+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1ce5 | ||||||
|---|---|---|---|---|---|---|---|
| Title | BOVINE PANCREAS BETA-TRYPSIN IN COMPLEX WITH BENZAMIDINE | ||||||
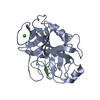
 Components Components | PROTEIN (TRYPSIN) | ||||||
 Keywords Keywords | HYDROLASE / HYDROLASE (SERINE PROTEINASE) | ||||||
| Function / homology |  Function and homology information Function and homology informationtrypsin / serpin family protein binding / serine protease inhibitor complex / digestion / endopeptidase activity / serine-type endopeptidase activity / proteolysis / : / metal ion binding Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 1.9 Å MOLECULAR REPLACEMENT / Resolution: 1.9 Å | ||||||
 Authors Authors | Ota, N. / Stroupe, C. / Ferreira-Da-Silva, J.M.S. / Shah, S.S. / Mares-Guia, M. / Brunger, A.T. | ||||||
 Citation Citation |  Journal: Proteins / Year: 1999 Journal: Proteins / Year: 1999Title: Non-Boltzmann thermodynamic integration (NBTI) for macromolecular systems: relative free energy of binding of trypsin to benzamidine and benzylamine. Authors: Ota, N. / Stroupe, C. / Ferreira-da-Silva, J.M. / Shah, S.A. / Mares-Guia, M. / Brunger, A.T. #1:  Journal: J.Mol.Biol. / Year: 1989 Journal: J.Mol.Biol. / Year: 1989Title: Crystal structure of bovine beta-trypsin at 1.5 A resolution in a crystal form with low molecular packing density. Active site geometry, ion pairs and solvent structure. Authors: Bartunik, H.D. / Summers, L.J. / Bartsch, H.H. #2:  Journal: Acta Crystallogr.,Sect.B / Year: 1983 Journal: Acta Crystallogr.,Sect.B / Year: 1983Title: The Geometry of the Reactive Site and of the Peptide Groups in Trypsin, Trypsinogen and its Complexes with Inhibitors Authors: Marquart, M. / Walter, J. / Deisenhofer, J. / Bode, W. / Huber, R. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1ce5.cif.gz 1ce5.cif.gz | 57.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1ce5.ent.gz pdb1ce5.ent.gz | 40.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1ce5.json.gz 1ce5.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ce/1ce5 https://data.pdbj.org/pub/pdb/validation_reports/ce/1ce5 ftp://data.pdbj.org/pub/pdb/validation_reports/ce/1ce5 ftp://data.pdbj.org/pub/pdb/validation_reports/ce/1ce5 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  2bzaC  3ptbS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 1 molecules A
| #1: Protein | Mass: 23324.287 Da / Num. of mol.: 1 / Source method: isolated from a natural source Details: BOVINE PANCREAS BETA-TRYPSIN PURCHASED FROM WORTHINGTON BIOCHEMICAL CORPORATION Source: (natural)  |
|---|
-Non-polymers , 5 types, 131 molecules 








| #2: Chemical | ChemComp-CA / |
|---|---|
| #3: Chemical | ChemComp-CL / |
| #4: Chemical | ChemComp-SO4 / |
| #5: Chemical | ChemComp-BEN / |
| #6: Water | ChemComp-HOH / |
-Details
| Has protein modification | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.99 Å3/Da / Density % sol: 58.92 % | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | pH: 6 / Details: pH 6.0 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Temperature: 20 ℃ / Method: vapor diffusion, hanging drop | ||||||||||||||||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 293 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU RU200 / Wavelength: 1.5418 ROTATING ANODE / Type: RIGAKU RU200 / Wavelength: 1.5418 |
| Detector | Type: RIGAKU / Detector: IMAGE PLATE / Date: Aug 1, 1997 / Details: MIRRORS |
| Radiation | Monochromator: NI FILTER / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.5418 Å / Relative weight: 1 |
| Reflection | Resolution: 1.9→24 Å / Num. obs: 21208 / % possible obs: 93.4 % / Observed criterion σ(I): 0 / Redundancy: 3.3 % / Biso Wilson estimate: 12.6 Å2 / Rsym value: 0.07 / Net I/σ(I): 19.25 |
| Reflection shell | Resolution: 1.9→1.97 Å / Redundancy: 1.96 % / Mean I/σ(I) obs: 5.1 / Rsym value: 0.201 / % possible all: 84 |
| Reflection | *PLUS Rmerge(I) obs: 0.07 |
| Reflection shell | *PLUS % possible obs: 84 % / Rmerge(I) obs: 0.201 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 3PTB Resolution: 1.9→24 Å / Rfactor Rfree error: 0.004 / Data cutoff high rms absF: 310285.4 / Isotropic thermal model: RESTRAINED / Cross valid method: THROUGHOUT / σ(F): 0
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Solvent model: FLAT MODEL / Bsol: 44.1 Å2 / ksol: 0.366 e/Å3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 22.2 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine analyze |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.9→24 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 1.9→2.02 Å / Rfactor Rfree error: 0.012 / Total num. of bins used: 6
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Xplor file |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Software | *PLUS Name: CNS / Version: 0.3 / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement | *PLUS Lowest resolution: 24 Å / σ(F): 0 / % reflection Rfree: 9.9 % / Rfactor obs: 0.161 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS Biso mean: 22.2 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints | *PLUS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | *PLUS Rfactor Rfree: 0.213 / % reflection Rfree: 9.9 % / Rfactor Rwork: 0.184 / Num. reflection obs: 1881 |
 Movie
Movie Controller
Controller






















 PDBj
PDBj






