| 登録情報 | データベース: PDB / ID: 3dow
|
|---|
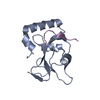
| タイトル | Complex structure of GABA type A receptor associated protein and its binding epitope on calreticulin |
|---|
 要素 要素 | - CRT peptide
- Gamma-aminobutyric acid receptor-associated protein
|
|---|
 キーワード キーワード | PROTEIN TRANSPORT / alpha-beta / beta-grasp fold / Cytoplasm / Cytoskeleton / Golgi apparatus / Membrane / Microtubule / Transport / Calcium / Chaperone / Endoplasmic reticulum / Extracellular matrix / Lectin / Metal-binding / Secreted / Zinc |
|---|
| 機能・相同性 |  機能・相同性情報 機能・相同性情報
response to biphenyl / Calnexin/calreticulin cycle / cytolytic granule / nuclear receptor-mediated glucocorticoid signaling pathway / Assembly of Viral Components at the Budding Site / positive regulation of protein K48-linked ubiquitination / ATF6 (ATF6-alpha) activates chaperone genes / positive regulation of dendritic cell chemotaxis / negative regulation of trophoblast cell migration / cortical granule ...response to biphenyl / Calnexin/calreticulin cycle / cytolytic granule / nuclear receptor-mediated glucocorticoid signaling pathway / Assembly of Viral Components at the Budding Site / positive regulation of protein K48-linked ubiquitination / ATF6 (ATF6-alpha) activates chaperone genes / positive regulation of dendritic cell chemotaxis / negative regulation of trophoblast cell migration / cortical granule / cellular response to electrical stimulus / regulation of meiotic nuclear division / negative regulation of retinoic acid receptor signaling pathway / : / complement component C1q complex binding / endoplasmic reticulum quality control compartment / protein folding in endoplasmic reticulum / regulation of Rac protein signal transduction / sarcoplasmic reticulum lumen / response to peptide / negative regulation of intracellular steroid hormone receptor signaling pathway / nuclear export signal receptor activity / GABA receptor binding / phosphatidylethanolamine binding / cardiac muscle cell differentiation / TBC/RABGAPs / cortical actin cytoskeleton organization / cellular response to nitrogen starvation / response to glycoside / microtubule associated complex / Scavenging by Class A Receptors / Scavenging by Class F Receptors / nuclear androgen receptor binding / cellular response to lithium ion / negative regulation of neuron differentiation / Macroautophagy / response to testosterone / regulation of neurotransmitter receptor localization to postsynaptic specialization membrane / smooth endoplasmic reticulum / hormone binding / autophagosome membrane / extrinsic apoptotic signaling pathway via death domain receptors / molecular sequestering activity / autophagosome maturation / axoneme / protein localization to nucleus / autophagosome assembly / protein targeting / mitophagy / beta-tubulin binding / ERAD pathway / peptide binding / positive regulation of cell cycle / endocytic vesicle lumen / endoplasmic reticulum-Golgi intermediate compartment membrane / positive regulation of substrate adhesion-dependent cell spreading / protein export from nucleus / protein folding chaperone / positive regulation of endothelial cell migration / autophagosome / positive regulation of phagocytosis / acrosomal vesicle / Antigen Presentation: Folding, assembly and peptide loading of class I MHC / lumenal side of endoplasmic reticulum membrane / peptide antigen assembly with MHC class I protein complex / MHC class I peptide loading complex / protein maturation / positive regulation of non-canonical NF-kappaB signal transduction / cellular response to virus / integrin binding / GABA-ergic synapse / microtubule cytoskeleton organization / phagocytic vesicle membrane / intracellular calcium ion homeostasis / cellular senescence / : / response to estradiol / nuclear envelope / positive regulation of proteasomal ubiquitin-dependent protein catabolic process / protein transport / actin cytoskeleton / carbohydrate binding / ER-Phagosome pathway / protein-folding chaperone binding / protein folding / extracellular matrix / cytoplasmic vesicle / sperm midpiece / regulation of apoptotic process / spermatogenesis / microtubule binding / chemical synaptic transmission / microtubule / lysosome / negative regulation of translation / postsynapse / protein stabilization / ribosome / iron ion binding / response to xenobiotic stimulus類似検索 - 分子機能 Calreticulin / Calreticulin family repeated motif signature. / Calreticulin/calnexin / Calreticulin/calnexin, P domain superfamily / Calreticulin/calnexin, conserved site / Calreticulin family / Calreticulin family signature 1. / Calreticulin family signature 2. / Autophagy protein Atg8 ubiquitin-like / Autophagy protein Atg8 ubiquitin like ...Calreticulin / Calreticulin family repeated motif signature. / Calreticulin/calnexin / Calreticulin/calnexin, P domain superfamily / Calreticulin/calnexin, conserved site / Calreticulin family / Calreticulin family signature 1. / Calreticulin family signature 2. / Autophagy protein Atg8 ubiquitin-like / Autophagy protein Atg8 ubiquitin like / Endoplasmic reticulum targeting sequence. / Phosphatidylinositol 3-kinase Catalytic Subunit; Chain A, domain 1 / Ubiquitin-like (UB roll) / Concanavalin A-like lectin/glucanase domain superfamily / Ubiquitin-like domain superfamily / Roll / Alpha Beta類似検索 - ドメイン・相同性 Gamma-aminobutyric acid receptor-associated protein / Calreticulin類似検索 - 構成要素 |
|---|
| 生物種 |  Homo sapiens (ヒト) Homo sapiens (ヒト) |
|---|
| 手法 |  X線回折 / X線回折 /  シンクロトロン / シンクロトロン /  分子置換 / 解像度: 2.3 Å 分子置換 / 解像度: 2.3 Å |
|---|
 データ登録者 データ登録者 | Thielmann, Y. / Weiergraeber, O.H. / Willbold, D. |
|---|
 引用 引用 |  ジャーナル: Febs J. / 年: 2009 ジャーナル: Febs J. / 年: 2009
タイトル: Structural framework of the GABARAP-calreticulin interface - implications for substrate binding to endoplasmic reticulum chaperones.
