[English] 日本語
 Yorodumi
Yorodumi- EMDB-28693: Cryo-EM structure of two IGF1 bound full-length mouse IGF1R mutan... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of two IGF1 bound full-length mouse IGF1R mutant (four glycine residues inserted in the alpha-CT; IGF1R-P674G4): symmetric conformation | |||||||||
 Map data Map data | Cryo-EM structure of two IGF1 bound full-length mouse IGF1R mutant (four glycine residues inserted in the alpha-CT; IGF1R-P674G4): symmetric conformation | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | IGF1R / IGF1 / SIGNALING PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of cholangiocyte apoptotic process / Signaling by Type 1 Insulin-like Growth Factor 1 Receptor (IGF1R) / IRS-related events triggered by IGF1R / SHC-related events triggered by IGF1R / positive regulation of steroid hormone biosynthetic process / glycolate metabolic process / muscle hypertrophy / negative regulation of oocyte development / insulin-like growth factor binding protein complex / insulin-like growth factor ternary complex ...negative regulation of cholangiocyte apoptotic process / Signaling by Type 1 Insulin-like Growth Factor 1 Receptor (IGF1R) / IRS-related events triggered by IGF1R / SHC-related events triggered by IGF1R / positive regulation of steroid hormone biosynthetic process / glycolate metabolic process / muscle hypertrophy / negative regulation of oocyte development / insulin-like growth factor binding protein complex / insulin-like growth factor ternary complex / positive regulation of trophectodermal cell proliferation / negative regulation of muscle cell apoptotic process / prostate gland stromal morphogenesis / positive regulation of type B pancreatic cell proliferation / type II pneumocyte differentiation / neuronal dense core vesicle lumen / positive regulation of glycoprotein biosynthetic process / proteoglycan biosynthetic process / regulation of establishment or maintenance of cell polarity / chondroitin sulfate proteoglycan biosynthetic process / positive regulation of transcription regulatory region DNA binding / insulin-like growth factor receptor activity / myotube cell development / positive regulation of DNA metabolic process / protein kinase complex / Extra-nuclear estrogen signaling / negative regulation of neuroinflammatory response / insulin-like growth factor binding / Signaling by Type 1 Insulin-like Growth Factor 1 Receptor (IGF1R) / skeletal muscle satellite cell maintenance involved in skeletal muscle regeneration / bone mineralization involved in bone maturation / positive regulation of cell growth involved in cardiac muscle cell development / IRS-related events triggered by IGF1R / negative regulation of vascular associated smooth muscle cell apoptotic process / positive regulation of cerebellar granule cell precursor proliferation / protein transporter activity / lung vasculature development / exocytic vesicle / cerebellar granule cell precursor proliferation / positive regulation of myoblast proliferation / transcytosis / positive regulation of meiotic cell cycle / positive regulation of axon regeneration / lung lobe morphogenesis / positive regulation of myelination / cell activation / negative regulation of hepatocyte apoptotic process / glial cell differentiation / negative regulation of androgen receptor signaling pathway / positive regulation of developmental growth / prostate gland epithelium morphogenesis / positive regulation of calcineurin-NFAT signaling cascade / transmembrane receptor protein tyrosine kinase activator activity / prostate gland growth / male sex determination / insulin receptor complex / positive regulation of protein-containing complex disassembly / insulin-like growth factor I binding / type B pancreatic cell proliferation / insulin receptor activity / mammary gland development / exocrine pancreas development / alphav-beta3 integrin-IGF-1-IGF1R complex / myoblast differentiation / cell surface receptor signaling pathway via STAT / positive regulation of insulin-like growth factor receptor signaling pathway / regulation of nitric oxide biosynthetic process / positive regulation of Ras protein signal transduction / response to L-glutamate / dendritic spine maintenance / activation of protein kinase B activity / positive regulation of smooth muscle cell migration / regulation of JNK cascade / growth hormone receptor signaling pathway / insulin binding / positive regulation of DNA binding / adrenal gland development / negative regulation of interleukin-1 beta production / lung alveolus development / muscle organ development / cellular response to insulin-like growth factor stimulus / branching morphogenesis of an epithelial tube / androgen receptor signaling pathway / prostate epithelial cord arborization involved in prostate glandular acinus morphogenesis / positive regulation of osteoblast proliferation / positive regulation of cardiac muscle hypertrophy / negative regulation of release of cytochrome c from mitochondria / positive regulation of cytokinesis / type I pneumocyte differentiation / establishment of cell polarity / negative regulation of amyloid-beta formation / inner ear development / negative regulation of smooth muscle cell apoptotic process / amyloid-beta clearance / myoblast proliferation / insulin receptor substrate binding / positive regulation of activated T cell proliferation / epithelial to mesenchymal transition / negative regulation of tumor necrosis factor production / Synthesis, secretion, and deacylation of Ghrelin Similarity search - Function | |||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 4.0 Å | |||||||||
