[English] 日本語
 Yorodumi
Yorodumi- PDB-1kc4: NMR Structural Analysis of the Complex Formed Between alpha-Bunga... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1kc4 | ||||||
|---|---|---|---|---|---|---|---|
| Title | NMR Structural Analysis of the Complex Formed Between alpha-Bungarotoxin and the Principal alpha-Neurotoxin Binding Sequence on the alpha7 Subunit of a Neuronal Nicotinic Acetylcholine Receptor | ||||||
 Components Components |
| ||||||
 Keywords Keywords | TOXIN / alpha-bungarotoxin / nicotinic acetylcholine receptor alpha 7 subunit / alpha-neurotoxin / ligand-gated ion channels / protein-protein interactions / protein-peptide complex | ||||||
| Function / homology |  Function and homology information Function and homology informationacetylcholine receptor activity / acetylcholine-gated channel complex / acetylcholine receptor inhibitor activity / chloride channel regulator activity / acetylcholine-gated monoatomic cation-selective channel activity / ion channel regulator activity / acetylcholine binding / synaptic transmission, cholinergic / negative regulation of tumor necrosis factor production / toxic substance binding ...acetylcholine receptor activity / acetylcholine-gated channel complex / acetylcholine receptor inhibitor activity / chloride channel regulator activity / acetylcholine-gated monoatomic cation-selective channel activity / ion channel regulator activity / acetylcholine binding / synaptic transmission, cholinergic / negative regulation of tumor necrosis factor production / toxic substance binding / negative regulation of cytokine production involved in inflammatory response / response to nicotine / regulation of membrane potential / cognition / positive regulation of angiogenesis / intracellular calcium ion homeostasis / calcium ion transport / amyloid-beta binding / toxin activity / monoatomic ion transmembrane transport / chemical synaptic transmission / perikaryon / response to hypoxia / positive regulation of MAPK cascade / postsynaptic membrane / neuron projection / axon / positive regulation of cell population proliferation / synapse / dendrite / signal transduction / protein homodimerization activity / extracellular region / metal ion binding / plasma membrane / cytoplasm Similarity search - Function | ||||||
| Biological species |   Bungarus multicinctus (many-banded krait) Bungarus multicinctus (many-banded krait) | ||||||
| Method | SOLUTION NMR / distance geometry dynamical simulated annealing | ||||||
 Authors Authors | Moise, L. / Piserchio, A. / Basus, V.J. / Hawrot, E. | ||||||
 Citation Citation |  Journal: J.Biol.Chem. / Year: 2002 Journal: J.Biol.Chem. / Year: 2002Title: NMR structural analysis of alpha-bungarotoxin and its complex with the principal alpha-neurotoxin-binding sequence on the alpha 7 subunit of a neuronal nicotinic acetylcholine receptor. Authors: Moise, L. / Piserchio, A. / Basus, V.J. / Hawrot, E. #1:  Journal: J.Biol.Chem. / Year: 1999 Journal: J.Biol.Chem. / Year: 1999Title: Chimeric analysis of a neuronal nicotinic acetylcholine receptor reveals amino acids conferring sensitivity to alpha-bungarotoxin. Authors: Levandoski, M.M. / Lin, Y. / Moise, L. / McLaughlin, J.T. / Cooper, E. / Hawrot, E. #2:  Journal: J.Biol.Chem. / Year: 1991 Journal: J.Biol.Chem. / Year: 1991Title: Identification of sequence segments forming the alpha-bungarotoxin binding sites on two nicotinic acetylcholine receptor alpha subunits from the avian brain. Authors: McLane, K.E. / Wu, X.D. / Schoepfer, R. / Lindstrom, J.M. / Conti-Tronconi, B.M. #3:  Journal: Biochemistry / Year: 1988 Journal: Biochemistry / Year: 1988Title: Structural studies of alpha-bungarotoxin. 1. Sequence-specific 1H NMR resonance assignments. Authors: Basus, V.J. / Billeter, M. / Love, R.A. / Stroud, R.M. / Kuntz, I.D. | ||||||
| History |
|
- Structure visualization
Structure visualization
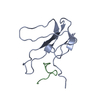
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1kc4.cif.gz 1kc4.cif.gz | 310.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1kc4.ent.gz pdb1kc4.ent.gz | 269.5 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1kc4.json.gz 1kc4.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/kc/1kc4 https://data.pdbj.org/pub/pdb/validation_reports/kc/1kc4 ftp://data.pdbj.org/pub/pdb/validation_reports/kc/1kc4 ftp://data.pdbj.org/pub/pdb/validation_reports/kc/1kc4 | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein | Mass: 8005.281 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Bungarus multicinctus (many-banded krait) / References: UniProt: P60615 Bungarus multicinctus (many-banded krait) / References: UniProt: P60615 |
|---|---|
| #2: Protein/peptide | Mass: 2348.629 Da / Num. of mol.: 1 / Fragment: alpha-neurotoxin binding site Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR experiment |
| ||||||||||||||||||||||||||||
| NMR details | Text: THE WELL-DEFINED REGIONS IN THE COMPLEX, EXHIBITING EXTENSIVE LONG RANGE NOES, ARE: BGTX: 1-16, 22-28, 39-48, AND 54-68 ALPHA 7 19MER: 185-194 |
- Sample preparation
Sample preparation
| Details |
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Sample conditions | pH: 5.5 / Pressure: ambient / Temperature: 308 K | |||||||||
| Crystal grow | *PLUS Method: other / Details: NMR |
-NMR measurement
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Radiation wavelength | Relative weight: 1 | |||||||||||||||
| NMR spectrometer |
|
- Processing
Processing
| NMR software |
| ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method: distance geometry dynamical simulated annealing / Software ordinal: 1 Details: RMS DEVIATIONS FROM IDEAL VALUES: BOND LENGTH(A) 0.0039, ANGLES(DEG) 0.61, IMPROPERS(DEG) 0.53 | ||||||||||||||||||||
| NMR representative | Selection criteria: determined by nmrclust 1.2 | ||||||||||||||||||||
| NMR ensemble | Conformer selection criteria: The submitted conformer models are the 10 with the lowest energy from a pool of over 100 accepted structures that had no restraint violations. Conformers submitted total number: 10 |
 Movie
Movie Controller
Controller




















 PDBj
PDBj


 HSQC
HSQC