[English] 日本語
 Yorodumi
Yorodumi- EMDB-21383: Cryo-EM Structure of CAP256-VRC26.25 Fab bound to HIV-1 Env trime... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-21383 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM Structure of CAP256-VRC26.25 Fab bound to HIV-1 Env trimer CAP256.wk34.c80 SOSIP.RnS2 | |||||||||
 Map data Map data | Sharpened map | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | V1V2 / VRC26 / CAP256 / HIV-1 / SOSIP / Vaccine / Superinfecting / VIRAL PROTEIN-IMMUNE SYSTEM complex | |||||||||
| Function / homology |  Function and homology information Function and homology informationimmunoglobulin complex / symbiont-mediated perturbation of host defense response / positive regulation of plasma membrane raft polarization / positive regulation of receptor clustering / host cell endosome membrane / clathrin-dependent endocytosis of virus by host cell / adaptive immune response / viral protein processing / fusion of virus membrane with host plasma membrane / fusion of virus membrane with host endosome membrane ...immunoglobulin complex / symbiont-mediated perturbation of host defense response / positive regulation of plasma membrane raft polarization / positive regulation of receptor clustering / host cell endosome membrane / clathrin-dependent endocytosis of virus by host cell / adaptive immune response / viral protein processing / fusion of virus membrane with host plasma membrane / fusion of virus membrane with host endosome membrane / viral envelope / virion attachment to host cell / host cell plasma membrane / virion membrane / structural molecule activity / extracellular region / membrane / plasma membrane Similarity search - Function | |||||||||
| Biological species |   Human immunodeficiency virus 1 / Human immunodeficiency virus 1 /  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.7 Å | |||||||||
 Authors Authors | Gorman J / Kwong PD | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Cell Rep / Year: 2020 Journal: Cell Rep / Year: 2020Title: Structure of Super-Potent Antibody CAP256-VRC26.25 in Complex with HIV-1 Envelope Reveals a Combined Mode of Trimer-Apex Recognition. Authors: Jason Gorman / Gwo-Yu Chuang / Yen-Ting Lai / Chen-Hsiang Shen / Jeffrey C Boyington / Aliaksandr Druz / Hui Geng / Mark K Louder / Krisha McKee / Reda Rawi / Raffaello Verardi / Yongping ...Authors: Jason Gorman / Gwo-Yu Chuang / Yen-Ting Lai / Chen-Hsiang Shen / Jeffrey C Boyington / Aliaksandr Druz / Hui Geng / Mark K Louder / Krisha McKee / Reda Rawi / Raffaello Verardi / Yongping Yang / Baoshan Zhang / Nicole A Doria-Rose / Bob Lin / Penny L Moore / Lynn Morris / Lawrence Shapiro / John R Mascola / Peter D Kwong /   Abstract: Antibodies targeting the V1V2 apex of the HIV-1 envelope (Env) trimer comprise one of the most commonly elicited categories of broadly neutralizing antibodies. Structures of these antibodies indicate ...Antibodies targeting the V1V2 apex of the HIV-1 envelope (Env) trimer comprise one of the most commonly elicited categories of broadly neutralizing antibodies. Structures of these antibodies indicate diverse modes of Env recognition typified by antibodies of the PG9 class and the PGT145 class. The mode of recognition, however, has been unclear for the most potent of the V1V2 apex-targeting antibodies, CAP256-VRC26.25 (named for donor-lineage.clone and referred to hereafter as VRC26.25). Here, we determine the cryoelectron microscopy structure at 3.7 Å resolution of the antigen-binding fragment of VRC26.25 in complex with the Env trimer thought to have initiated the lineage. The 36-residue protruding loop of VRC26.25 displays recognition incorporating both strand-C interactions similar to the PG9 class and V1V2 apex insertion similar to the PGT145 class. Structural elements of separate antibody classes can thus intermingle to form a "combined" class, which in this case yields an antibody of extraordinary potency. | |||||||||
| History |
|
- Structure visualization
Structure visualization
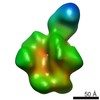
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_21383.map.gz emd_21383.map.gz | 108.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-21383-v30.xml emd-21383-v30.xml emd-21383.xml emd-21383.xml | 24.8 KB 24.8 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_21383.png emd_21383.png | 79 KB | ||
| Masks |  emd_21383_msk_1.map emd_21383_msk_1.map | 115.9 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-21383.cif.gz emd-21383.cif.gz | 7.3 KB | ||
| Others |  emd_21383_additional.map.gz emd_21383_additional.map.gz emd_21383_half_map_1.map.gz emd_21383_half_map_1.map.gz emd_21383_half_map_2.map.gz emd_21383_half_map_2.map.gz | 23.4 MB 107.6 MB 107.6 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-21383 http://ftp.pdbj.org/pub/emdb/structures/EMD-21383 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-21383 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-21383 | HTTPS FTP |
-Related structure data
| Related structure data |  6vttMC  6vrwC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_21383.map.gz / Format: CCP4 / Size: 115.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_21383.map.gz / Format: CCP4 / Size: 115.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Sharpened map | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.0961 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Mask #1
| File |  emd_21383_msk_1.map emd_21383_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Unsharpened map
| File | emd_21383_additional.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Unsharpened map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half map A
| File | emd_21383_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map A | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half map B
| File | emd_21383_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map B | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Cryo-EM Structure of CAP256-VRC26.25 Fab bound to HIV-1 Env trime...