 Authors Authors | Li J / Wu JY / Hall C / Bai XC / Choi E | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: Elife / Year: 2022 Journal: Elife / Year: 2022Title: Molecular basis for the role of disulfide-linked αCTs in the activation of insulin-like growth factor 1 receptor and insulin receptor. Authors: Jie Li / Jiayi Wu / Catherine Hall / Xiao-Chen Bai / Eunhee Choi /  Abstract: The insulin receptor (IR) and insulin-like growth factor 1 receptor (IGF1R) control metabolic homeostasis and cell growth and proliferation. The IR and IGF1R form similar disulfide bonds linked ...The insulin receptor (IR) and insulin-like growth factor 1 receptor (IGF1R) control metabolic homeostasis and cell growth and proliferation. The IR and IGF1R form similar disulfide bonds linked homodimers in the apo-state; however, their ligand binding properties and the structures in the active state differ substantially. It has been proposed that the disulfide-linked C-terminal segment of α-chain (αCTs) of the IR and IGF1R control the cooperativity of ligand binding and regulate the receptor activation. Nevertheless, the molecular basis for the roles of disulfide-linked αCTs in IR and IGF1R activation are still unclear. Here, we report the cryo-EM structures of full-length mouse IGF1R/IGF1 and IR/insulin complexes with modified αCTs that have increased flexibility. Unlike the -shaped asymmetric IGF1R dimer with a single IGF1 bound, the IGF1R with the enhanced flexibility of αCTs can form a -shaped symmetric dimer with two IGF1s bound. Meanwhile, the IR with non-covalently linked αCTs predominantly adopts an asymmetric conformation with four insulins bound, which is distinct from the -shaped symmetric IR. Using cell-based experiments, we further showed that both IGF1R and IR with the modified αCTs cannot activate the downstream signaling potently. Collectively, our studies demonstrate that the certain structural rigidity of disulfide-linked αCTs is critical for optimal IR and IGF1R signaling activation. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_28693.map.gz emd_28693.map.gz | 110.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-28693-v30.xml emd-28693-v30.xml emd-28693.xml emd-28693.xml | 18.5 KB 18.5 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_28693.png emd_28693.png | 35.6 KB | ||
| Filedesc metadata |  emd-28693.cif.gz emd-28693.cif.gz | 6.7 KB | ||
| Others |  emd_28693_half_map_1.map.gz emd_28693_half_map_1.map.gz emd_28693_half_map_2.map.gz emd_28693_half_map_2.map.gz | 139.2 MB 139.1 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-28693 http://ftp.pdbj.org/pub/emdb/structures/EMD-28693 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-28693 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-28693 | HTTPS FTP |
-Related structure data
| Related structure data |  8eyrMC  8eyxC  8eyyC  8ez0C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_28693.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_28693.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cryo-EM structure of two IGF1 bound full-length mouse IGF1R mutant (four glycine residues inserted in the alpha-CT; IGF1R-P674G4): symmetric conformation | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.83 Å | ||||||||||||||||||||||||||||||||||||
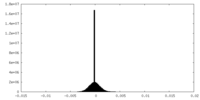
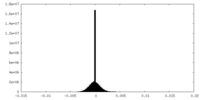
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: Cryo-EM structure of two IGF1 bound full-length mouse...
| File | emd_28693_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cryo-EM structure of two IGF1 bound full-length mouse IGF1R mutant (four glycine residues inserted in the alpha-CT; IGF1R-P674G4): symmetric conformation Unfiltered half1 map | ||||||||||||
| Projections & Slices |
| ||||||||||||
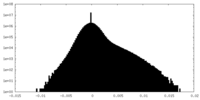
| Density Histograms |
-Half map: Cryo-EM structure of two IGF1 bound full-length mouse...
| File | emd_28693_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cryo-EM structure of two IGF1 bound full-length mouse IGF1R mutant (four glycine residues inserted in the alpha-CT; IGF1R-P674G4): symmetric conformation Unfiltered half2 map | ||||||||||||
| Projections & Slices |
| ||||||||||||
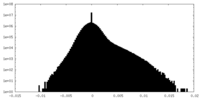
| Density Histograms |
- Sample components
Sample components
-Entire : Two IGF1 bound full-length mouse IGF1R mutant (four glycine resid...
| Entire | Name: Two IGF1 bound full-length mouse IGF1R mutant (four glycine residues inserted in the alpha-CT; IGF1R-P674G4): symmetric conformation |
|---|---|
| Components |
|
-Supramolecule #1: Two IGF1 bound full-length mouse IGF1R mutant (four glycine resid...
