+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-9508 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM map of the human 26S proteasome with C1 symmetry | |||||||||
 Map data Map data | overall map at 3.8A, apply C1 symmetry | |||||||||
 Sample Sample |
| |||||||||
| Function / homology |  Function and homology information Function and homology informationthyrotropin-releasing hormone receptor binding / nuclear proteasome complex / host-mediated perturbation of viral transcription / Impaired BRCA2 translocation to the nucleus / Impaired BRCA2 binding to SEM1 (DSS1) / positive regulation of inclusion body assembly / proteasome accessory complex / meiosis I / integrator complex / purine ribonucleoside triphosphate binding ...thyrotropin-releasing hormone receptor binding / nuclear proteasome complex / host-mediated perturbation of viral transcription / Impaired BRCA2 translocation to the nucleus / Impaired BRCA2 binding to SEM1 (DSS1) / positive regulation of inclusion body assembly / proteasome accessory complex / meiosis I / integrator complex / purine ribonucleoside triphosphate binding / proteasome regulatory particle / cytosolic proteasome complex / positive regulation of proteasomal protein catabolic process / proteasome-activating activity / Antigen processing: Ub, ATP-independent proteasomal degradation / proteasome regulatory particle, lid subcomplex / proteasome regulatory particle, base subcomplex / sperm glycocalyx / protein K63-linked deubiquitination / negative regulation of programmed cell death / metal-dependent deubiquitinase activity / Regulation of ornithine decarboxylase (ODC) / Proteasome assembly / proteasome core complex / perinuclear theca / Cross-presentation of soluble exogenous antigens (endosomes) / K63-linked deubiquitinase activity / Somitogenesis / transcription factor binding / Homologous DNA Pairing and Strand Exchange / Defective homologous recombination repair (HRR) due to BRCA1 loss of function / Defective HDR through Homologous Recombination Repair (HRR) due to PALB2 loss of BRCA1 binding function / Defective HDR through Homologous Recombination Repair (HRR) due to PALB2 loss of BRCA2/RAD51/RAD51C binding function / Resolution of D-loop Structures through Synthesis-Dependent Strand Annealing (SDSA) / Resolution of D-loop Structures through Holliday Junction Intermediates / proteasome binding / Impaired BRCA2 binding to RAD51 / myofibril / regulation of protein catabolic process / proteasome storage granule / proteasomal ubiquitin-independent protein catabolic process / sperm head-tail coupling apparatus / positive regulation of RNA polymerase II transcription preinitiation complex assembly / general transcription initiation factor binding / Presynaptic phase of homologous DNA pairing and strand exchange / blastocyst development / protein deubiquitination / polyubiquitin modification-dependent protein binding / immune system process / proteasome endopeptidase complex / NF-kappaB binding / proteasome core complex, beta-subunit complex / endopeptidase activator activity / threonine-type endopeptidase activity / proteasome assembly / mRNA export from nucleus / proteasome core complex, alpha-subunit complex / SARS-CoV-1 targets host intracellular signalling and regulatory pathways / enzyme regulator activity / ERAD pathway / regulation of proteasomal protein catabolic process / inclusion body / TBP-class protein binding / : / ciliary tip / proteasome complex / stem cell differentiation / Regulation of activated PAK-2p34 by proteasome mediated degradation / sarcomere / Autodegradation of Cdh1 by Cdh1:APC/C / APC/C:Cdc20 mediated degradation of Securin / ubiquitin binding / Asymmetric localization of PCP proteins / N-glycan trimming in the ER and Calnexin/Calreticulin cycle / Ubiquitin-dependent degradation of Cyclin D / SCF-beta-TrCP mediated degradation of Emi1 / NIK-->noncanonical NF-kB signaling / AUF1 (hnRNP D0) binds and destabilizes mRNA / TNFR2 non-canonical NF-kB pathway / centriole / sperm end piece / negative regulation of inflammatory response to antigenic stimulus / P-body / Assembly of the pre-replicative complex / Vpu mediated degradation of CD4 / lipopolysaccharide binding / Cdc20:Phospho-APC/C mediated degradation of Cyclin A / Dectin-1 mediated noncanonical NF-kB signaling / Degradation of DVL / Degradation of AXIN / Degradation of CRY and PER proteins / Hh mutants are degraded by ERAD / Activation of NF-kappaB in B cells / G2/M Checkpoints / Degradation of GLI1 by the proteasome / Hedgehog ligand biogenesis / Regulation of RUNX3 expression and activity / Autodegradation of the E3 ubiquitin ligase COP1 / Defective CFTR causes cystic fibrosis / GSK3B and BTRC:CUL1-mediated-degradation of NFE2L2 Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.8 Å | |||||||||
