+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2v70 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Third LRR domain of human Slit2 | ||||||
 Components Components | SLIT HOMOLOG 2 PROTEIN N-PRODUCT | ||||||
 Keywords Keywords | STRUCTURAL PROTEIN / NEUROGENESIS / GLYCOPROTEIN / SLIT2 / SECRETED / CHEMOTAXIS / LRR DOMAIN / DIFFERENTIATION / EGF-LIKE DOMAIN / LEUCINE-RICH REPEAT / DEVELOPMENTAL PROTEIN | ||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of leukocyte chemotaxis / corticospinal neuron axon guidance through spinal cord / induction of negative chemotaxis / negative regulation of mononuclear cell migration / apoptotic process involved in luteolysis / negative regulation of monocyte chemotaxis / chemorepulsion involved in postnatal olfactory bulb interneuron migration / negative regulation of retinal ganglion cell axon guidance / Roundabout binding / cellular response to heparin ...negative regulation of leukocyte chemotaxis / corticospinal neuron axon guidance through spinal cord / induction of negative chemotaxis / negative regulation of mononuclear cell migration / apoptotic process involved in luteolysis / negative regulation of monocyte chemotaxis / chemorepulsion involved in postnatal olfactory bulb interneuron migration / negative regulation of retinal ganglion cell axon guidance / Roundabout binding / cellular response to heparin / negative regulation of cellular response to growth factor stimulus / negative regulation of lamellipodium assembly / negative regulation of small GTPase mediated signal transduction / negative regulation of chemokine-mediated signaling pathway / laminin-1 binding / axon extension involved in axon guidance / Netrin-1 signaling / Formation of the ureteric bud / negative regulation of smooth muscle cell chemotaxis / Regulation of commissural axon pathfinding by SLIT and ROBO / negative regulation of smooth muscle cell migration / negative regulation of actin filament polymerization / GTPase inhibitor activity / Role of ABL in ROBO-SLIT signaling / SLIT2:ROBO1 increases RHOA activity / negative regulation of neutrophil chemotaxis / Inactivation of CDC42 and RAC1 / Roundabout signaling pathway / response to cortisol / Signaling by ROBO receptors / pulmonary valve morphogenesis / cell migration involved in sprouting angiogenesis / negative regulation of vascular permeability / motor neuron axon guidance / Activation of RAC1 / proteoglycan binding / negative regulation of endothelial cell migration / aortic valve morphogenesis / retinal ganglion cell axon guidance / ureteric bud development / positive regulation of axonogenesis / negative chemotaxis / branching morphogenesis of an epithelial tube / ventricular septum morphogenesis / negative regulation of protein phosphorylation / cellular response to hormone stimulus / negative regulation of cell migration / axon guidance / negative regulation of cell growth / Regulation of expression of SLITs and ROBOs / heparin binding / positive regulation of apoptotic process / calcium ion binding / protein homodimerization activity / : / extracellular exosome / extracellular region / membrane / identical protein binding / cytoplasm Similarity search - Function | ||||||
| Biological species |  HOMO SAPIENS (human) HOMO SAPIENS (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 3.01 Å MOLECULAR REPLACEMENT / Resolution: 3.01 Å | ||||||
 Authors Authors | Morlot, C. / Cusack, S. / McCarthy, A.A. | ||||||
 Citation Citation |  Journal: Acta Crystallogr.,Sect.D / Year: 2007 Journal: Acta Crystallogr.,Sect.D / Year: 2007Title: Production of Slit2 Lrr Domains in Mammalian Cells for Structural Studies and the Structure of Human Slit2 Domain 3. Authors: Morlot, C. / Hemrika, W. / Romijn, R.A. / Gros, P. / Cusack, S. / Mccarthy, A.A. | ||||||
| History |
| ||||||
| Remark 650 | HELIX DETERMINATION METHOD: AUTHOR PROVIDED. |
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2v70.cif.gz 2v70.cif.gz | 168.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2v70.ent.gz pdb2v70.ent.gz | 133.5 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2v70.json.gz 2v70.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/v7/2v70 https://data.pdbj.org/pub/pdb/validation_reports/v7/2v70 ftp://data.pdbj.org/pub/pdb/validation_reports/v7/2v70 ftp://data.pdbj.org/pub/pdb/validation_reports/v7/2v70 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  1w8aS S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 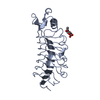
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2 | 
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3 | 
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 4 | 
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unit cell |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Noncrystallographic symmetry (NCS) | NCS domain:
NCS domain segments:
NCS oper:
|
- Components
Components
| #1: Protein | Mass: 24886.449 Da / Num. of mol.: 4 / Fragment: THIRD LRR DOMAIN, RESIDUES 504-714 Source method: isolated from a genetically manipulated source Details: GLYCOSYLATION ON N623 / Source: (gene. exp.)  HOMO SAPIENS (human) / Cell line (production host): HEK293 / Production host: HOMO SAPIENS (human) / Cell line (production host): HEK293 / Production host:  HOMO SAPIENS (human) / References: UniProt: O94813 HOMO SAPIENS (human) / References: UniProt: O94813#2: Sugar | ChemComp-NAG / Has protein modification | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.7 Å3/Da / Density % sol: 54.6 % / Description: NONE |
|---|---|
| Crystal grow | pH: 5.6 Details: 15% PEG 4000, 0.2M NH4 ACETATE, 0.1M NACITRATE, PH=5.6. |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ESRF ESRF  / Beamline: ID14-4 / Wavelength: 0.9393 / Beamline: ID14-4 / Wavelength: 0.9393 |
| Detector | Type: ADSC CCD / Detector: CCD / Date: Apr 22, 2005 / Details: TORODIAL |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.9393 Å / Relative weight: 1 |
| Reflection | Resolution: 3→30 Å / Num. obs: 45165 / % possible obs: 91.9 % / Observed criterion σ(I): 0 / Redundancy: 2.3 % / Rmerge(I) obs: 0.09 / Net I/σ(I): 11.1 |
| Reflection shell | Resolution: 3→3.5 Å / Redundancy: 2.4 % / Rmerge(I) obs: 0.3 / Mean I/σ(I) obs: 3.2 / % possible all: 94.5 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: MODIFIED PDB ENTRY 1W8A Resolution: 3.01→30 Å / Cor.coef. Fo:Fc: 0.911 / Cor.coef. Fo:Fc free: 0.869 / SU B: 50.217 / SU ML: 0.396 / TLS residual ADP flag: LIKELY RESIDUAL / Cross valid method: THROUGHOUT / ESU R Free: 0.501 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.4 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 34.97 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 3.01→30 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller













 PDBj
PDBj












