+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2a06 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Bovine cytochrome bc1 complex with stigmatellin bound | ||||||||||||
 Components Components |
| ||||||||||||
 Keywords Keywords | OXIDOREDUCTASE / CYTOCHROME BC1 / MEMBRANE PROTEIN / HEME PROTEIN / RIESKE IRON SULFUR PROTEIN / CYTOCHROME B / CYTOCHROME C1 / COMPLEX III / MITOCHONDRIAL PROCESSING PROTEASE / UBIQUINONE / REDOX ENZYME / RESPIRATORY CHAIN / STIGMATELLIN | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationComplex III assembly / subthalamus development / pons development / cerebellar Purkinje cell layer development / Respiratory electron transport / pyramidal neuron development / thalamus development / Mitochondrial translation termination / respiratory chain complex III / quinol-cytochrome-c reductase ...Complex III assembly / subthalamus development / pons development / cerebellar Purkinje cell layer development / Respiratory electron transport / pyramidal neuron development / thalamus development / Mitochondrial translation termination / respiratory chain complex III / quinol-cytochrome-c reductase / quinol-cytochrome-c reductase activity / mitochondrial electron transport, ubiquinol to cytochrome c / Mitochondrial protein degradation / hypothalamus development / midbrain development / ubiquinone binding / respiratory electron transport chain / hippocampus development / metalloendopeptidase activity / 2 iron, 2 sulfur cluster binding / mitochondrial membrane / oxidoreductase activity / mitochondrial inner membrane / heme binding / protein-containing complex / mitochondrion / proteolysis / membrane / metal ion binding Similarity search - Function | ||||||||||||
| Biological species |  | ||||||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / MOLECULAR REPLACEMENT USING NATIVE BOVINE, STIGMATELLIN-BOUND CHICKEN STRUCTURES / Resolution: 2.1 Å SYNCHROTRON / MOLECULAR REPLACEMENT USING NATIVE BOVINE, STIGMATELLIN-BOUND CHICKEN STRUCTURES / Resolution: 2.1 Å | ||||||||||||
 Authors Authors | Huang, L.S. / Cobessi, D. / Tung, E.Y. / Berry, E.A. | ||||||||||||
 Citation Citation |  Journal: J.Mol.Biol. / Year: 2005 Journal: J.Mol.Biol. / Year: 2005Title: Binding of the Respiratory Chain Inhibitor Antimycin to the Mitochondrial bc(1) Complex: A New Crystal Structure Reveals an Altered Intramolecular Hydrogen-bonding Pattern. Authors: Huang, L.S. / Cobessi, D. / Tung, E.Y. / Berry, E.A. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2a06.cif.gz 2a06.cif.gz | 865.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2a06.ent.gz pdb2a06.ent.gz | 686.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2a06.json.gz 2a06.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/a0/2a06 https://data.pdbj.org/pub/pdb/validation_reports/a0/2a06 ftp://data.pdbj.org/pub/pdb/validation_reports/a0/2a06 ftp://data.pdbj.org/pub/pdb/validation_reports/a0/2a06 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  1pp9C  1ppjC  1be3S  2bccS C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
| ||||||||
| Noncrystallographic symmetry (NCS) | NCS oper: (Code: given Matrix: (-0.610395, 0.250688, 0.751381), Vector: |
- Components
Components
-Ubiquinol-cytochrome-c reductase complex core protein ... , 2 types, 4 molecules ANBO
| #1: Protein | Mass: 49266.254 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  #2: Protein | Mass: 46575.469 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  |
|---|
-Protein , 2 types, 4 molecules CPDQ
| #3: Protein | Mass: 42620.340 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  #4: Protein | Mass: 27323.277 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  |
|---|
-Ubiquinol-cytochrome c reductase iron-sulfur subunit, ... , 2 types, 4 molecules ERIV
| #5: Protein | Mass: 21640.580 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  #9: Protein | Mass: 7964.259 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  |
|---|
-Ubiquinol-cytochrome c reductase complex ... , 4 types, 8 molecules FSGTHUJW
| #6: Protein | Mass: 13371.190 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  #7: Protein | Mass: 9606.027 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  #8: Protein | Mass: 9189.116 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  #10: Protein | Mass: 7209.311 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  |
|---|
-Sugars , 1 types, 6 molecules 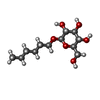
| #11: Sugar | ChemComp-JZR / |
|---|
-Non-polymers , 12 types, 1729 molecules 



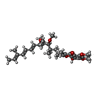
















| #12: Chemical | ChemComp-PO4 / #13: Chemical | ChemComp-AZI / #14: Chemical | ChemComp-UNL / Num. of mol.: 74 / Source method: obtained synthetically #15: Chemical | ChemComp-PEE / #16: Chemical | ChemComp-HEM / #17: Chemical | #18: Chemical | #19: Chemical | ChemComp-CDL / #20: Chemical | ChemComp-GOL / #21: Chemical | #22: Chemical | #23: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.76 Å3/Da / Density % sol: 55.16 % / Description: OPTICAL RESOLUTION 1.70 A |
|---|---|
| Crystal grow | Temperature: 277 K / Method: vapor diffusion, sitting drop / pH: 6.65 Details: PEG-3350, JEFFAMINE, GLYCEROL, CACODYLATE, HEXYLGLUCOSIDE , pH 6.65, VAPOR DIFFUSION, SITTING DROP, temperature 277K |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ALS ALS  / Beamline: 5.0.1 / Wavelength: 1 Å / Beamline: 5.0.1 / Wavelength: 1 Å |
| Detector | Type: ADSC QUANTUM 4 / Detector: CCD / Date: Mar 28, 2002 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1 Å / Relative weight: 1 |
| Reflection | Resolution: 1.99→60 Å / Num. all: 305544 / Num. obs: 305544 / % possible obs: 89.7 % / Observed criterion σ(F): 0 / Observed criterion σ(I): -3 / Redundancy: 2.6 % / Biso Wilson estimate: 29 Å2 / Rsym value: 0.106 / Net I/σ(I): 6.1 |
| Reflection shell | Resolution: 1.99→2.03 Å / Mean I/σ(I) obs: 0.684 / Num. unique all: 8753 / Rsym value: 0.914 / % possible all: 51.9 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure: MOLECULAR REPLACEMENT USING NATIVE BOVINE, STIGMATELLIN-BOUND CHICKEN STRUCTURES Starting model: PDB ENTRIES 1BE3, 2BCC Resolution: 2.1→37.87 Å / Rfactor Rfree error: 0.002 / Data cutoff high absF: 9807698.03 / Data cutoff low absF: 0 / Isotropic thermal model: RESTRAINED / Cross valid method: THROUGHOUT / σ(F): 0 / Stereochemistry target values: MAXIMUM LIKELIHOOD Details: There are sequence link problems in both chain B and chain O. Residue (B GLU 12 ) and residue (B VAL 17 ) are linked together. Residue (O GLU 12 ) and residue (O VAL 17 ) are linked together.
| ||||||||||||||||||||||||||||||||||||
| Solvent computation | Solvent model: FLAT MODEL / Bsol: 74.5142 Å2 / ksol: 0.389315 e/Å3 | ||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 50.3 Å2
| ||||||||||||||||||||||||||||||||||||
| Refine analyze |
| ||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.1→37.87 Å
| ||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||
| Refine LS restraints NCS | NCS model details: CONSTR | ||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.1→2.15 Å / Rfactor Rfree error: 0.014 / Total num. of bins used: 15
| ||||||||||||||||||||||||||||||||||||
| Xplor file |
|
 Movie
Movie Controller
Controller













 PDBj
PDBj

