 Authors Authors | Huang XL / Luan B / Wu JP / Shi YG | |||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2016 Journal: Nat Struct Mol Biol / Year: 2016Title: An atomic structure of the human 26S proteasome. Authors: Xiuliang Huang / Bai Luan / Jianping Wu / Yigong Shi /  Abstract: We report the cryo-EM structure of the human 26S proteasome at an average resolution of 3.5 Å, allowing atomic modeling of 28 subunits in the core particle (CP) and 18 subunits in the regulatory ...We report the cryo-EM structure of the human 26S proteasome at an average resolution of 3.5 Å, allowing atomic modeling of 28 subunits in the core particle (CP) and 18 subunits in the regulatory particle (RP). The C-terminal residues of Rpt3 and Rpt5 subunits in the RP can be seen inserted into surface pockets formed between adjacent α subunits in the CP. Each of the six Rpt subunits contains a bound nucleotide, and the central gate of the CP α-ring is closed despite RP association. The six pore 1 loops in the Rpt ring are arranged similarly to a spiral staircase along the axial channel of substrate transport, which is constricted by the pore 2 loops. We also determined the cryo-EM structure of the human proteasome bound to the deubiquitinating enzyme USP14 at 4.35-Å resolution. Together, our structures provide a framework for mechanistic understanding of eukaryotic proteasome function. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_9508.map.gz emd_9508.map.gz | 479 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-9508-v30.xml emd-9508-v30.xml emd-9508.xml emd-9508.xml | 13 KB 13 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_9508.png emd_9508.png | 37.8 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-9508 http://ftp.pdbj.org/pub/emdb/structures/EMD-9508 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9508 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9508 | HTTPS FTP |
-Related structure data
| Related structure data |  9507C  9509C  9510C  9511C  9512C  5gjqC  5gjrC C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_9508.map.gz / Format: CCP4 / Size: 512 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_9508.map.gz / Format: CCP4 / Size: 512 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | overall map at 3.8A, apply C1 symmetry | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.07 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
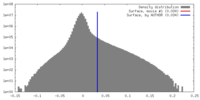
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : human 26S proteasome
| Entire | Name: human 26S proteasome |
|---|---|
| Components |
|
-Supramolecule #1: human 26S proteasome
| Supramolecule | Name: human 26S proteasome / type: complex / ID: 1 / Parent: 0 Details: cryo-EM map of human 26S proteasome by single particle reconstruction |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 2.5 MDa |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1 mg/mL | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 8 Component:
| ||||||||||||
| Grid | Material: COPPER / Support film - Material: CARBON / Support film - topology: CONTINUOUS / Support film - Film thickness: 3.0 nm / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Atmosphere: AIR / Pretreatment - Pressure: 101.325 kPa | ||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 281 K / Instrument: FEI VITROBOT MARK IV / Details: blot for 2 seconds before plunging. | ||||||||||||
| Details | This sample was monodisperse. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Temperature | Min: 70.0 K |
| Details | Preliminary grid screening was performed manually |
| Image recording | Film or detector model: FEI FALCON II (4k x 4k) / Digitization - Dimensions - Width: 4096 pixel / Digitization - Dimensions - Height: 4096 pixel / Digitization - Frames/image: 1-26 / Average exposure time: 1.6 sec. / Average electron dose: 37.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated defocus max: 0.0026 µm / Calibrated defocus min: 0.0016 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller




















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)