+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-9292 | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
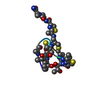
| Title | MicroED structure of thiostrepton at 1.9 A resolution | ||||||||||||||||||
 Map data Map data | 2Fo-Fc map of thiostrepton | ||||||||||||||||||
 Sample Sample |
| ||||||||||||||||||
 Keywords Keywords | ANTIBIOTIC | ||||||||||||||||||
| Function / homology | Thiazolylpeptide-type bacteriocin precursor / Thiopeptide-type bacteriocin precursor / defense response to bacterium / extracellular region / Thiostrepton Function and homology information Function and homology information | ||||||||||||||||||
| Biological species |  Streptomyces azureus (bacteria) Streptomyces azureus (bacteria) | ||||||||||||||||||
| Method | electron crystallography / cryo EM / Resolution: 1.91 Å | ||||||||||||||||||
 Authors Authors | Jones CG / Martynowycz MW | ||||||||||||||||||
| Funding support |  United States, 5 items United States, 5 items
| ||||||||||||||||||
 Citation Citation |  Journal: ACS Cent Sci / Year: 2018 Journal: ACS Cent Sci / Year: 2018Title: The CryoEM Method MicroED as a Powerful Tool for Small Molecule Structure Determination. Authors: Christopher G Jones / Michael W Martynowycz / Johan Hattne / Tyler J Fulton / Brian M Stoltz / Jose A Rodriguez / Hosea M Nelson / Tamir Gonen /  Abstract: In the many scientific endeavors that are driven by organic chemistry, unambiguous identification of small molecules is of paramount importance. Over the past 50 years, NMR and other powerful ...In the many scientific endeavors that are driven by organic chemistry, unambiguous identification of small molecules is of paramount importance. Over the past 50 years, NMR and other powerful spectroscopic techniques have been developed to address this challenge. While almost all of these techniques rely on inference of connectivity, the unambiguous determination of a small molecule's structure requires X-ray and/or neutron diffraction studies. In practice, however, X-ray crystallography is rarely applied in routine organic chemistry due to intrinsic limitations of both the analytes and the technique. Here we report the use of the electron cryo-microscopy (cryoEM) method microcrystal electron diffraction (MicroED) to provide routine and unambiguous structural determination of small organic molecules. From simple powders, with minimal sample preparation, we could collect high-quality MicroED data from nanocrystals (∼100 nm, ∼10 g) resulting in atomic resolution (<1 Å) crystal structures in minutes. | ||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_9292.map.gz emd_9292.map.gz | 255.9 KB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-9292-v30.xml emd-9292-v30.xml emd-9292.xml emd-9292.xml | 16.2 KB 16.2 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_9292.png emd_9292.png | 177.2 KB | ||
| Filedesc metadata |  emd-9292.cif.gz emd-9292.cif.gz | 5.6 KB | ||
| Filedesc structureFactors |  emd_9292_sf.cif.gz emd_9292_sf.cif.gz | 20.4 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-9292 http://ftp.pdbj.org/pub/emdb/structures/EMD-9292 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9292 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9292 | HTTPS FTP |
-Related structure data
| Related structure data |  6mxfMC  9282C  9283C  9284C  9285C  9286C  9287C  9288C  9289C  9290C  9291C C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_9292.map.gz / Format: CCP4 / Size: 363.3 KB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_9292.map.gz / Format: CCP4 / Size: 363.3 KB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | 2Fo-Fc map of thiostrepton | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. generated in cubic-lattice coordinate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X: 0.59589 Å / Y: 0.59589 Å / Z: 0.68834 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 96 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Thiostrepton
| Entire | Name: Thiostrepton |
|---|---|
| Components |
|
-Supramolecule #1: Thiostrepton
| Supramolecule | Name: Thiostrepton / type: organelle_or_cellular_component / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Streptomyces azureus (bacteria) Streptomyces azureus (bacteria) |
| Molecular weight | Theoretical: 1.619538 KDa |
-Macromolecule #1: Thiostrepton
| Macromolecule | Name: Thiostrepton / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Streptomyces azureus (bacteria) Streptomyces azureus (bacteria) |
| Molecular weight | Theoretical: 1.805985 KDa |
| Sequence | String: (QUA)IA(DHA)AS(BB9)T(DBU)(DCY) (TS9)(BB9)T(BB9)(MH6)(BB9)(DHA)(DHA)(NH2) |
-Macromolecule #2: water
| Macromolecule | Name: water / type: ligand / ID: 2 / Number of copies: 2 / Formula: HOH |
|---|---|
| Molecular weight | Theoretical: 18.015 Da |
| Chemical component information |  ChemComp-HOH: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | electron crystallography |
| Aggregation state | 3D array |
- Sample preparation
Sample preparation
| Buffer | pH: 7 / Details: Powder |
|---|---|
| Grid | Model: Quantifoil R2/2 / Material: COPPER / Mesh: 300 |
| Vitrification | Cryogen name: NITROGEN / Chamber humidity: 100 % / Chamber temperature: 298 K / Details: Hand-plunged. |
| Details | Powder |
| Crystal formation | Details: Powder |
- Electron microscopy
Electron microscopy
| Microscope | FEI TALOS ARCTICA |
|---|---|
| Image recording | Film or detector model: FEI CETA (4k x 4k) / Digitization - Dimensions - Width: 2048 pixel / Digitization - Dimensions - Height: 2048 pixel / Number grids imaged: 1 / Number diffraction images: 214 / Average exposure time: 2.21 sec. / Average electron dose: 0.09 e/Å2 / Details: FEI CetaD |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: DIFFRACTION / Camera length: 960 mm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller




 Z (Sec.)
Z (Sec.) X (Row.)
X (Row.) Y (Col.)
Y (Col.)