[English] 日本語
 Yorodumi
Yorodumi- EMDB-42230: In-situ structure of typeO supercomplex in respiratory chain (com... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
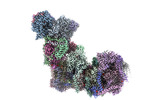
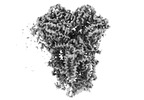
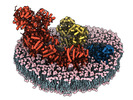
| Title | In-situ structure of typeO supercomplex in respiratory chain (composite) | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | in-situ cryo-EM structure / mammalian / mitochondria / respiratory supercomplex / proton pumping / membrane protein / electron transport | |||||||||
| Function / homology |  Function and homology information Function and homology informationCytoprotection by HMOX1 / TP53 Regulates Metabolic Genes / respiratory chain complex IV assembly / mitochondrial respirasome assembly / RHOG GTPase cycle / respiratory chain complex IV / Complex I biogenesis / Respiratory electron transport / Neutrophil degranulation / : ...Cytoprotection by HMOX1 / TP53 Regulates Metabolic Genes / respiratory chain complex IV assembly / mitochondrial respirasome assembly / RHOG GTPase cycle / respiratory chain complex IV / Complex I biogenesis / Respiratory electron transport / Neutrophil degranulation / : / regulation of oxidative phosphorylation / Mitochondrial protein degradation / : / cardiac muscle tissue development / cytochrome-c oxidase / : / quinol-cytochrome-c reductase / ubiquinone-6 biosynthetic process / cellular respiration / ubiquinol-cytochrome-c reductase activity / oxidoreductase activity, acting on NAD(P)H / mitochondrial electron transport, cytochrome c to oxygen / cytochrome-c oxidase activity / NADH dehydrogenase activity / mitochondrial electron transport, ubiquinol to cytochrome c / NADH:ubiquinone reductase (H+-translocating) / positive regulation of vasoconstriction / acyl binding / ubiquinone binding / acyl carrier activity / quinone binding / electron transport coupled proton transport / : / enzyme regulator activity / ATP synthesis coupled electron transport / negative regulation of intrinsic apoptotic signaling pathway / mitochondrial ATP synthesis coupled electron transport / mitochondrial respiratory chain complex I assembly / response to cAMP / respiratory electron transport chain / reactive oxygen species metabolic process / : / mitochondrial electron transport, NADH to ubiquinone / central nervous system development / NADH dehydrogenase (ubiquinone) activity / electron transport chain / regulation of protein phosphorylation / brain development / aerobic respiration / mitochondrial intermembrane space / negative regulation of cell growth / metalloendopeptidase activity / 2 iron, 2 sulfur cluster binding / positive regulation of fibroblast proliferation / NAD binding / FMN binding / nervous system development / 4 iron, 4 sulfur cluster binding / response to oxidative stress / oxidoreductase activity / nuclear body / electron transfer activity / mitochondrial inner membrane / mitochondrial matrix / copper ion binding / heme binding / protein-containing complex binding / mitochondrion / proteolysis / nucleoplasm / membrane / metal ion binding / plasma membrane / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.7 Å | |||||||||
 Authors Authors | Zheng W / Zhang K / Zhu J | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2024 Journal: Nature / Year: 2024Title: High-resolution in situ structures of mammalian respiratory supercomplexes. Authors: Wan Zheng / Pengxin Chai / Jiapeng Zhu / Kai Zhang /   Abstract: Mitochondria play a pivotal part in ATP energy production through oxidative phosphorylation, which occurs within the inner membrane through a series of respiratory complexes. Despite extensive in ...Mitochondria play a pivotal part in ATP energy production through oxidative phosphorylation, which occurs within the inner membrane through a series of respiratory complexes. Despite extensive in vitro structural studies, determining the atomic details of their molecular mechanisms in physiological states remains a major challenge, primarily because of loss of the native environment during purification. Here we directly image porcine mitochondria using an in situ cryo-electron microscopy approach. This enables us to determine the structures of various high-order assemblies of respiratory supercomplexes in their native states. We identify four main supercomplex organizations: IIIIIV, IIIIIV, IIIIIV and IIIIIV, which potentially expand into higher-order arrays on the inner membranes. These diverse supercomplexes are largely formed by 