508 / S288c, 1710b / Gene: SMT3, YDR510W, D9719.15, BURPS1710b_A0907 / Plasmid: pET28-HisSMT / Production host:  References: UniProt: Q12306, UniProt: Q3JK38, peptidylprolyl isomerase |
|---|---|
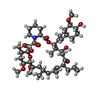
| #2: Chemical | ChemComp-FK5 / |
| #3: Water | ChemComp-HOH / |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 1.71 Å3/Da / Density % sol: 27.96 % |
|---|---|
| Crystal grow | Temperature: 290 K / Method: vapor diffusion, sitting drop / pH: 8.5 Details: Internal tracking number 226593. PACT well H3. 0.2M Sodium Iodide, 0.1M Bis-Tris Propane, pH 8.5, 20.0% w/v PEG3350, 30% PEG400 Cryo. BupsA.00130.a.D24 PD00194 19.6mg/ml., vapor diffusion, ...Details: Internal tracking number 226593. PACT well H3. 0.2M Sodium Iodide, 0.1M Bis-Tris Propane, pH 8.5, 20.0% w/v PEG3350, 30% PEG400 Cryo. BupsA.00130.a.D24 PD00194 19.6mg/ml., vapor diffusion, sitting drop, temperature 290K |
-Data collection
| Diffraction | Mean temperature: 100 K | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ALS ALS  / Beamline: 5.0.1 / Wavelength: 0.9774 Å / Beamline: 5.0.1 / Wavelength: 0.9774 Å | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Detector | Type: ADSC QUANTUM 315r / Detector: CCD / Date: Nov 17, 2011 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation | Monochromator: Si(220) Asymmetric cut single crystal / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation wavelength | Wavelength: 0.9774 Å / Relative weight: 1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection | Resolution: 1.55→40.47 Å / Num. all: 22924 / Num. obs: 22306 / % possible obs: 97.3 % / Observed criterion σ(F): -3 / Observed criterion σ(I): -3 / Redundancy: 3.88 % / Biso Wilson estimate: 19.899 Å2 / Rmerge(I) obs: 0.036 / Net I/σ(I): 24.33 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection shell | Diffraction-ID: 1
|
-Phasing
| Phasing | Method:  molecular replacement molecular replacement |
|---|
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  molecular replacement molecular replacementStarting model: pdb entry 3uqa Resolution: 1.55→40.47 Å / Cor.coef. Fo:Fc: 0.962 / Cor.coef. Fo:Fc free: 0.948 / SU B: 2.519 / SU ML: 0.05 / Cross valid method: THROUGHOUT / σ(F): 0 / σ(I): 2 / ESU R: 0.088 / ESU R Free: 0.086 / Stereochemistry target values: MAXIMUM LIKELIHOOD Details: U VALUES : WITH TLS ADDED HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 14.153 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.55→40.47 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 1.55→1.59 Å / Total num. of bins used: 20
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Origin x: 1.5402 Å / Origin y: 0.7207 Å / Origin z: 20.748 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller














 PDBj
PDBj