| 登録情報 | データベース: PDB / ID: 2mgv
|
|---|
| タイトル | NMR structure of PASTA domain of PonA2 from Mycobacterium tuberculosis |
|---|
 要素 要素 | Bifunctional membrane-associated penicillin-binding protein 1A/1B ponA2 |
|---|
 キーワード キーワード | PENICILLIN BINDING PROTEIN / PASTA domain |
|---|
| 機能・相同性 |  機能・相同性情報 機能・相同性情報
peptidoglycan glycosyltransferase / peptidoglycan glycosyltransferase activity / serine-type D-Ala-D-Ala carboxypeptidase / serine-type D-Ala-D-Ala carboxypeptidase activity / penicillin binding / peptidoglycan biosynthetic process / outer membrane-bounded periplasmic space / proteolysis類似検索 - 分子機能 Trypsin Inhibitor V; Chain A - #20 / PASTA domain / PASTA domain / PASTA domain profile. / PASTA / Trypsin Inhibitor V; Chain A / Glycosyl transferase, family 51 / Penicillin binding protein transglycosylase domain / : / Transglycosylase ...Trypsin Inhibitor V; Chain A - #20 / PASTA domain / PASTA domain / PASTA domain profile. / PASTA / Trypsin Inhibitor V; Chain A / Glycosyl transferase, family 51 / Penicillin binding protein transglycosylase domain / : / Transglycosylase / Penicillin-binding protein, transpeptidase / Penicillin binding protein transpeptidase domain / Beta-lactamase/transpeptidase-like / Lysozyme-like domain superfamily / 2-Layer Sandwich / Alpha Beta類似検索 - ドメイン・相同性 |
|---|
| 生物種 |   Mycobacterium tuberculosis (結核菌) Mycobacterium tuberculosis (結核菌) |
|---|
| 手法 | 溶液NMR / torsion angle dynamics |
|---|
| Model details | closest to the average, model1 |
|---|
 データ登録者 データ登録者 | Calvanese, L. / Falcigno, L. / Maglione, C. / Marasco, D. / Ruggiero, A. / Squeglia, F. / Berisio, R. / D'Auria, G. |
|---|
 引用 引用 |  ジャーナル: Biopolymers / 年: 2014 ジャーナル: Biopolymers / 年: 2014
タイトル: Structural and binding properties of the PASTA domain of PonA2, a key penicillin binding protein from Mycobacterium tuberculosis.
著者: Calvanese, L. / Falcigno, L. / Maglione, C. / Marasco, D. / Ruggiero, A. / Squeglia, F. / Berisio, R. / D'Auria, G. |
|---|
| 履歴 | | 登録 | 2013年11月11日 | 登録サイト: BMRB / 処理サイト: RCSB |
|---|
| 改定 1.0 | 2013年12月25日 | Provider: repository / タイプ: Initial release |
|---|
| 改定 1.1 | 2014年5月7日 | Group: Database references |
|---|
| 改定 1.2 | 2023年6月14日 | Group: Data collection / Database references / Other
カテゴリ: database_2 / pdbx_database_status / pdbx_nmr_software
Item: _database_2.pdbx_DOI / _database_2.pdbx_database_accession ..._database_2.pdbx_DOI / _database_2.pdbx_database_accession / _pdbx_database_status.status_code_nmr_data / _pdbx_nmr_software.name |
|---|
| 改定 1.3 | 2024年5月15日 | Group: Data collection / Database references / カテゴリ: chem_comp_atom / chem_comp_bond / database_2 / Item: _database_2.pdbx_DOI |
|---|
|
|---|
 データを開く
データを開く 基本情報
基本情報 要素
要素 キーワード
キーワード 機能・相同性情報
機能・相同性情報
 データ登録者
データ登録者 引用
引用 ジャーナル: Biopolymers / 年: 2014
ジャーナル: Biopolymers / 年: 2014 構造の表示
構造の表示 Molmil
Molmil Jmol/JSmol
Jmol/JSmol ダウンロードとリンク
ダウンロードとリンク ダウンロード
ダウンロード 2mgv.cif.gz
2mgv.cif.gz PDBx/mmCIF形式
PDBx/mmCIF形式 pdb2mgv.ent.gz
pdb2mgv.ent.gz PDB形式
PDB形式 2mgv.json.gz
2mgv.json.gz PDBx/mmJSON形式
PDBx/mmJSON形式 その他のダウンロード
その他のダウンロード https://data.pdbj.org/pub/pdb/validation_reports/mg/2mgv
https://data.pdbj.org/pub/pdb/validation_reports/mg/2mgv ftp://data.pdbj.org/pub/pdb/validation_reports/mg/2mgv
ftp://data.pdbj.org/pub/pdb/validation_reports/mg/2mgv リンク
リンク 集合体
集合体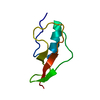
 要素
要素

 試料調製
試料調製 解析
解析 ムービー
ムービー コントローラー
コントローラー













 PDBj
PDBj
 HSQC
HSQC