| Entire | Name: Cryo-EM Structure of CAP256-VRC26.25 Fab bound to HIV-1 Env trimer CAP256.wk34.c80 SOSIP.RnS2 |
|---|---|
| Components |
|
-Supramolecule #1: Cryo-EM Structure of CAP256-VRC26.25 Fab bound to HIV-1 Env trime...
| Supramolecule | Name: Cryo-EM Structure of CAP256-VRC26.25 Fab bound to HIV-1 Env trimer CAP256.wk34.c80 SOSIP.RnS2 type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#4 |
|---|
-Supramolecule #2: HIV-1 Env trimer CAP256.wk34.c80 SOSIP.RnS2
| Supramolecule | Name: HIV-1 Env trimer CAP256.wk34.c80 SOSIP.RnS2 / type: complex / ID: 2 / Parent: 1 / Macromolecule list: #1, #4 |
|---|---|
| Source (natural) | Organism:   Human immunodeficiency virus 1 Human immunodeficiency virus 1 |
-Supramolecule #3: CAP256-VRC26.25 Fab
| Supramolecule | Name: CAP256-VRC26.25 Fab / type: complex / ID: 3 / Parent: 1 / Macromolecule list: #2-#3 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Envelope glycoprotein gp120
| Macromolecule | Name: Envelope glycoprotein gp120 / type: protein_or_peptide / ID: 1 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Human immunodeficiency virus 1 Human immunodeficiency virus 1 |
| Molecular weight | Theoretical: 53.322434 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: GLWVTVYYGV PVWREAKTTL FCASDAKSYE KEVHNVWATH ACVPTDPNPQ ELVLENVTEN FNMWKNDMVD QMHEDIISLW DQSLKPCVK LTPLCVTLNC SDAKVNATYK GTREEIKNCS FNATTELRDK KRREYALFYR LDIVPLSGEG NNNSEYRLIN C NTSVITQI ...String: GLWVTVYYGV PVWREAKTTL FCASDAKSYE KEVHNVWATH ACVPTDPNPQ ELVLENVTEN FNMWKNDMVD QMHEDIISLW DQSLKPCVK LTPLCVTLNC SDAKVNATYK GTREEIKNCS FNATTELRDK KRREYALFYR LDIVPLSGEG NNNSEYRLIN C NTSVITQI CPKVTFDPIP IHYCAPAGYA ILKCNNKTFN GTGPCNNVST VQCTHGIKPV VSTQLLLNGS LAEEEIIIRS EN LTDNVKT IIVHLNESVE ITCTRPNNMT RKSVRIGPGQ TFYALGDIIG DIRQPHCNIS EIKWEKTLQR VSEKLREHFN KTI IFNQSS GGDLEITTHS FNCGGEFFYC NTSDLFFNKT FNETYSTGSN STNSTITLPC RIKQIINMWQ EVGRAMYAPP IAGN ITCKS NITGLLLTRD GGGNNSTKET FRPGGGNMRD NWRSELYKYK VVEVKPLGIA PTECNRTVVQ RRRRRR |
-Macromolecule #2: VRC26.25 Heavy Chain
| Macromolecule | Name: VRC26.25 Heavy Chain / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 28.083482 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: QVQLVESGGG VVQPGTSLRL SCAASQFRFD GYGMHWVRQA PGKGLEWVAS ISHDGIKKYH AEKVWGRFTI SRDNSKNTLY LQMNSLRPE DTALYYCAKD LREDECEEWW SD(TYS)(TYS)DFGKQL PCAKSRGGLV GIADNWGQGT MVTVSSASTK GPS VFPLAP ...String: QVQLVESGGG VVQPGTSLRL SCAASQFRFD GYGMHWVRQA PGKGLEWVAS ISHDGIKKYH AEKVWGRFTI SRDNSKNTLY LQMNSLRPE DTALYYCAKD LREDECEEWW SD(TYS)(TYS)DFGKQL PCAKSRGGLV GIADNWGQGT MVTVSSASTK GPS VFPLAP SSKSTSGGTA ALGCLVKDYF PEPVTVSWNS GALTSGVHTF PAVLQSSGLY SLSSVVTVPS SSLGTQTYIC NVNH KPSNT KVDKRVEPKS CDKGLEVLFQ |