'protein-lipids-protein' interactions, which in turn have a substantial impact on the local geometry of the surrounding membranes. Our in situ structures also capture numerous reactive intermediates within these respiratory supercomplexes, shedding light on the dynamic processes of the ubiquinone/ubiquinol exchange mechanism in complex I and the Q-cycle in complex III. Structural comparison of supercomplexes from mitochondria treated under different conditions indicates a possible correlation between conformational states of complexes I and III, probably in response to environmental changes. By preserving the native membrane environment, our approach enables structural studies of mitochondrial respiratory supercomplexes in reaction at high resolution across multiple scales, from atomic-level details to the broader subcellular context. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_42230.map.gz emd_42230.map.gz | 21.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-42230-v30.xml emd-42230-v30.xml emd-42230.xml emd-42230.xml | 93.5 KB 93.5 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_42230.png emd_42230.png | 85.9 KB | ||
| Filedesc metadata |  emd-42230.cif.gz emd-42230.cif.gz | 18.7 KB | ||
| Others |  emd_42230_additional_1.map.gz emd_42230_additional_1.map.gz emd_42230_additional_2.map.gz emd_42230_additional_2.map.gz | 478.8 MB 254.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-42230 http://ftp.pdbj.org/pub/emdb/structures/EMD-42230 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-42230 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-42230 | HTTPS FTP |
-Validation report
| Summary document |  emd_42230_validation.pdf.gz emd_42230_validation.pdf.gz | 401.3 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_42230_full_validation.pdf.gz emd_42230_full_validation.pdf.gz | 400.9 KB | Display | |
| Data in XML |  emd_42230_validation.xml.gz emd_42230_validation.xml.gz | 7.9 KB | Display | |
| Data in CIF |  emd_42230_validation.cif.gz emd_42230_validation.cif.gz | 9.1 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-42230 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-42230 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-42230 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-42230 | HTTPS FTP |
-Related structure data
| Related structure data |  8ugnMC  8ud1C  8ueoC  8uepC  8ueqC  8uerC  8uesC  8uetC  8ueuC  8uevC  8uewC  8uexC  8ueyC  8uezC  8ugdC  8ugeC  8ugfC  8uggC  8ughC  8ugiC  8ugjC  8ugkC  8uglC  8ugpC  8ugrC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_42230.map.gz / Format: CCP4 / Size: 634.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_42230.map.gz / Format: CCP4 / Size: 634.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 1.17 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: #1
| File | emd_42230_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
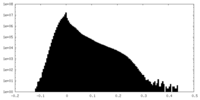
| Projections & Slices |
| ||||||||||||
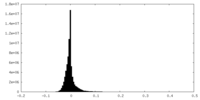
| Density Histograms |
-Additional map: #2
| File | emd_42230_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
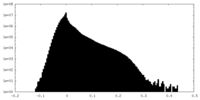
| Projections & Slices |
| ||||||||||||
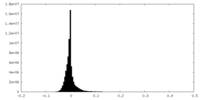
| Density Histograms |
- Sample components
Sample components
+Entire : In-situ cryo-EM structure of respiratory supercomplex by directly...
+Supramolecule #1: In-situ cryo-EM structure of respiratory supercomplex by directly...
+Macromolecule #1: NADH-ubiquinone oxidoreductase chain 3
+Macromolecule #2: NADH dehydrogenase [ubiquinone] iron-sulfur protein 7, mitochondrial
+Macromolecule #3: NADH dehydrogenase [ubiquinone] iron-sulfur protein 3, mitochondrial
+Macromolecule #4: NADH dehydrogenase [ubiquinone] iron-sulfur protein 2, mitochondrial
+Macromolecule #5: NADH dehydrogenase [ubiquinone] flavoprotein 2, mitochondrial
+Macromolecule #6: NADH dehydrogenase [ubiquinone] flavoprotein 1, mitochondrial
+Macromolecule #7: NADH-ubiquinone oxidoreductase 75 kDa subunit, mitochondrial
+Macromolecule #8: NADH-ubiquinone oxidoreductase chain 1
+Macromolecule #9: NADH dehydrogenase [ubiquinone] iron-sulfur protein 8, mitochondrial
+Macromolecule #10: NADH-ubiquinone oxidoreductase chain 6
+Macromolecule #11: NADH-ubiquinone oxidoreductase chain 4L
+Macromolecule #12: NADH-ubiquinone oxidoreductase chain 5
+Macromolecule #13: NADH-ubiquinone oxidoreductase chain 4
+Macromolecule #14: NADH-ubiquinone oxidoreductase chain 2
+Macromolecule #15: NADH dehydrogenase [ubiquinone] 1 alpha subcomplex subunit 10, mi...
