[English] 日本語
 Yorodumi
Yorodumi- PDB-3jcc: Structure of Simian Immunodeficiency Virus Envelope Spikes bound ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 3jcc | ||||||
|---|---|---|---|---|---|---|---|
| Title | Structure of Simian Immunodeficiency Virus Envelope Spikes bound with CD4 and Monoclonal Antibody 36D5 | ||||||
 Components Components |
| ||||||
 Keywords Keywords | VIRAL PROTEIN/IMMUNE SYSTEM / Cryoelectron tomography / immunology / AIDS / HIV / VIRAL PROTEIN-IMMUNE SYSTEM complex | ||||||
| Function / homology |  Function and homology information Function and homology informationhelper T cell enhancement of adaptive immune response / interleukin-16 binding / interleukin-16 receptor activity / maintenance of protein location in cell / cellular response to ionomycin / response to methamphetamine hydrochloride / T cell selection / MHC class II protein binding / interleukin-15-mediated signaling pathway / cellular response to granulocyte macrophage colony-stimulating factor stimulus ...helper T cell enhancement of adaptive immune response / interleukin-16 binding / interleukin-16 receptor activity / maintenance of protein location in cell / cellular response to ionomycin / response to methamphetamine hydrochloride / T cell selection / MHC class II protein binding / interleukin-15-mediated signaling pathway / cellular response to granulocyte macrophage colony-stimulating factor stimulus / positive regulation of monocyte differentiation / Alpha-defensins / Nef Mediated CD4 Down-regulation / regulation of T cell activation / response to vitamin D / Other interleukin signaling / T cell receptor complex / extracellular matrix structural constituent / enzyme-linked receptor protein signaling pathway / Translocation of ZAP-70 to Immunological synapse / Phosphorylation of CD3 and TCR zeta chains / Generation of second messenger molecules / macrophage differentiation / T cell differentiation / immunoglobulin binding / Co-inhibition by PD-1 / positive regulation of calcium ion transport into cytosol / Binding and entry of HIV virion / positive regulation of interleukin-2 production / coreceptor activity / positive regulation of T cell proliferation / positive regulation of calcium-mediated signaling / cell surface receptor protein tyrosine kinase signaling pathway / protein tyrosine kinase binding / calcium-mediated signaling / clathrin-coated endocytic vesicle membrane / Vpu mediated degradation of CD4 / MHC class II protein complex binding / response to estradiol / transmembrane signaling receptor activity / Downstream TCR signaling / Cargo recognition for clathrin-mediated endocytosis / T cell receptor signaling pathway / Clathrin-mediated endocytosis / virus receptor activity / signaling receptor activity / clathrin-dependent endocytosis of virus by host cell / defense response to Gram-negative bacterium / adaptive immune response / response to ethanol / early endosome / cell surface receptor signaling pathway / cell adhesion / immune response / membrane raft / endoplasmic reticulum lumen / external side of plasma membrane / fusion of virus membrane with host endosome membrane / viral envelope / symbiont entry into host cell / lipid binding / endoplasmic reticulum membrane / protein kinase binding / virion attachment to host cell / virion membrane / enzyme binding / signal transduction / protein homodimerization activity / zinc ion binding / identical protein binding / plasma membrane Similarity search - Function | ||||||
| Biological species |  Simian immunodeficiency virus Simian immunodeficiency virus Homo sapiens (human) Homo sapiens (human) | ||||||
| Method | ELECTRON MICROSCOPY / electron tomography / cryo EM | ||||||
 Authors Authors | Hu, G. / Liu, J. / Roux, K. / Taylor, K.A. | ||||||
 Citation Citation |  Journal: J Virol / Year: 2017 Journal: J Virol / Year: 2017Title: Structure of Simian Immunodeficiency Virus Envelope Spikes Bound with CD4 and Monoclonal Antibody 36D5. Authors: Guiqing Hu / Jun Liu / Kenneth H Roux / Kenneth A Taylor /  Abstract: The human immunodeficiency virus type 1 (HIV-1)/simian immunodeficiency virus (SIV) envelope spike (Env) mediates viral entry into host cells. The V3 loop of the gp120 component of the Env trimer ...The human immunodeficiency virus type 1 (HIV-1)/simian immunodeficiency virus (SIV) envelope spike (Env) mediates viral entry into host cells. The V3 loop of the gp120 component of the Env trimer contributes to the coreceptor binding site and is a target for neutralizing antibodies. We used cryo-electron tomography to visualize the binding of CD4 and the V3 loop monoclonal antibody (MAb) 36D5 to gp120 of the SIV Env trimer. Our results show that 36D5 binds gp120 at the base of the V3 loop and suggest that the antibody exerts its neutralization effect by blocking the coreceptor binding site. The antibody does this without altering the dynamics