[English] 日本語
 Yorodumi
Yorodumi- EMDB-2077: Electron cryo-microscopy of microtubule-bound human kinesin-5 mot... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-2077 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
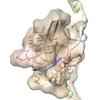
| Title | Electron cryo-microscopy of microtubule-bound human kinesin-5 motor domain in AMPPNP state. | |||||||||
 Map data Map data | 3D reconstruction of microtubule-bound human kinesin-5 motor domain with AMPPNP bound in the nucleotide-binding site | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | cancer / electron cryo-microscopy / kinesin / microtubule / mitosis | |||||||||
| Function / homology |  Function and homology information Function and homology informationspindle elongation / regulation of mitotic centrosome separation / plus-end-directed microtubule motor activity / mitotic centrosome separation / Kinesins / positive regulation of axon guidance / spindle organization / microtubule motor activity / kinesin complex / COPI-dependent Golgi-to-ER retrograde traffic ...spindle elongation / regulation of mitotic centrosome separation / plus-end-directed microtubule motor activity / mitotic centrosome separation / Kinesins / positive regulation of axon guidance / spindle organization / microtubule motor activity / kinesin complex / COPI-dependent Golgi-to-ER retrograde traffic / microtubule-based movement / mitotic spindle assembly / microtubule-based process / MHC class II antigen presentation / mitotic spindle organization / sperm end piece / structural constituent of cytoskeleton / microtubule cytoskeleton organization / spindle / neuron migration / spindle pole / mitotic spindle / mitotic cell cycle / sperm principal piece / microtubule cytoskeleton / sperm midpiece / microtubule binding / Hydrolases; Acting on acid anhydrides; Acting on GTP to facilitate cellular and subcellular movement / microtubule / ciliary basal body / protein heterodimerization activity / cell division / hydrolase activity / GTPase activity / protein kinase binding / GTP binding / protein-containing complex / ATP binding / membrane / metal ion binding / nucleus / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 9.7 Å | |||||||||
 Authors Authors | Goulet A / Behnke-Parks WM / Sindelar C / Rosenfeld S / Moores C | |||||||||
 Citation Citation |  Journal: J Biol Chem / Year: 2012 Journal: J Biol Chem / Year: 2012Title: The structural basis of force generation by the mitotic motor kinesin-5. Authors: Adeline Goulet / William M Behnke-Parks / Charles V Sindelar / Jennifer Major / Steven S Rosenfeld / Carolyn A Moores /  Abstract: Kinesin-5 is required for forming the bipolar spindle during mitosis. Its motor domain, which contains nucleotide and microtubule binding sites and mechanical elements to generate force, has evolved ...Kinesin-5 is required for forming the bipolar spindle during mitosis. Its motor domain, which contains nucleotide and microtubule binding sites and mechanical elements to generate force, has evolved distinct properties for its spindle-based functions. In this study, we report subnanometer resolution cryoelectron microscopy reconstructions of microtubule-bound human kinesin-5 before and after nucleotide binding and combine this information with studies of the kinetics of nucleotide-induced neck linker and cover strand movement. These studies reveal coupled, nucleotide-dependent conformational changes that explain many of this motor's properties. We find that ATP binding induces a ratchet-like docking of the neck linker and simultaneous, parallel docking of the N-terminal cover strand. Loop L5, the binding site for allosteric inhibitors of kinesin-5, also undergoes a dramatic reorientation when ATP binds, suggesting that it is directly involved in controlling nucleotide binding. Our structures indicate that allosteric inhibitors of human kinesin-5, which are being developed as anti-cancer therapeutics, bind to a motor conformation that occurs in the course of normal function. However, due to evolutionarily defined sequence variations in L5, this conformation is not adopted by invertebrate kinesin-5s, explaining their resistance to drug inhibition. Together, our data reveal the precision with which the molecular mechanism of kinesin-5 motors has evolved for force generation. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_2077.map.gz emd_2077.map.gz | 238.1 KB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-2077-v30.xml emd-2077-v30.xml emd-2077.xml emd-2077.xml | 12.3 KB 12.3 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_2077.jpg emd_2077.jpg | 144.6 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-2077 http://ftp.pdbj.org/pub/emdb/structures/EMD-2077 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2077 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2077 | HTTPS FTP |
-Related structure data
| Related structure data |  4aqvMC  2078C  2079C  2080C  2081C  2152C  4aqwC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_2077.map.gz / Format: CCP4 / Size: 348.6 KB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_2077.map.gz / Format: CCP4 / Size: 348.6 KB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | 3D reconstruction of microtubule-bound human kinesin-5 motor domain with AMPPNP bound in the nucleotide-binding site | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 2.8 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : 13-protofilament microtubule-bound human kinesin-5 motor domain w...
