[English] 日本語
 Yorodumi
Yorodumi- EMDB-1367: Three-dimensional structure of a voltage-gated potassium channel ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-1367 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Three-dimensional structure of a voltage-gated potassium channel at 2.5 nm resolution. | |||||||||
 Map data Map data | This is an image of surface rendered view of Shaker potassium voltage-gated channel | |||||||||
 Sample Sample |
| |||||||||
| Function / homology |  Function and homology information Function and homology informationdelayed rectifier potassium channel activity / action potential / voltage-gated potassium channel activity / voltage-gated potassium channel complex / protein homooligomerization / identical protein binding Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / negative staining / Resolution: 25.0 Å | |||||||||
 Authors Authors | Sokolova O / Kolmakova-Partensky L / Grigorieff N | |||||||||
 Citation Citation |  Journal: Structure / Year: 2001 Journal: Structure / Year: 2001Title: Three-dimensional structure of a voltage-gated potassium channel at 2.5 nm resolution. Authors: O Sokolova / L Kolmakova-Partensky / N Grigorieff /  Abstract: BACKGROUND: The voltage-gated potassium channel Shaker from Drosophila consists of a tetramer of identical subunits, each containing six transmembrane segments. The atomic structure of a bacterial ...BACKGROUND: The voltage-gated potassium channel Shaker from Drosophila consists of a tetramer of identical subunits, each containing six transmembrane segments. The atomic structure of a bacterial homolog, the potassium channel KcsA, is much smaller than Shaker. It does not have a voltage sensor and other important domains like the N-terminal tetramerization (T1) domain. The structure of these additional elements has to be studied in the more complex voltage-gated channels. RESULTS: We determined the three-dimensional structure of the entire Shaker channel at 2.5 nm resolution using electron microscopy. The four-fold symmetric structure shows a large and a small domain ...RESULTS: We determined the three-dimensional structure of the entire Shaker channel at 2.5 nm resolution using electron microscopy. The four-fold symmetric structure shows a large and a small domain linked by thin 2 nm long connectors. To interpret the structure, we used the crystal structures of the isolated T1 domain and the KcsA channel. A unique density assignment was made based on the symmetry and dimensions of the crystal structures and domains, identifying the smaller domain as the cytoplasmic mass of Shaker containing T1 and the larger domain as embedded in the membrane. CONCLUSIONS: The two-domain architecture of the Shaker channel is consistent with the recently proposed "hanging gondola" model for the T1 domain, putting the T1 domain at a distance from the ...CONCLUSIONS: The two-domain architecture of the Shaker channel is consistent with the recently proposed "hanging gondola" model for the T1 domain, putting the T1 domain at a distance from the membrane domain but attached to it by thin connectors. The space between the two domains is sufficient to permit cytoplasmic access of ions and the N-terminal inactivation domain to the pore region. A hanging gondola architecture has also been observed in the nicotinic acetylcholine receptor and the KcsA structure, suggesting that it is a common element of ion channels. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_1367.map.gz emd_1367.map.gz | 908.6 KB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-1367-v30.xml emd-1367-v30.xml emd-1367.xml emd-1367.xml | 9.8 KB 9.8 KB | Display Display |  EMDB header EMDB header |
| Images |  1367.gif 1367.gif | 47.8 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-1367 http://ftp.pdbj.org/pub/emdb/structures/EMD-1367 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-1367 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-1367 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_1367.map.gz / Format: CCP4 / Size: 1001 KB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_1367.map.gz / Format: CCP4 / Size: 1001 KB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | This is an image of surface rendered view of Shaker potassium voltage-gated channel | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 3.5 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
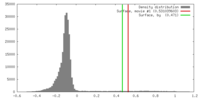
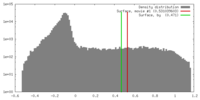
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Shaker B channel
| Entire | Name: Shaker B channel |
|---|---|
| Components |
|
-Supramolecule #1000: Shaker B channel
| Supramolecule | Name: Shaker B channel / type: sample / ID: 1000 Details: This is an inactivation-removed Shaker B channel construct, delta6-46/F425G, with a C-terminal 1D4 immunoaffinity tag. The molecular weight given above includes glycosylation. Oligomeric state: One homotetramer / Number unique components: 1 |
|---|---|
| Molecular weight | Experimental: 860 KDa / Theoretical: 860 KDa / Method: SDS PAGE electrophoresis |
-Macromolecule #1: Kv1.1
| Macromolecule | Name: Kv1.1 / type: protein_or_peptide / ID: 1 / Name.synonym: Shaker Kv channel / Number of copies: 4 / Oligomeric state: Tetramer / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Experimental: 860 KDa / Theoretical: 860 KDa |
| Recombinant expression | Organism: Mammalian (mammals) / Recombinant plasmid: pmt3 |
-Experimental details
-Structure determination
| Method | negative staining, cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 Details: 80 mM KCl, 2 mM NaEDTA, 40 mM HEPES-KOH, 0.7% CHAPS, 1 mM DTT, 50 mM NaCl, 0.2 mM leupeptin/pepstatin,1 mM PMSF |
|---|---|
| Staining | Type: NEGATIVE Details: Grids floated on drops of sample buffer twice with blotting inbetween, than floated on 1% solution of uranyl acetate twice (blot inbetween) for 10 seconds each |
| Grid | Details: Continuous carbon on 400 mesh copper grid |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI/PHILIPS CM120T |
|---|---|
| Alignment procedure | Legacy - Astigmatism: Objective lens astigmatism was corrected at 100,000 times magnification |
| Image recording | Category: FILM / Film or detector model: KODAK SO-163 FILM / Digitization - Scanner: ZEISS SCAI / Digitization - Sampling interval: 3.5 µm / Number real images: 40 / Average electron dose: 10 e/Å2 / Bits/pixel: 8 |
| Electron beam | Acceleration voltage: 120 kV / Electron source: LAB6 |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.0 mm / Nominal defocus max: 1.9 µm / Nominal defocus min: 1.5 µm / Nominal magnification: 60000 |
| Sample stage | Specimen holder: Side-entry room temperature / Specimen holder model: OTHER |
- Image processing
Image processing
| Details | Manual selection of particles |
|---|---|
| CTF correction | Details: Each particle |
| Final reconstruction | Applied symmetry - Point group: C4 (4 fold cyclic) / Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 25.0 Å / Resolution method: FSC 0.5 CUT-OFF / Software - Name: IMAGIC, FREALIGN / Number images used: 6000 |
| Final two d classification | Number classes: 50 |
 Movie
Movie Controller
Controller


 UCSF Chimera
UCSF Chimera















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)