of the spike motion between closed and open states when CD4 is bound. The interaction between 36D5 and SIV gp120 is similar to the interaction between some broadly neutralizing anti-V3 loop antibodies and HIV-1 gp120. Two conformations of gp120 bound with CD4 are revealed, suggesting an intrinsic dynamic nature of the liganded Env trimer. CD4 binding substantially increases the binding of 36D5 to gp120 in the intact Env trimer, consistent with CD4-induced changes in the conformation of gp120 and the antibody binding site. Binding by MAb 36D5 does not substantially alter the proportions of the two CD4-bound conformations. The position of MAb 36D5 at the V3 base changes little between conformations, indicating that the V3 base serves as a pivot point during the transition between these two states. Glycoprotein spikes on the surfaces of SIV and HIV are the sole targets available to the immune system for antibody neutralization. Spikes evade the immune system by a combination of a thick layer of polysaccharide on the surface (the glycan shield) and movement between spike domains that masks the epitope conformation. Using SIV virions whose spikes were "decorated" with the primary cellular receptor (CD4) and an antibody (36D5) at part of the coreceptor binding site, we visualized multiple conformations trapped by the rapid freezing step, which were separated using statistical analysis. Our results show that the CD4-induced conformational dynamics of the spike enhances binding of the antibody. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  3jcc.cif.gz 3jcc.cif.gz | 377.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb3jcc.ent.gz pdb3jcc.ent.gz | 302 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  3jcc.json.gz 3jcc.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/jc/3jcc https://data.pdbj.org/pub/pdb/validation_reports/jc/3jcc ftp://data.pdbj.org/pub/pdb/validation_reports/jc/3jcc ftp://data.pdbj.org/pub/pdb/validation_reports/jc/3jcc | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  6543MC  6538C  6539C 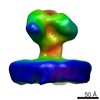 6540C 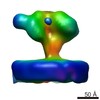 6541C 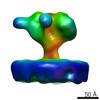 6542C  3jcbC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
| #1: Protein | Mass: 50130.594 Da / Num. of mol.: 3 / Fragment: SEE REMARK 999 / Source method: isolated from a natural source / Source: (natural)  Simian immunodeficiency virus / Strain: SIV239/251tail/Supt-CCR5 CL.30 / References: UniProt: A0A3B6UDT5*PLUS Simian immunodeficiency virus / Strain: SIV239/251tail/Supt-CCR5 CL.30 / References: UniProt: A0A3B6UDT5*PLUS#2: Antibody | | Mass: 22291.643 Da / Num. of mol.: 1 / Fragment: Fab / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) Homo sapiens (human)#3: Antibody | | Mass: 25115.289 Da / Num. of mol.: 1 / Fragment: Fab / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) Homo sapiens (human)#4: Protein | | Mass: 19442.045 Da / Num. of mol.: 1 / Fragment: UNP residues 26-200 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P01730 Homo sapiens (human) / References: UniProt: P01730Has protein modification | Y | Sequence details | ENVELOPE GLYCOPROTE | |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: CELL / 3D reconstruction method: electron tomography |
- Sample preparation
Sample preparation
| Component |
| ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Details of virus | Empty: NO / Enveloped: YES / Host category: VERTEBRATES / Isolate: STRAIN / Type: VIRION | ||||||||||||||||||||
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES | ||||||||||||||||||||
| Vitrification | Instrument: HOMEMADE PLUNGER / Cryogen name: ETHANE / Details: Plunged into liquid ethane. |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Tecnai Polara / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI POLARA 300 / Date: Aug 16, 2008 |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal magnification: 31000 X / Nominal defocus max: 5000 nm / Nominal defocus min: 4000 nm |
| Specimen holder | Specimen holder model: OTHER / Specimen holder type: unidentified / Tilt angle max: 65 ° / Tilt angle min: -65 ° |
| Image recording | Electron dose: 100 e/Å2 / Film or detector model: TVIPS TEMCAM-F415 (4k x 4k) |
- Processing
Processing
| Symmetry | Point symmetry: C1 (asymmetric) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 3D reconstruction | Num. of particles: 1796 / Nominal pixel size: 5.7 Å / Actual pixel size: 5.7 Å / Details: (Subtomogram Averaging--Applied Symmetry: C1) / Symmetry type: POINT | ||||||||||||
| Atomic model building | Protocol: RIGID BODY FIT / Space: REAL | ||||||||||||
| Atomic model building | PDB-ID: 4NCO Accession code: 4NCO / Source name: PDB / Type: experimental model | ||||||||||||
| Refinement step | Cycle: LAST
|
 Movie
Movie Controller
Controller










 PDBj
PDBj




