著者: Thielmann, Y. / Weiergraber, O.H. / Mohrluder, J. / Willbold, D. |
|---|
| 履歴 | | 登録 | 2008年7月7日 | 登録サイト: RCSB / 処理サイト: RCSB |
|---|
| 改定 1.0 | 2009年2月24日 | Provider: repository / タイプ: Initial release |
|---|
| 改定 1.1 | 2011年7月13日 | Group: Version format compliance |
|---|
| 改定 1.2 | 2023年8月30日 | Group: Data collection / Database references ...Data collection / Database references / Derived calculations / Refinement description
カテゴリ: chem_comp_atom / chem_comp_bond ...chem_comp_atom / chem_comp_bond / database_2 / pdbx_initial_refinement_model / struct_conn / struct_conn_type / struct_ref_seq_dif / struct_site
Item: _database_2.pdbx_DOI / _database_2.pdbx_database_accession ..._database_2.pdbx_DOI / _database_2.pdbx_database_accession / _struct_conn.conn_type_id / _struct_conn.id / _struct_conn.pdbx_dist_value / _struct_conn.pdbx_leaving_atom_flag / _struct_conn.ptnr1_auth_asym_id / _struct_conn.ptnr1_auth_comp_id / _struct_conn.ptnr1_auth_seq_id / _struct_conn.ptnr1_label_asym_id / _struct_conn.ptnr1_label_atom_id / _struct_conn.ptnr1_label_comp_id / _struct_conn.ptnr1_label_seq_id / _struct_conn.ptnr2_auth_asym_id / _struct_conn.ptnr2_auth_comp_id / _struct_conn.ptnr2_auth_seq_id / _struct_conn.ptnr2_label_asym_id / _struct_conn.ptnr2_label_atom_id / _struct_conn.ptnr2_label_comp_id / _struct_conn.ptnr2_label_seq_id / _struct_conn_type.id / _struct_ref_seq_dif.details / _struct_site.pdbx_auth_asym_id / _struct_site.pdbx_auth_comp_id / _struct_site.pdbx_auth_seq_id |
|---|
| 改定 1.3 | 2024年10月30日 | Group: Structure summary
カテゴリ: pdbx_entry_details / pdbx_modification_feature |
|---|
|
|---|
 データを開く
データを開く 基本情報
基本情報 要素
要素 キーワード
キーワード 機能・相同性情報
機能・相同性情報 Homo sapiens (ヒト)
Homo sapiens (ヒト) X線回折 /
X線回折 /  シンクロトロン /
シンクロトロン /  分子置換 / 解像度: 2.3 Å
分子置換 / 解像度: 2.3 Å  データ登録者
データ登録者 引用
引用 ジャーナル: Febs J. / 年: 2009
ジャーナル: Febs J. / 年: 2009 構造の表示
構造の表示 Molmil
Molmil Jmol/JSmol
Jmol/JSmol ダウンロードとリンク
ダウンロードとリンク ダウンロード
ダウンロード 3dow.cif.gz
3dow.cif.gz PDBx/mmCIF形式
PDBx/mmCIF形式 pdb3dow.ent.gz
pdb3dow.ent.gz PDB形式
PDB形式 3dow.json.gz
3dow.json.gz PDBx/mmJSON形式
PDBx/mmJSON形式 その他のダウンロード
その他のダウンロード https://data.pdbj.org/pub/pdb/validation_reports/do/3dow
https://data.pdbj.org/pub/pdb/validation_reports/do/3dow ftp://data.pdbj.org/pub/pdb/validation_reports/do/3dow
ftp://data.pdbj.org/pub/pdb/validation_reports/do/3dow
 リンク
リンク 集合体
集合体
 要素
要素 Homo sapiens (ヒト) / 遺伝子: GABARAP, FLC3B / 発現宿主:
Homo sapiens (ヒト) / 遺伝子: GABARAP, FLC3B / 発現宿主: 
 X線回折 / 使用した結晶の数: 1
X線回折 / 使用した結晶の数: 1  試料調製
試料調製 シンクロトロン / サイト:
シンクロトロン / サイト:  ESRF
ESRF  / ビームライン: ID14-1 / 波長: 0.934 Å
/ ビームライン: ID14-1 / 波長: 0.934 Å 解析
解析 分子置換
分子置換 ムービー
ムービー コントローラー
コントローラー













 PDBj
PDBj













