[English] 日本語
 Yorodumi
Yorodumi- EMDB-0553: Cryo-EM structure of AAV-2 in complex with AAVR PKD domains 1 and 2 -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-0553 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of AAV-2 in complex with AAVR PKD domains 1 and 2 | |||||||||
 Map data Map data | EM sharpened map | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | AAV / AAVR / receptor / coreceptor / VIRUS / KIAA0319L / PKD / parvovirus | |||||||||
| Function / homology |  Function and homology information Function and homology informationproacrosomal vesicle fusion / symbiont entry into host cell via permeabilization of host membrane / response to auditory stimulus / host cell nucleolus / flagellated sperm motility / T=1 icosahedral viral capsid / receptor-mediated endocytosis of virus by host cell / trans-Golgi network membrane / neuron migration / cytoplasmic vesicle ...proacrosomal vesicle fusion / symbiont entry into host cell via permeabilization of host membrane / response to auditory stimulus / host cell nucleolus / flagellated sperm motility / T=1 icosahedral viral capsid / receptor-mediated endocytosis of virus by host cell / trans-Golgi network membrane / neuron migration / cytoplasmic vesicle / clathrin-dependent endocytosis of virus by host cell / Golgi membrane / virion attachment to host cell / nucleolus / structural molecule activity / Golgi apparatus / membrane / plasma membrane Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) / Homo sapiens (human) /  Adeno-associated virus - 2 Adeno-associated virus - 2 | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.4 Å | |||||||||
 Authors Authors | Meyer NL / Xie Q | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: Elife / Year: 2019 Journal: Elife / Year: 2019Title: Structure of the gene therapy vector, adeno-associated virus with its cell receptor, AAVR. Authors: Nancy L Meyer / Guiqing Hu / Omar Davulcu / Qing Xie / Alex J Noble / Craig Yoshioka / Drew S Gingerich / Andrew Trzynka / Larry David / Scott M Stagg / Michael Stewart Chapman /  Abstract: Adeno-associated virus (AAV) vectors are preeminent in emerging clinical gene therapies. Generalizing beyond the most tractable genetic diseases will require modulation of cell specificity and immune ...Adeno-associated virus (AAV) vectors are preeminent in emerging clinical gene therapies. Generalizing beyond the most tractable genetic diseases will require modulation of cell specificity and immune neutralization. Interactions of AAV with its cellular receptor, AAVR, are key to understanding cell-entry and trafficking with the rigor needed to engineer tissue-specific vectors. -electron tomography shows ordered binding of part of the flexible receptor to the viral surface, with distal domains in multiple conformations. Regions of the virus and receptor in close physical proximity can be identified by cross-linking/mass spectrometry. -electron microscopy with a two-domain receptor fragment reveals the interactions at 2.4 Å resolution. AAVR binds between AAV's spikes on a plateau that is conserved, except in one clade whose structure is AAVR-incompatible. AAVR's footprint overlaps the epitopes of several neutralizing antibodies, prompting a re-evaluation of neutralization mechanisms. The structure provides a roadmap for experimental probing and manipulation of viral-receptor interactions. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_0553.map.gz emd_0553.map.gz | 42.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-0553-v30.xml emd-0553-v30.xml emd-0553.xml emd-0553.xml | 25.3 KB 25.3 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_0553.png emd_0553.png | 319.1 KB | ||
| Masks |  emd_0553_msk_1.map emd_0553_msk_1.map | 216 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-0553.cif.gz emd-0553.cif.gz | 8.2 KB | ||
| Others |  emd_0553_half_map_1.map.gz emd_0553_half_map_1.map.gz emd_0553_half_map_2.map.gz emd_0553_half_map_2.map.gz | 170.7 MB 170.8 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-0553 http://ftp.pdbj.org/pub/emdb/structures/EMD-0553 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-0553 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-0553 | HTTPS FTP |
-Related structure data
| Related structure data |  6nz0MC  0621C 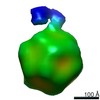 0622C  0623C  0624C C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_0553.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_0553.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | EM sharpened map | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.046 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Mask #1
| File |  emd_0553_msk_1.map emd_0553_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: half map 2
| File | emd_0553_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: half map 1
| File | emd_0553_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map 1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Adeno-associated virus - 2
| Entire | Name:  Adeno-associated virus - 2 Adeno-associated virus - 2 |
|---|---|
| Components |
|
-Supramolecule #1: Adeno-associated virus - 2
| Supramolecule | Name: Adeno-associated virus - 2 / type: virus / ID: 1 / Parent: 0 / Macromolecule list: #1-#2 Details: AAV-2 virus-like particles expressed in Sf9 cells using Invitrogen's Bac-to-Bac expression system NCBI-ID: 10804 / Sci species name: Adeno-associated virus - 2 / Virus type: VIRUS-LIKE PARTICLE / Virus isolate: SEROTYPE / Virus enveloped: No / Virus empty: Yes |
|---|---|
| Host (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 22 KDa |
| Virus shell | Shell ID: 1 / Diameter: 250.0 Å / T number (triangulation number): 1 |
-Supramolecule #2: Binary complex of AAV-2 with a fragment of its cellular receptor,...
