[English] 日本語
 Yorodumi
Yorodumi- EMDB-21979: Bridging of double-strand DNA break activates PARP2/HPF1 to modif... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-21979 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
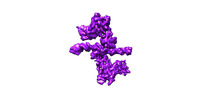
| Title | Bridging of double-strand DNA break activates PARP2/HPF1 to modify chromatin | |||||||||
 Map data Map data | 2xPARP2/HPF1 from PARP2/HPF1_Nucleosome complex | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | DNA repair / PARP1 / PARP2 / HPF1 / ADP-ribosylation / chromatin / histone modifications / GENE REGULATION | |||||||||
| Function / homology |  Function and homology information Function and homology informationregulation of protein ADP-ribosylation / protein ADP-ribosyltransferase-substrate adaptor activity / response to oxygen-glucose deprivation / poly-ADP-D-ribose binding / positive regulation of cell growth involved in cardiac muscle cell development / NAD+-protein-serine ADP-ribosyltransferase activity / NAD DNA ADP-ribosyltransferase activity / DNA ADP-ribosylation / poly-ADP-D-ribose modification-dependent protein binding / HDR through MMEJ (alt-NHEJ) ...regulation of protein ADP-ribosylation / protein ADP-ribosyltransferase-substrate adaptor activity / response to oxygen-glucose deprivation / poly-ADP-D-ribose binding / positive regulation of cell growth involved in cardiac muscle cell development / NAD+-protein-serine ADP-ribosyltransferase activity / NAD DNA ADP-ribosyltransferase activity / DNA ADP-ribosylation / poly-ADP-D-ribose modification-dependent protein binding / HDR through MMEJ (alt-NHEJ) / hippocampal neuron apoptotic process / NAD+ ADP-ribosyltransferase / protein auto-ADP-ribosylation / NAD+-protein-aspartate ADP-ribosyltransferase activity / protein poly-ADP-ribosylation / NAD+-protein-glutamate ADP-ribosyltransferase activity / DNA repair-dependent chromatin remodeling / NAD+-protein mono-ADP-ribosyltransferase activity / decidualization / Transferases; Glycosyltransferases; Pentosyltransferases / POLB-Dependent Long Patch Base Excision Repair / NAD+ poly-ADP-ribosyltransferase activity / site of DNA damage / nucleosome binding / extrinsic apoptotic signaling pathway / nucleotidyltransferase activity / base-excision repair / DNA Damage Recognition in GG-NER / Dual Incision in GG-NER / Formation of Incision Complex in GG-NER / double-strand break repair / histone binding / damaged DNA binding / DNA repair / DNA damage response / chromatin binding / chromatin / nucleolus / nucleoplasm / nucleus / cytosol Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) / synthetic construct (others) Homo sapiens (human) / synthetic construct (others) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 6.3 Å | |||||||||
 Authors Authors | Halic M / Bilokapic S | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2020 Journal: Nature / Year: 2020Title: Bridging of DNA breaks activates PARP2-HPF1 to modify chromatin. Authors: Silvija Bilokapic / Marcin J Suskiewicz / Ivan Ahel / Mario Halic /   Abstract: Breaks in DNA strands recruit the protein PARP1 and its paralogue PARP2 to modify histones and other substrates through the addition of mono- and poly(ADP-ribose) (PAR). In the DNA damage responses, ...Breaks in DNA strands recruit the protein PARP1 and its paralogue PARP2 to modify histones and other substrates through the addition of mono- and poly(ADP-ribose) (PAR). In the DNA damage responses, this post-translational modification occurs predominantly on serine residues and requires HPF1, an accessory factor that switches the amino acid specificity of PARP1 and PARP2 from aspartate or glutamate to serine. Poly(ADP) ribosylation (PARylation) is important for subsequent chromatin decompaction and provides an anchor for the recruitment of downstream signalling and repair factors to the sites of DNA breaks. Here, to understand the molecular mechanism by which PARP enzymes recognize DNA breaks within chromatin, we determined the cryo-electron-microscopic structure of human PARP2-HPF1 bound to a nucleosome. This showed that PARP2-HPF1 bridges two nucleosomes, with the broken DNA aligned in a position suitable for ligation, revealing the initial step in the repair of double-strand DNA breaks. The bridging induces structural changes in PARP2 that signal the recognition of a DNA break to the catalytic domain, which licenses HPF1 binding and PARP2 activation. Our data suggest that active PARP2 cycles through different conformational states to exchange NAD and substrate, which may enable PARP enzymes to act processively while bound to chromatin. The processes of PARP activation and the PARP catalytic cycle we describe can explain mechanisms of resistance to PARP inhibitors and will aid the development of better inhibitors as cancer treatments. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_21979.map.gz emd_21979.map.gz | 49.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-21979-v30.xml emd-21979-v30.xml emd-21979.xml emd-21979.xml | 19.7 KB 19.7 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_21979.png emd_21979.png | 84.2 KB | ||
| Filedesc metadata |  emd-21979.cif.gz emd-21979.cif.gz | 6.8 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-21979 http://ftp.pdbj.org/pub/emdb/structures/EMD-21979 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-21979 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-21979 | HTTPS FTP |
-Related structure data
| Related structure data |  6x0mMC  6wz5C  6wz9C  6x0lC  6x0nC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_21979.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_21979.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | 2xPARP2/HPF1 from PARP2/HPF1_Nucleosome complex | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 2.12 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Histone PARylation factor 1, Poly [ADP-ribose] polymerase 2/DNA C...