-Macromolecule #3: VRC26.25 Light Chain
| Macromolecule | Name: VRC26.25 Light Chain / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 23.052611 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: QSVLTQPPSV SAAPGQKVTI SCSGNTSNIG NNFVSWYQQR PGRAPQLLIY ETDKRPSGIP DRFSASKSGT SGTLAITGLQ TGDEADYYC ATWAASLSSA RVFGTGTKVI VLVQPKANPT VTLFPPSSEE LQANKATLVC LISDFYPGAV TVAWKADSSP V KAGVETTT ...String: QSVLTQPPSV SAAPGQKVTI SCSGNTSNIG NNFVSWYQQR PGRAPQLLIY ETDKRPSGIP DRFSASKSGT SGTLAITGLQ TGDEADYYC ATWAASLSSA RVFGTGTKVI VLVQPKANPT VTLFPPSSEE LQANKATLVC LISDFYPGAV TVAWKADSSP V KAGVETTT PSKQSNNKYA ASSYLSLTPE QWKSHRSYSC QVTHEGSTVE KTVAPTECS UniProtKB: Lambda-chain (AA -20 to 215) |
-Macromolecule #4: Envelope glycoprotein gp41
| Macromolecule | Name: Envelope glycoprotein gp41 / type: protein_or_peptide / ID: 4 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Human immunodeficiency virus 1 Human immunodeficiency virus 1 |
| Molecular weight | Theoretical: 17.355703 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: AVVGLGAVFL GFLGAAGSTM GAASNTLTVQ ARQLLSGIVQ QQSNLLRAPE AQQHMLQLGV WGFKQLQARV LAIERYLEVQ QLLGMWGCS GKLICCTNVP WNSSWSNKTY NEIWDNMTWM QWDREIGNYT DTIYKLLEVS QFQQEINEKD NLTLD |
-Macromolecule #11: 2-acetamido-2-deoxy-beta-D-glucopyranose
| Macromolecule | Name: 2-acetamido-2-deoxy-beta-D-glucopyranose / type: ligand / ID: 11 / Number of copies: 41 / Formula: NAG |
|---|---|
| Molecular weight | Theoretical: 221.208 Da |
| Chemical component information |  ChemComp-NAG: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 2 mg/mL |
|---|---|
| Buffer | pH: 7.4 / Component - Formula: PBS |
| Grid | Model: C-flat-1.2/1.3 4C / Material: COPPER / Support film - Material: CARBON / Support film - topology: HOLEY |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 90 % / Chamber temperature: 293 K / Instrument: FEI VITROBOT MARK IV |
| Details | Cryo-EM Structure of Fab CAP256-VRC26.25 bound to HIV-1 Env trimer CAP256 SU.wk34.80 SOSIP.RnS2 |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Number grids imaged: 1 / Number real images: 1777 / Average exposure time: 10.0 sec. / Average electron dose: 64.48 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 70.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model |
| ||||||
|---|---|---|---|---|---|---|---|
| Refinement | Space: REAL / Protocol: FLEXIBLE FIT | ||||||
| Output model |  PDB-6vtt: |
 Movie
Movie Controller
Controller














 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)