| Supramolecule | Name: Binary complex of AAV-2 with a fragment of its cellular receptor, AAVR or dyslexia-associated protein KIAA0319-like protein. type: complex / ID: 2 / Parent: 1 / Macromolecule list: #1 Details: Expressed N-terminally His6-tagged fragment of AAVR containing the PKD1 and PKD2 domains. |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Dyslexia-associated protein KIAA0319-like protein
| Macromolecule | Name: Dyslexia-associated protein KIAA0319-like protein / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 31.837385 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MASVSAGESV QITLPKNEVQ LNAYVLQEPP KGETYTYDWQ LITHPRDYSG EMEGKHSQIL KLSKLTPGLY EFKVIVEGQN AHGEGYVNV TVKPEPRKNR PPIAIVSPQF QEISLPTTST VIDGSQSTDD DKIVQYHWEE LKGPLREEKI SEDTAILKLS K LVPGNYTF ...String: MASVSAGESV QITLPKNEVQ LNAYVLQEPP KGETYTYDWQ LITHPRDYSG EMEGKHSQIL KLSKLTPGLY EFKVIVEGQN AHGEGYVNV TVKPEPRKNR PPIAIVSPQF QEISLPTTST VIDGSQSTDD DKIVQYHWEE LKGPLREEKI SEDTAILKLS K LVPGNYTF SLTVVDSDGA TNSTTANLTV NKAVDYPPVA NAGPNQVITL PQNSITLFGN QSTDDHGITS YEWSLSPSSK GK VVEMQGV RTPTLQLSAM QEGDYTYQLT VTDTIGQQAT AQVTVIVQPE NNK UniProtKB: Dyslexia-associated protein KIAA0319-like protein |
-Macromolecule #2: Capsid protein VP1
| Macromolecule | Name: Capsid protein VP1 / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Adeno-associated virus - 2 Adeno-associated virus - 2 |
| Molecular weight | Theoretical: 82.077445 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MAADGYLPDW LEDTLSEGIR QWWKLKPGPP PPKPAERHKD DSRGLVLPGY KYLGPFNGLD KGEPVNEADA AALEHDKAYD RQLDSGDNP YLKYNHADAE FQERLKEDTS FGGNLGRAVF QAKKRVLEPL GLVEEPVKTA PGKKRPVEHS PVEPDSSSGT G KAGQQPAR ...String: MAADGYLPDW LEDTLSEGIR QWWKLKPGPP PPKPAERHKD DSRGLVLPGY KYLGPFNGLD KGEPVNEADA AALEHDKAYD RQLDSGDNP YLKYNHADAE FQERLKEDTS FGGNLGRAVF QAKKRVLEPL GLVEEPVKTA PGKKRPVEHS PVEPDSSSGT G KAGQQPAR KRLNFGQTGD ADSVPDPQPL GQPPAAPSGL GTNTMATGSG APMADNNEGA DGVGNSSGNW HCDSTWMGDR VI TTSTRTW ALPTYNNHLY KQISSQSGAS NDNHYFGYST PWGYFDFNRF HCHFSPRDWQ RLINNNWGFR PKRLNFKLFN IQV KEVTQN DGTTTIANNL TSTVQVFTDS EYQLPYVLGS AHQGCLPPFP ADVFMVPQYG YLTLNNGSQA VCRSSFYCLE YFPS QMLRT GNNFTFSYTF EDVPFHSSYA HSQSLDRLMN PLIDQYLYYL SRTNTPSGTT TQSRLQFSQA GASDIRDQSR NWLPG PCYR QQRVSKTSAD NNNSEYSWTG ATKYHLNGRD SLVNPGPAMA SHKDDEEKFF PQSGVLIFGK QGSEKTNVDI EKVMIT DEE EIRTTNPVAT EQYGSVSTNL QRGNRQAATA DVNTQGVLPG MVWQDRDVYL QGPIWAKIPH TDGHFHPSPL MGGFGLK HP PPQILIKNTP VPANPSTTFS AAKFASFITQ YSTGQVSVEI EWELQKENSK RWNPEIQYTS NYNKSVNVDF TVDTNGVY S EPRPIGTRYL TRNL UniProtKB: Capsid protein VP1 |
-Macromolecule #3: MAGNESIUM ION
| Macromolecule | Name: MAGNESIUM ION / type: ligand / ID: 3 / Number of copies: 1 / Formula: MG |
|---|---|
| Molecular weight | Theoretical: 24.305 Da |
-Macromolecule #4: water
| Macromolecule | Name: water / type: ligand / ID: 4 / Number of copies: 143 / Formula: HOH |
|---|---|
| Molecular weight | Theoretical: 18.015 Da |