+Macromolecule #16: NADH:ubiquinone oxidoreductase subunit A9
+Macromolecule #17: NADH dehydrogenase [ubiquinone] iron-sulfur protein 4, mitochondrial
+Macromolecule #18: NADH dehydrogenase [ubiquinone] iron-sulfur protein 6, mitochondrial
+Macromolecule #19: NADH dehydrogenase [ubiquinone] 1 alpha subcomplex subunit 2
+Macromolecule #20: NADH:ubiquinone oxidoreductase subunit AB1
+Macromolecule #21: NADH dehydrogenase [ubiquinone] 1 alpha subcomplex subunit 5 isof...
+Macromolecule #22: NADH dehydrogenase [ubiquinone] 1 alpha subcomplex subunit 6
+Macromolecule #23: NADH dehydrogenase [ubiquinone] 1 alpha subcomplex subunit 8
+Macromolecule #24: NADH dehydrogenase [ubiquinone] 1 alpha subcomplex subunit 11
+Macromolecule #25: NADH:ubiquinone oxidoreductase subunit A13
+Macromolecule #26: NADH dehydrogenase [ubiquinone] 1 alpha subcomplex subunit 1
+Macromolecule #27: NADH dehydrogenase [ubiquinone] 1 alpha subcomplex subunit 3
+Macromolecule #28: NADH dehydrogenase [ubiquinone] 1 subunit C1, mitochondrial
+Macromolecule #29: NADH dehydrogenase [ubiquinone] 1 subunit C2
+Macromolecule #30: NADH dehydrogenase [ubiquinone] iron-sulfur protein 5
+Macromolecule #31: NADH dehydrogenase [ubiquinone] 1 beta subcomplex subunit 1 [Sus ...
+Macromolecule #32: NADH dehydrogenase [ubiquinone] 1 beta subcomplex subunit 11, mit...
+Macromolecule #33: NADH dehydrogenase [ubiquinone] 1 beta subcomplex subunit 5, mito...
+Macromolecule #34: NADH dehydrogenase [ubiquinone] 1 beta subcomplex subunit 6
+Macromolecule #35: NADH:ubiquinone oxidoreductase subunit B2
+Macromolecule #36: NADH dehydrogenase [ubiquinone] 1 beta subcomplex subunit 3
+Macromolecule #37: NADH dehydrogenase [ubiquinone] 1 beta subcomplex subunit 8, mito...
+Macromolecule #38: NADH dehydrogenase [ubiquinone] 1 beta subcomplex subunit 4
+Macromolecule #39: NADH dehydrogenase [ubiquinone] 1 beta subcomplex subunit 9
+Macromolecule #40: NADH dehydrogenase [ubiquinone] 1 beta subcomplex subunit 7
+Macromolecule #41: NADH dehydrogenase [ubiquinone] 1 beta subcomplex subunit 10
+Macromolecule #42: NADH dehydrogenase [ubiquinone] 1 alpha subcomplex subunit 12
+Macromolecule #43: NADH dehydrogenase [ubiquinone] 1 alpha subcomplex subunit 7
+Macromolecule #44: NADH dehydrogenase [ubiquinone] flavoprotein 3, mitochondrial
+Macromolecule #45: Cytochrome b-c1 complex subunit 1, mitochondrial
+Macromolecule #46: Cytochrome b-c1 complex subunit 2, mitochondrial
+Macromolecule #47: Cytochrome b
+Macromolecule #48: Cytochrome c1
+Macromolecule #49: Cytochrome b-c1 complex subunit Rieske, mitochondrial
+Macromolecule #50: Cytochrome b-c1 complex subunit 7
+Macromolecule #51: Cytochrome b-c1 complex subunit 8
+Macromolecule #52: Cytochrome b-c1 complex subunit 6, mitochondrial
+Macromolecule #53: Ubiquinol-cytochrome c reductase complex 7.2 kDa protein 53 compl...