| Entire | Name: Histone PARylation factor 1, Poly [ADP-ribose] polymerase 2/DNA Complex |
|---|---|
| Components |
|
-Supramolecule #1: Histone PARylation factor 1, Poly [ADP-ribose] polymerase 2/DNA C...
| Supramolecule | Name: Histone PARylation factor 1, Poly [ADP-ribose] polymerase 2/DNA Complex type: complex / ID: 1 / Parent: 0 / Macromolecule list: all / Details: 2xPARP2/HPF1 from PARP2/HPF1_Nucleosome complex |
|---|---|
| Molecular weight | Theoretical: 100 KDa |
-Supramolecule #2: Histone PARylation factor 1, Poly [ADP-ribose] polymerase 2
| Supramolecule | Name: Histone PARylation factor 1, Poly [ADP-ribose] polymerase 2 type: complex / ID: 2 / Parent: 1 / Macromolecule list: #1-#2 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Supramolecule #3: DNA
| Supramolecule | Name: DNA / type: complex / ID: 3 / Parent: 1 / Macromolecule list: #3-#4 |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
-Macromolecule #1: Histone PARylation factor 1
| Macromolecule | Name: Histone PARylation factor 1 / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 40.598293 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MGHHHHHHGG MVGGGGKRRP GGEGPQCEKT TDVKKSKFCE ADVSSDLRKE VENHYKLSLP EDFYHFWKFC EELDPEKPSD SLSASLGLQ LVGPYDILAG KHKTKKKSTG LNFNLHWRFY YDPPEFQTII IGDNKTQYHM GYFRDSPDEF PVYVGINEAK K NCIIVPNG ...String: MGHHHHHHGG MVGGGGKRRP GGEGPQCEKT TDVKKSKFCE ADVSSDLRKE VENHYKLSLP EDFYHFWKFC EELDPEKPSD SLSASLGLQ LVGPYDILAG KHKTKKKSTG LNFNLHWRFY YDPPEFQTII IGDNKTQYHM GYFRDSPDEF PVYVGINEAK K NCIIVPNG DNVFAAVKLF LTKKLKEITD KKKINLLKNI DEKLTEAARE LGYSLEQRTV KMKQRDKKVV TKTFHGAGLV VP VDKNDVG YRELPETDAD LKRICKTIVE AASDEERLKA FAPIQEMMTF VQFANDECDY GMGLELGMDL FCYGSHYFHK VAG QLLPLA YNLLKRNLFA EIIEEHLANR SQENIDQLAA UniProtKB: Histone PARylation factor 1 |
-Macromolecule #2: Poly [ADP-ribose] polymerase 2
| Macromolecule | Name: Poly [ADP-ribose] polymerase 2 / type: protein_or_peptide / ID: 2 / Number of copies: 2 / Enantiomer: LEVO / EC number: NAD+ ADP-ribosyltransferase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 67.0725 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MGSSHHHHHH SSGLVPRGSH MAARRRRSTG GGRARALNES KRVNNGNTAP EDSSPAKKTR RCQRQESKKM PVAGGKANKD RTEDKQDES VKALLLKGKA PVDPECTAKV GKAHVYCEGN DVYDVMLNQT NLQFNNNKYY LIQLLEDDAQ RNFSVWMRWG R VGKMGQHS ...String: MGSSHHHHHH SSGLVPRGSH MAARRRRSTG GGRARALNES KRVNNGNTAP EDSSPAKKTR RCQRQESKKM PVAGGKANKD RTEDKQDES VKALLLKGKA PVDPECTAKV GKAHVYCEGN DVYDVMLNQT NLQFNNNKYY LIQLLEDDAQ RNFSVWMRWG R VGKMGQHS LVACSGNLNK AKEIFQKKFL DKTKNNWEDR EKFEKVPGKY DMLQMDYATN TQDEEETKKE ESLKSPLKPE SQ LDLRVQE LIKLICNVQA MEEMMMEMKY NTKKAPLGKL TVAQIKAGYQ SLKKIEDCIR AGQHGRALME ACNEFYTRIP HDF GLRTPP LIRTQKELSE KIQLLEALGD IEIAIKLVKT ELQSPEHPLD QHYRNLHCAL RPLDHESYEF KVISQYLQST HAPT HSDYT MTLLDLFEVE KDGEKEAFRE DLHNRMLLWH GSRMSNWVGI LSHGLRIAPP EAPITGYMFG KGIYFADMSS KSANY CFAS RLKNTGLLLL SEVALGQCNE LLEANPKAEG LLQGKHSTKG LGKMAPSSAH FVTLNGSTVP LGPASDTGIL NPDGYT LNY NEYIVYNPNQ VRMRYLLKVQ FNFLQLW UniProtKB: Poly [ADP-ribose] polymerase 2 |