| Supramolecule | Name: Two IGF1 bound full-length mouse IGF1R mutant (four glycine residues inserted in the alpha-CT; IGF1R-P674G4): symmetric conformation type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: Insulin-like growth factor 1 receptor
| Macromolecule | Name: Insulin-like growth factor 1 receptor / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO / EC number: receptor protein-tyrosine kinase |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 144.481953 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: EICGPGIDIR NDYQQLKRLE NCTVIEGFLH ILLISKAEDY RSYRFPKLTV ITEYLLLFRV AGLESLGDLF PNLTVIRGWK LFYNYALVI FEMTNLKDIG LYNLRNITRG AIRIEKNADL CYLSTIDWSL ILDAVSNNYI VGNKPPKECG DLCPGTLEEK P MCEKTTIN ...String: EICGPGIDIR NDYQQLKRLE NCTVIEGFLH ILLISKAEDY RSYRFPKLTV ITEYLLLFRV AGLESLGDLF PNLTVIRGWK LFYNYALVI FEMTNLKDIG LYNLRNITRG AIRIEKNADL CYLSTIDWSL ILDAVSNNYI VGNKPPKECG DLCPGTLEEK P MCEKTTIN NEYNYRCWTT NRCQKMCPSV CGKRACTENN ECCHPECLGS CHTPDDNTTC VACRHYYYKG VCVPACPPGT YR FEGWRCV DRDFCANIPN AESSDSDGFV IHDDECMQEC PSGFIRNSTQ SMYCIPCEGP CPKVCGDEEK KTKTIDSVTS AQM LQGCTI LKGNLLINIR RGNNIASELE NFMGLIEVVT GYVKIRHSHA LVSLSFLKNL RLILGEEQLE GNYSFYVLDN QNLQ QLWDW NHRNLTVRSG KMYFAFNPKL CVSEIYRMEE VTGTKGRQSK GDINTRNNGE RASCESDVLR FTSTTTWKNR IIITW HRYR PPDYRDLISF TVYYKEAPFK NVTEYDGQDA CGSNSWNMVD VDLPPNKEGE PGILLHGLKP WTQYAVYVKA VTLTMV END HIRGAKSEIL YIRTNASVPS IPLDVLSASN SSSQLIVKWN PPTLPNGNLS YYIVRWQRQP QDGYLYRHNY CSKDKIP IR KYADGTIDVE EVTENPKTEV CGGDKGPCCA CPGGGGKTEA EKQAEKEEAE YRKVFENFLH NSIFVPRPER RRRDVMQV A NTTMSSRSRN TTVADTYNIT DPEEFETEYP FFESRVDNKE RTVISNLRPF TLYRIDIHSC NHEAEKLGCS ASNFVFART MPAEGADDIP GPVTWEPRPE NSIFLKWPEP ENPNGLILMY EIKYGSQVED QRECVSRQEY RKYGGAKLNR LNPGNYTARI QATSLSGNG SWTDPVFFYV PAKTTYENFM HLIIALPVAI LLIVGGLVIM LYVFHRKRNN SRLGNGVLYA SVNPEYFSAA D VYVPDEWE VAREKITMNR ELGQGSFGMV YEGVAKGVVK DEPETRVAIK TVNEAASMRE RIEFLNEASV MKEFNCHHVV RL LGVVSQG QPTLVIMELM TRGDLKSYLR SLRPEVEQNN LVLIPPSLSK MIQMAGEIAD GMAYLNANKF VHRDLAARNC MVA EDFTVK IGDFGMTRDI YETDYYRKGG KGLLPVRWMS PESLKDGVFT THSDVWSFGV VLWEIATLAE QPYQGLSNEQ VLRF VMEGG LLDKPDNCPD MLFELMRMCW QYNPKMRPSF LEIIGSIKDE MEPSFQEVSF YYSEENKPPE P UniProtKB: Insulin-like growth factor 1 receptor |
-Macromolecule #2: Insulin-like growth factor I
| Macromolecule | Name: Insulin-like growth factor I / type: protein_or_peptide / ID: 2 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 21.88132 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MGKISSLPTQ LFKCCFCDFL KVKMHTMSSS HLFYLALCLL TFTSSATAGP ETLCGAELVD ALQFVCGDRG FYFNKPTGYG SSSRRAPQT GIVDECCFRS CDLRRLEMYC APLKPAKSAR SVRAQRHTDM PKTQKYQPPS TNKNTKSQRR KGWPKTHPGG E QKEGTEAS ...String: MGKISSLPTQ LFKCCFCDFL KVKMHTMSSS HLFYLALCLL TFTSSATAGP ETLCGAELVD ALQFVCGDRG FYFNKPTGYG SSSRRAPQT GIVDECCFRS CDLRRLEMYC APLKPAKSAR SVRAQRHTDM PKTQKYQPPS TNKNTKSQRR KGWPKTHPGG E QKEGTEAS LQIRGKKKEQ RREIGSRNAE CRGKKGK UniProtKB: Insulin-like growth factor 1 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 6 mg/mL |
|---|---|
| Buffer | pH: 7.4 |
| Grid | Model: Quantifoil / Material: GOLD / Mesh: 300 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Bioquantum / Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.6 µm / Nominal defocus min: 1.6 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller




















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)