| Entire | Name: 13-protofilament microtubule-bound human kinesin-5 motor domain with AMPPNP |
|---|---|
| Components |
|
-Supramolecule #1000: 13-protofilament microtubule-bound human kinesin-5 motor domain w...
| Supramolecule | Name: 13-protofilament microtubule-bound human kinesin-5 motor domain with AMPPNP type: sample / ID: 1000 Oligomeric state: 13-protofilament microtubule with one kineisn-5 motor domain bound every tubulin heterodimers Number unique components: 2 |
|---|
-Macromolecule #1: alpha-beta tubulin dimer
| Macromolecule | Name: alpha-beta tubulin dimer / type: protein_or_peptide / ID: 1 / Oligomeric state: heterodimer / Recombinant expression: No / Database: NCBI |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #2: Kinesin-5 motor domain
| Macromolecule | Name: Kinesin-5 motor domain / type: protein_or_peptide / ID: 2 / Oligomeric state: monomer / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / synonym: Human Homo sapiens (human) / synonym: Human |
| Recombinant expression | Organism:  |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 6.8 / Details: 80 mM PIPES, 5 mM MgCl2, 1 mM EGTA, 5mM AMPPNP |
|---|---|
| Grid | Details: 400 mesh holey carbon grids |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Instrument: FEI VITROBOT MARK I / Method: chamber at 24 degrees C, blot 2.5 sec |
- Electron microscopy
Electron microscopy
| Microscope | FEI TECNAI F20 |
|---|---|
| Temperature | Average: 90 K |
| Alignment procedure | Legacy - Astigmatism: Objective lens astigmatism was corrected at 150,000 times magnification |
| Date | Jan 10, 2011 |
| Image recording | Category: FILM / Film or detector model: KODAK SO-163 FILM / Digitization - Scanner: ZEISS SCAI / Digitization - Sampling interval: 7 µm / Number real images: 46 / Average electron dose: 18 e/Å2 / Bits/pixel: 8 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.0 mm / Nominal defocus max: 2.2 µm / Nominal defocus min: 0.7 µm / Nominal magnification: 50000 |
| Sample stage | Specimen holder model: GATAN LIQUID NITROGEN |
| Experimental equipment |  Model: Tecnai F20 / Image courtesy: FEI Company |
- Image processing
Image processing
| Details | The particles were selected along individual microtubules. |
|---|---|
| CTF correction | Details: FREALIGN |
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 9.7 Å / Resolution method: FSC 0.5 CUT-OFF / Software - Name: SPIDER, FREALIGN Details: Approximately 50,000 asymmetric units were averaged in the final reconstruction. Number images used: 3587 |
-Atomic model buiding 1
| Initial model | PDB ID: Chain - Chain ID: A |
|---|---|
| Software | Name: Chimera, FlexEM |
| Details | Protocol: rigid body then flexible fitting. The domain was fitted as a rigid body. The N-terminal residues 6 to 16 were built in the EM map and the final model was refined by flexible fitting. |
| Refinement | Space: REAL / Protocol: FLEXIBLE FIT / Target criteria: cross-correlation |
| Output model |  PDB-4aqv: |
-Atomic model buiding 2
| Initial model | PDB ID: Chain - #0 - Chain ID: A / Chain - #1 - Chain ID: B |
|---|---|
| Software | Name:  Chimera Chimera |
| Details | Protocol: rigid body. alpha- and b-tubulin were separately fitted. |
| Refinement | Space: REAL / Protocol: RIGID BODY FIT / Target criteria: cross-correlation |
| Output model |  PDB-4aqv: |
 Movie
Movie Controller
Controller



















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)