+Macromolecule #54: Cytochrome b-c1 complex subunit 10
+Macromolecule #55: Cytochrome c oxidase subunit 1
+Macromolecule #56: Cytochrome c oxidase subunit 2
+Macromolecule #57: Cytochrome c oxidase subunit 3
+Macromolecule #58: Cytochrome c oxidase subunit 4
+Macromolecule #59: Cytochrome c oxidase subunit 5A, mitochondrial
+Macromolecule #60: Cytochrome c oxidase subunit 5B, mitochondrial
+Macromolecule #61: Cytochrome c oxidase subunit 6A2
+Macromolecule #62: Cytochrome c oxidase subunit 6B1
+Macromolecule #63: Cytochrome c oxidase subunit 6C
+Macromolecule #64: Cytochrome c oxidase subunit 7A1, mitochondrial
+Macromolecule #65: Cytochrome c oxidase subunit 7B
+Macromolecule #66: Cytochrome c oxidase subunit 7C, mitochondrial
+Macromolecule #67: Cytochrome c oxidase subunit 8
+Macromolecule #68: Cytochrome c oxidase subunit NDUFA4
+Macromolecule #69: 1,2-DIACYL-SN-GLYCERO-3-PHOSPHOCHOLINE
+Macromolecule #70: IRON/SULFUR CLUSTER
+Macromolecule #71: FE2/S2 (INORGANIC) CLUSTER
+Macromolecule #72: FLAVIN MONONUCLEOTIDE
+Macromolecule #73: POTASSIUM ION
+Macromolecule #74: CARDIOLIPIN
+Macromolecule #75: N-ACETYLALANINE
+Macromolecule #76: 1,2-Distearoyl-sn-glycerophosphoethanolamine
+Macromolecule #77: GUANOSINE-5'-TRIPHOSPHATE
+Macromolecule #78: MAGNESIUM ION
+Macromolecule #79: NADPH DIHYDRO-NICOTINAMIDE-ADENINE-DINUCLEOTIDE PHOSPHATE
+Macromolecule #80: ZINC ION
+Macromolecule #81: ~{S}-[2-[3-[[(2~{R})-3,3-dimethyl-2-oxidanyl-4-phosphonooxy-butan...
+Macromolecule #82: N-ACETYLMETHIONINE
+Macromolecule #83: MYRISTIC ACID
+Macromolecule #84: PROTOPORPHYRIN IX CONTAINING FE
+Macromolecule #85: HEME C
+Macromolecule #86: (1R)-2-{[{[(2S)-2,3-DIHYDROXYPROPYL]OXY}(HYDROXY)PHOSPHORYL]OXY}-...
+Macromolecule #87: HEME-A
+Macromolecule #88: COPPER (II) ION
+Macromolecule #89: SODIUM ION
+Macromolecule #90: (7R,17E,20E)-4-HYDROXY-N,N,N-TRIMETHYL-9-OXO-7-[(PALMITOYLOXY)MET...
+Macromolecule #91: DINUCLEAR COPPER ION
+Macromolecule #92: (1S)-2-{[(2-AMINOETHOXY)(HYDROXY)PHOSPHORYL]OXY}-1-[(STEAROYLOXY)...
+Macromolecule #93: PHOSPHATE ION
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 3.0 µm / Nominal defocus min: 1.3 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: NONE |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 2.7 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 80000 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller









































 Z
Z Y
Y X
X