[English] 日本語
 Yorodumi
Yorodumi- EMDB-1418: Binding of a neutralizing antibody to dengue virus alters the arr... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-1418 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
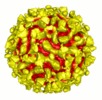
| Title | Binding of a neutralizing antibody to dengue virus alters the arrangement of surface glycoproteins | |||||||||
 Map data Map data | This is a map of Fab 1A1D-2 complexed with Dengue virus | |||||||||
 Sample Sample |
| |||||||||
| Function / homology |  Function and homology information Function and homology informationimmunoglobulin complex / viral nucleocapsid / clathrin-dependent endocytosis of virus by host cell / adaptive immune response / protein dimerization activity / symbiont-mediated suppression of host innate immune response / host cell endoplasmic reticulum membrane / fusion of virus membrane with host endosome membrane / viral envelope / virion attachment to host cell ...immunoglobulin complex / viral nucleocapsid / clathrin-dependent endocytosis of virus by host cell / adaptive immune response / protein dimerization activity / symbiont-mediated suppression of host innate immune response / host cell endoplasmic reticulum membrane / fusion of virus membrane with host endosome membrane / viral envelope / virion attachment to host cell / virion membrane / extracellular region Similarity search - Function | |||||||||
| Biological species |   Dengue virus 2 Dengue virus 2 | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 24.0 Å | |||||||||
 Authors Authors | Lok SM | |||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2008 Journal: Nat Struct Mol Biol / Year: 2008Title: Binding of a neutralizing antibody to dengue virus alters the arrangement of surface glycoproteins. Authors: Shee-Mei Lok / Victor Kostyuchenko / Grant E Nybakken / Heather A Holdaway / Anthony J Battisti / Soila Sukupolvi-Petty / Dagmar Sedlak / Daved H Fremont / Paul R Chipman / John T Roehrig / ...Authors: Shee-Mei Lok / Victor Kostyuchenko / Grant E Nybakken / Heather A Holdaway / Anthony J Battisti / Soila Sukupolvi-Petty / Dagmar Sedlak / Daved H Fremont / Paul R Chipman / John T Roehrig / Michael S Diamond / Richard J Kuhn / Michael G Rossmann /  Abstract: The monoclonal antibody 1A1D-2 has been shown to strongly neutralize dengue virus serotypes 1, 2 and 3, primarily by inhibiting attachment to host cells. A crystal structure of its antigen binding ...The monoclonal antibody 1A1D-2 has been shown to strongly neutralize dengue virus serotypes 1, 2 and 3, primarily by inhibiting attachment to host cells. A crystal structure of its antigen binding fragment (Fab) complexed with domain III of the viral envelope glycoprotein, E, showed that the epitope would be partially occluded in the known structure of the mature dengue virus. Nevertheless, antibody could bind to the virus at 37 degrees C, suggesting that the virus is in dynamic motion making hidden epitopes briefly available. A cryo-electron microscope image reconstruction of the virus:Fab complex showed large changes in the organization of the E protein that exposed the epitopes on two of the three E molecules in each of the 60 icosahedral asymmetric units of the virus. The changes in the structure of the viral surface are presumably responsible for inhibiting attachment to cells. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_1418.map.gz emd_1418.map.gz | 59.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-1418-v30.xml emd-1418-v30.xml emd-1418.xml emd-1418.xml | 11.7 KB 11.7 KB | Display Display |  EMDB header EMDB header |
| Images |  1418.gif 1418.gif | 118.9 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-1418 http://ftp.pdbj.org/pub/emdb/structures/EMD-1418 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-1418 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-1418 | HTTPS FTP |
-Related structure data
| Related structure data |  2r6pMC  2r29C  2r69C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_1418.map.gz / Format: CCP4 / Size: 89 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_1418.map.gz / Format: CCP4 / Size: 89 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | This is a map of Fab 1A1D-2 complexed with Dengue virus | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 2.74 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
