+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1svr | ||||||
|---|---|---|---|---|---|---|---|
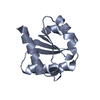
| Title | STRUCTURE OF SEVERIN DOMAIN 2 IN SOLUTION | ||||||
 Components Components | SEVERIN | ||||||
 Keywords Keywords | ACTIN-BINDING | ||||||
| Function / homology |  Function and homology information Function and homology informationactin filament severing activity / Neutrophil degranulation / actin filament fragmentation / actin filament severing / barbed-end actin filament capping / actin polymerization or depolymerization / actin filament bundle assembly / actin monomer binding / phagocytic vesicle / phosphatidylinositol-4,5-bisphosphate binding ...actin filament severing activity / Neutrophil degranulation / actin filament fragmentation / actin filament severing / barbed-end actin filament capping / actin polymerization or depolymerization / actin filament bundle assembly / actin monomer binding / phagocytic vesicle / phosphatidylinositol-4,5-bisphosphate binding / calcium-dependent protein binding / actin filament binding / actin cytoskeleton / calcium ion binding / cytoplasm Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method | SOLUTION NMR | ||||||
 Authors Authors | Schnuchel, A. / Holak, T.A. | ||||||
 Citation Citation |  Journal: J.Mol.Biol. / Year: 1995 Journal: J.Mol.Biol. / Year: 1995Title: Structure of severin domain 2 in solution. Authors: Schnuchel, A. / Wiltscheck, R. / Eichinger, L. / Schleicher, M. / Holak, T.A. #1:  Journal: Biochemistry / Year: 1992 Journal: Biochemistry / Year: 1992Title: Characterization of Actin-and Lipid-Binding Domains in Severin, a Ca(2+)-Dependent F-Actin Fragmenting Protein Authors: Eichinger, L. / Schleicher, M. #2:  Journal: J.Biol.Chem. / Year: 1988 Journal: J.Biol.Chem. / Year: 1988Title: Severin, Gelsolin, and Villin Share a Homologous Sequence in Regions Presumed to Contain F-Actin Severing Domains Authors: Andre, E. / Lottspeich, F. / Schleicher, M. / Noegel, A. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1svr.cif.gz 1svr.cif.gz | 43.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1svr.ent.gz pdb1svr.ent.gz | 30.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1svr.json.gz 1svr.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/sv/1svr https://data.pdbj.org/pub/pdb/validation_reports/sv/1svr ftp://data.pdbj.org/pub/pdb/validation_reports/sv/1svr ftp://data.pdbj.org/pub/pdb/validation_reports/sv/1svr | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein | Mass: 12260.842 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  |
|---|---|
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR |
|---|
- Sample preparation
Sample preparation
| Crystal grow | *PLUS Method: other / Details: NMR |
|---|
- Processing
Processing
| NMR ensemble | Conformers submitted total number: 1 |
|---|
 Movie
Movie Controller
Controller













 PDBj
PDBj