| Chemical component information |  ChemComp-HOH: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.1 mg/mL | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.4 Component:
| ||||||||
| Grid | Material: COPPER / Mesh: 400 / Support film - Material: CARBON / Support film - topology: LACEY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 25 sec. / Pretreatment - Atmosphere: AIR / Pretreatment - Pressure: 0.039 kPa Details: Glow discharge settings: Pelco easiGlow, 25s, 25mA, 0.39mBar, negative polarity Grid: Ted Pella Cat#01824, ultrathin carbon film (<3nm) on lacey carbon support film | ||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 298.15 K / Instrument: FEI VITROBOT MARK IV Details: 4ul of AAV-2 (1.7uM) was added to the grid, followed by 4ul of AAVR-PKD1-2 (16.7uM), with manual blotting after each addition with Whatman paper (Cat. no. 1001-110.) 4ul of buffer containing ...Details: 4ul of AAV-2 (1.7uM) was added to the grid, followed by 4ul of AAVR-PKD1-2 (16.7uM), with manual blotting after each addition with Whatman paper (Cat. no. 1001-110.) 4ul of buffer containing 25mM HEPES, 150mM NaCl, pH 7.4 was added to the grid before final blotting and plunge-freezing in the Vitrobot (blot time 1.5sec, blot force -1).. | ||||||||
| Details | Virus-like particles at 0.10 mg/mL were adhered to thin continuous carbon-coated grids, then, after initial blotting, AAVR-PKD1-2 was added at 0.74 mg/mL (a 10-fold molar excess over AAV2 subunits) before blotting again. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON III (4k x 4k) / Detector mode: SUPER-RESOLUTION / Digitization - Dimensions - Width: 7676 pixel / Digitization - Dimensions - Height: 7420 pixel / Number grids imaged: 1 / Number real images: 2329 / Average exposure time: 40.0 sec. / Average electron dose: 25.4 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: -2.0 µm / Nominal defocus min: -0.8 µm / Nominal magnification: 75000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model | PDB ID: Chain - Chain ID: A / Chain - Residue range: 217-735 / Chain - Source name: PDB / Chain - Initial model type: experimental model |
|---|---|
| Details | An initial model for AAVR was constructed by homology modeling using Modeller 9.2. After remodeling AAVR and the crystal structure of AAV2, the structure was refined using RSRef embedded in CNS for stereochemically restrained torsion angle simulated annealing and gradient descent optimization. Stand-alone RSRef was used for refinement of EM envelope corrections, resolution estimates, and restrained atomic B-factors. |
| Refinement | Space: REAL / Protocol: FLEXIBLE FIT Target criteria: Map values at grid points within 2 Angstrom of atoms |
| Output model |  PDB-6nz0: |
 Movie
Movie Controller
Controller















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)