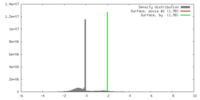
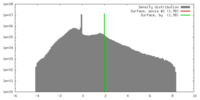
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Fab Fragment of MAb 1A1D-2 complexed with Dengue 2 virus
| Entire | Name: Fab Fragment of MAb 1A1D-2 complexed with Dengue 2 virus |
|---|---|
| Components |
|
-Supramolecule #1000: Fab Fragment of MAb 1A1D-2 complexed with Dengue 2 virus
| Supramolecule | Name: Fab Fragment of MAb 1A1D-2 complexed with Dengue 2 virus type: sample / ID: 1000 Oligomeric state: The Fab molecule binds to two of the three E proteins in the icosahedral assymmetric unit Number unique components: 2 |
|---|
-Supramolecule #1: Dengue virus 2
| Supramolecule | Name: Dengue virus 2 / type: virus / ID: 1 / Name.synonym: DENV / NCBI-ID: 11060 / Sci species name: Dengue virus 2 / Virus type: VIRION / Virus isolate: SEROTYPE / Virus enveloped: Yes / Virus empty: No / Syn species name: DENV |
|---|---|
| Host (natural) | Organism:  Homo sapiens (human) / synonym: VERTEBRATES Homo sapiens (human) / synonym: VERTEBRATES |
| Virus shell | Shell ID: 1 / Name: envelope / Diameter: 240 Å / T number (triangulation number): 3 |
-Macromolecule #1: Fab fragment of MAb 1A1D-2
| Macromolecule | Name: Fab fragment of MAb 1A1D-2 / type: protein_or_peptide / ID: 1 / Name.synonym: 1A1D-2 / Number of copies: 2 / Oligomeric state: heterodimer / Recombinant expression: No |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Experimental: 50 MDa / Theoretical: 50 MDa |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.6 mg/mL |
|---|---|
| Buffer | pH: 7.6 / Details: 12 mM Tris-HCl, 120 mM NaCl, 1 mM EDTA |
| Grid | Details: 400 mesh copper grid |
| Vitrification | Cryogen name: ETHANE / Instrument: HOMEMADE PLUNGER Details: Vitrification instrument: Guillotine-style plunge freezeing device Method: A small vial of ethane is placed inside a larger liquid nitrogen reservoir. The grid holding a few microliters of the sample is held in place at the bottom of a plunger by the means of fine ...Method: A small vial of ethane is placed inside a larger liquid nitrogen reservoir. The grid holding a few microliters of the sample is held in place at the bottom of a plunger by the means of fine tweezers. Once the ethane in the vial is completely frozen, it needs to be slightly melted. When the liquid ethane is ready, a piece of filter paper is then pressed against the sample to blot of excess buffer, sufficient to leave a thin layer on the grid. After a predetermined time, the filter paper is removed, and the plunger is allowed to drop into the liquid ethane. Once the grid enters the liquid ethane, the sample is rapidly frozen, and the grid is transferred under liquid nitrogen to a storage box immersed liquid nitrogen for later use in the microscope. |
- Electron microscopy
Electron microscopy
| Microscope | FEI/PHILIPS CM200T |
|---|---|
| Temperature | Average: 98 K |
| Alignment procedure | Legacy - Astigmatism: live FFT |
| Details | low dose |
| Date | Mar 21, 2007 |
| Image recording | Category: CCD / Film or detector model: KODAK SO-163 FILM / Digitization - Sampling interval: 7 µm / Average electron dose: 24 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated magnification: 51040 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.0 mm / Nominal defocus max: 3.37 µm / Nominal defocus min: 2.27 µm / Nominal magnification: 50000 |
| Sample stage | Specimen holder: Eucentric / Specimen holder model: GATAN LIQUID NITROGEN |
- Image processing
Image processing
| CTF correction | Details: each particle |
|---|---|
| Final reconstruction | Applied symmetry - Point group: I (icosahedral) / Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 24.0 Å / Resolution method: FSC 0.5 CUT-OFF / Software - Name: spider,XMIPP / Number images used: 2885 |
-Atomic model buiding 1
| Initial model | PDB ID: Chain - Chain ID - 0: 2 / Chain - Chain ID - 1: R / Chain - Chain ID - 2: 2 / Chain - Chain ID - 3: 9 |
|---|---|
| Software | Name: EMFIT |
| Details | Protocol: Rigid body |
| Refinement | Protocol: RIGID BODY FIT |
| Output model |  PDB-2r6p: |
 Movie
Movie Controller
Controller










 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)