-Macromolecule #3: DNA (167-MER)
| Macromolecule | Name: DNA (167-MER) / type: dna / ID: 3 / Number of copies: 2 / Classification: DNA |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 51.776004 KDa |
| Sequence | String: (DC)(DA)(DA)(DT)(DA)(DC)(DA)(DT)(DG)(DC) (DA)(DC)(DA)(DG)(DG)(DA)(DT)(DG)(DT)(DA) (DT)(DA)(DT)(DA)(DT)(DC)(DT)(DG)(DA) (DC)(DA)(DC)(DG)(DT)(DG)(DC)(DC)(DT)(DG) (DG) (DA)(DG)(DA)(DC)(DT)(DA) ...String: (DC)(DA)(DA)(DT)(DA)(DC)(DA)(DT)(DG)(DC) (DA)(DC)(DA)(DG)(DG)(DA)(DT)(DG)(DT)(DA) (DT)(DA)(DT)(DA)(DT)(DC)(DT)(DG)(DA) (DC)(DA)(DC)(DG)(DT)(DG)(DC)(DC)(DT)(DG) (DG) (DA)(DG)(DA)(DC)(DT)(DA)(DG)(DG) (DG)(DA)(DG)(DT)(DA)(DA)(DT)(DC)(DC)(DC) (DC)(DT) (DT)(DG)(DG)(DC)(DG)(DG)(DT) (DT)(DA)(DA)(DA)(DA)(DC)(DG)(DC)(DG)(DG) (DG)(DG)(DG) (DA)(DC)(DA)(DG)(DC)(DG) (DC)(DG)(DT)(DA)(DC)(DG)(DT)(DG)(DC)(DG) (DT)(DT)(DT)(DA) (DA)(DG)(DC)(DG)(DG) (DT)(DG)(DC)(DT)(DA)(DG)(DA)(DG)(DC)(DT) (DG)(DT)(DC)(DT)(DA) (DC)(DG)(DA)(DC) (DC)(DA)(DA)(DT)(DT)(DG)(DA)(DG)(DC)(DG) (DG)(DC)(DC)(DT)(DC)(DG) (DG)(DC)(DA) (DC)(DC)(DG)(DG)(DG)(DA)(DT)(DT)(DC)(DT) (DC)(DC)(DA)(DG)(DG)(DG)(DC) (DA)(DT) (DC)(DA)(DT)(DA)(DG) |
-Macromolecule #4: DNA (167-MER)
| Macromolecule | Name: DNA (167-MER) / type: dna / ID: 4 / Number of copies: 2 / Classification: DNA |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 51.330684 KDa |
| Sequence | String: (DC)(DT)(DA)(DT)(DG)(DA)(DT)(DG)(DC)(DC) (DC)(DT)(DG)(DG)(DA)(DG)(DA)(DA)(DT)(DC) (DC)(DC)(DG)(DG)(DT)(DG)(DC)(DC)(DG) (DA)(DG)(DG)(DC)(DC)(DG)(DC)(DT)(DC)(DA) (DA) (DT)(DT)(DG)(DG)(DT)(DC) ...String: (DC)(DT)(DA)(DT)(DG)(DA)(DT)(DG)(DC)(DC) (DC)(DT)(DG)(DG)(DA)(DG)(DA)(DA)(DT)(DC) (DC)(DC)(DG)(DG)(DT)(DG)(DC)(DC)(DG) (DA)(DG)(DG)(DC)(DC)(DG)(DC)(DT)(DC)(DA) (DA) (DT)(DT)(DG)(DG)(DT)(DC)(DG)(DT) (DA)(DG)(DA)(DC)(DA)(DG)(DC)(DT)(DC)(DT) (DA)(DG) (DC)(DA)(DC)(DC)(DG)(DC)(DT) (DT)(DA)(DA)(DA)(DC)(DG)(DC)(DA)(DC)(DG) (DT)(DA)(DC) (DG)(DC)(DG)(DC)(DT)(DG) (DT)(DC)(DC)(DC)(DC)(DC)(DG)(DC)(DG)(DT) (DT)(DT)(DT)(DA) (DA)(DC)(DC)(DG)(DC) (DC)(DA)(DA)(DG)(DG)(DG)(DG)(DA)(DT)(DT) (DA)(DC)(DT)(DC)(DC) (DC)(DT)(DA)(DG) (DT)(DC)(DT)(DC)(DC)(DA)(DG)(DG)(DC)(DA) (DC)(DG)(DT)(DG)(DT)(DC) (DA)(DG)(DA) (DT)(DA)(DT)(DA)(DT)(DA)(DC)(DA)(DT)(DC) (DC)(DT)(DG)(DT)(DG)(DC)(DA) (DT)(DG) (DT)(DA)(DT)(DT)(DG) |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.1 mg/mL |
|---|---|
| Buffer | pH: 7.5 |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | #0 - Image recording ID: 1 / #0 - Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / #0 - Detector mode: COUNTING / #0 - Average electron dose: 80.0 e/Å2 / #1 - Image recording ID: 2 / #1 - Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / #1 - Average electron dose: 80.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller


















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)





















