[English] 日本語
 Yorodumi
Yorodumi- PDB-1w6m: X-RAY CRYSTAL STRUCTURE OF C2S HUMAN GALECTIN-1 COMPLEXED WITH GA... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1w6m | ||||||
|---|---|---|---|---|---|---|---|
| Title | X-RAY CRYSTAL STRUCTURE OF C2S HUMAN GALECTIN-1 COMPLEXED WITH GALACTOSE | ||||||
 Components Components | (GALECTIN-1) x 2 | ||||||
 Keywords Keywords | SUGAR BINDING PROTEIN / LECTIN / CARBOHYDRATE-BINDING PROTEIN | ||||||
| Function / homology |  Function and homology information Function and homology informationgalectin complex / lactose binding / negative regulation of T-helper 17 cell lineage commitment / myoblast differentiation / plasma cell differentiation / T cell costimulation / laminin binding / Post-translational protein phosphorylation / cell-cell adhesion / Regulation of Insulin-like Growth Factor (IGF) transport and uptake by Insulin-like Growth Factor Binding Proteins (IGFBPs) ...galectin complex / lactose binding / negative regulation of T-helper 17 cell lineage commitment / myoblast differentiation / plasma cell differentiation / T cell costimulation / laminin binding / Post-translational protein phosphorylation / cell-cell adhesion / Regulation of Insulin-like Growth Factor (IGF) transport and uptake by Insulin-like Growth Factor Binding Proteins (IGFBPs) / positive regulation of inflammatory response / extracellular matrix / regulation of apoptotic process / positive regulation of viral entry into host cell / positive regulation of canonical NF-kappaB signal transduction / positive regulation of apoptotic process / endoplasmic reticulum lumen / receptor ligand activity / apoptotic process / : / RNA binding / extracellular exosome / extracellular region / plasma membrane / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |  HOMO SAPIENS (human) HOMO SAPIENS (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 2.3 Å MOLECULAR REPLACEMENT / Resolution: 2.3 Å | ||||||
 Authors Authors | Lopez-Lucendo, M.I.F. / Gabius, H.J. / Romero, A. | ||||||
 Citation Citation |  Journal: J.Mol.Biol. / Year: 2004 Journal: J.Mol.Biol. / Year: 2004Title: Growth-Regulatory Human Galectin-1: Crystallographic Characterisation of the Structural Changes Induced by Single-Site Mutations and Their Impact on the Thermodynamics of Ligand Binding. Authors: Lopez-Lucendo, M.I.F. / Solis, D. / Andre, S. / Hirabayashi, J. / Kasai, K. / Kaltner, H. / Gabius, H.J. / Romero, A. | ||||||
| History |
| ||||||
| Remark 700 | SHEET THE SHEET STRUCTURE OF THIS MOLECULE IS BIFURCATED. IN ORDER TO REPRESENT THIS FEATURE IN ... SHEET THE SHEET STRUCTURE OF THIS MOLECULE IS BIFURCATED. IN ORDER TO REPRESENT THIS FEATURE IN THE SHEET RECORDS BELOW, TWO SHEETS ARE DEFINED. |
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1w6m.cif.gz 1w6m.cif.gz | 70.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1w6m.ent.gz pdb1w6m.ent.gz | 51 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1w6m.json.gz 1w6m.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/w6/1w6m https://data.pdbj.org/pub/pdb/validation_reports/w6/1w6m ftp://data.pdbj.org/pub/pdb/validation_reports/w6/1w6m ftp://data.pdbj.org/pub/pdb/validation_reports/w6/1w6m | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  1gzwSC  1w6nC  1w6oC  1w6pC  1w6qC S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
| ||||||||
| Noncrystallographic symmetry (NCS) | NCS oper: (Code: given Matrix: (-0.3715, -0.35986, -0.85586), Vector: |
- Components
Components
-Protein , 2 types, 2 molecules AB
| #1: Protein | Mass: 14657.447 Da / Num. of mol.: 1 / Mutation: YES Source method: isolated from a genetically manipulated source Source: (gene. exp.)  HOMO SAPIENS (human) / Plasmid: PH14GAL / Production host: HOMO SAPIENS (human) / Plasmid: PH14GAL / Production host:  |
|---|---|
| #2: Protein | Mass: 14641.447 Da / Num. of mol.: 1 / Mutation: YES Source method: isolated from a genetically manipulated source Source: (gene. exp.)  HOMO SAPIENS (human) / Plasmid: PH14GAL / Production host: HOMO SAPIENS (human) / Plasmid: PH14GAL / Production host:  |
-Sugars , 1 types, 2 molecules 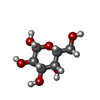
| #5: Sugar |
|---|
-Non-polymers , 3 types, 138 molecules 




| #3: Chemical | ChemComp-BME / #4: Chemical | #6: Water | ChemComp-HOH / | |
|---|
-Details
| Compound details | MAY REGULATE CELL APOPTOSIS AND CELL DIFFERENTIATION. ENGINEERED RESIDUE IN CHAIN A, CYS 2 TO SER ...MAY REGULATE CELL APOPTOSIS AND CELL DIFFERENTI |
|---|---|
| Sequence details | CYS 2 ENGINEERED |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.63 Å3/Da / Density % sol: 52.9 % |
|---|---|
| Crystal grow | Method: vapor diffusion, sitting drop / pH: 5.6 Details: CRYSTALS WERE OBTAINED IN SITTING DROPS BY MIXING EQUAL VOLUMES OF THE PROTEIN SOLUTION (10 MG/ML) AND THE PRECIPITATING BUFFER (2M AMMONIUM SULPHATE AND 1% BETA-MERCAPTO ETHANOL, PH 5.6). ...Details: CRYSTALS WERE OBTAINED IN SITTING DROPS BY MIXING EQUAL VOLUMES OF THE PROTEIN SOLUTION (10 MG/ML) AND THE PRECIPITATING BUFFER (2M AMMONIUM SULPHATE AND 1% BETA-MERCAPTO ETHANOL, PH 5.6). THE GALACTOSE COMPLEX WAS OBTAINED BY SOAKING C2S CRYSTALS FOR 72H IN THE MOTHER LIQUOR SUPPLEMENTED WITH 10 MM OF GALACTOSE |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: ENRAF-NONIUS FR591 / Wavelength: 1.5418 ROTATING ANODE / Type: ENRAF-NONIUS FR591 / Wavelength: 1.5418 |
| Detector | Type: MARRESEARCH / Detector: IMAGE PLATE / Details: OSMIC MIRRORS |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.5418 Å / Relative weight: 1 |
| Reflection | Resolution: 2.3→25.6 Å / Num. obs: 13774 / % possible obs: 96.4 % / Redundancy: 5.1 % / Biso Wilson estimate: 24.6 Å2 / Rmerge(I) obs: 0.08 |
| Reflection shell | Resolution: 2.3→2.45 Å / Redundancy: 4.2 % / Rmerge(I) obs: 0.23 / Mean I/σ(I) obs: 3.1 / % possible all: 94.8 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 1GZW Resolution: 2.3→25.6 Å / Rfactor Rfree error: 0.008 / Data cutoff high absF: 1341364.2 / Isotropic thermal model: RESTRAINED / Cross valid method: THROUGHOUT / σ(F): 0
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Solvent model: FLAT MODEL / Bsol: 33.6187 Å2 / ksol: 0.361353 e/Å3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 22.7 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine analyze |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.3→25.6 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.3→2.44 Å / Rfactor Rfree error: 0.026 / Total num. of bins used: 6
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Xplor file |
|
 Movie
Movie Controller
Controller















 PDBj
PDBj







