[English] 日本語
 Yorodumi
Yorodumi- EMDB-12367: Nanodisc reconstituted human ABCB4 in complex with 4B1-Fab (posac... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-12367 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Nanodisc reconstituted human ABCB4 in complex with 4B1-Fab (posaconazole-bound, inward-open conformation) | ||||||||||||
 Map data Map data | Nanodisc reconstituted human ABCB4 in complex with 4B1-Fab (posaconazole-bound, inward-open conformation) | ||||||||||||
 Sample Sample |
| ||||||||||||
| Function / homology |  Function and homology information Function and homology informationresponse to fenofibrate / Defective ABCB4 causes PFIC3, ICP3 and GBD1 / positive regulation of phospholipid transport / positive regulation of cholesterol transport / positive regulation of phospholipid translocation / bile acid secretion / phospholipid transporter activity / cellular response to bile acid / phosphatidylcholine floppase activity / intercellular canaliculus ...response to fenofibrate / Defective ABCB4 causes PFIC3, ICP3 and GBD1 / positive regulation of phospholipid transport / positive regulation of cholesterol transport / positive regulation of phospholipid translocation / bile acid secretion / phospholipid transporter activity / cellular response to bile acid / phosphatidylcholine floppase activity / intercellular canaliculus / lipid homeostasis / P-type phospholipid transporter / phospholipid translocation / clathrin-coated vesicle / ATPase-coupled transmembrane transporter activity / ABC-type transporter activity / lipid metabolic process / PPARA activates gene expression / ABC-family proteins mediated transport / transmembrane transport / apical plasma membrane / membrane raft / focal adhesion / ATP hydrolysis activity / extracellular exosome / nucleoplasm / ATP binding / membrane / plasma membrane / cytoplasm / cytosol Similarity search - Function | ||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||||||||
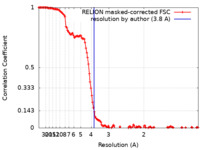
| Method | single particle reconstruction / cryo EM / Resolution: 3.8 Å | ||||||||||||
 Authors Authors | Nosol K / Locher KP | ||||||||||||
| Funding support |  Switzerland, Switzerland,  United States, 3 items United States, 3 items
| ||||||||||||
 Citation Citation |  Journal: Proc Natl Acad Sci U S A / Year: 2021 Journal: Proc Natl Acad Sci U S A / Year: 2021Title: Structures of ABCB4 provide insight into phosphatidylcholine translocation. Authors: Kamil Nosol / Rose Bang-Sørensen / Rossitza N Irobalieva / Satchal K Erramilli / Bruno Stieger / Anthony A Kossiakoff / Kaspar P Locher /   Abstract: ABCB4 is expressed in hepatocytes and translocates phosphatidylcholine into bile canaliculi. The mechanism of specific lipid recruitment from the canalicular membrane, which is essential to mitigate ...ABCB4 is expressed in hepatocytes and translocates phosphatidylcholine into bile canaliculi. The mechanism of specific lipid recruitment from the canalicular membrane, which is essential to mitigate the cytotoxicity of bile salts, is poorly understood. We present cryogenic electron microscopy structures of human ABCB4 in three distinct functional conformations. An apo-inward structure reveals how phospholipid can be recruited from the inner leaflet of the membrane without flipping its orientation. An occluded structure reveals a single phospholipid molecule in a central cavity. Its choline moiety is stabilized by cation-π interactions with an essential tryptophan residue, rationalizing the specificity of ABCB4 for phosphatidylcholine. In an inhibitor-bound structure, a posaconazole molecule blocks phospholipids from reaching the central cavity. Using a proteoliposome-based translocation assay with fluorescently labeled phosphatidylcholine analogs, we recapitulated the substrate specificity of ABCB4 in vitro and confirmed the role of the key tryptophan residue. Our results provide a structural basis for understanding an essential translocation step in the generation of bile and its sensitivity to azole drugs. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_12367.map.gz emd_12367.map.gz | 182 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-12367-v30.xml emd-12367-v30.xml emd-12367.xml emd-12367.xml | 16.8 KB 16.8 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_12367_fsc.xml emd_12367_fsc.xml | 13.8 KB | Display |  FSC data file FSC data file |
| Images |  emd_12367.png emd_12367.png | 101.7 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-12367 http://ftp.pdbj.org/pub/emdb/structures/EMD-12367 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12367 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12367 | HTTPS FTP |
-Validation report
| Summary document |  emd_12367_validation.pdf.gz emd_12367_validation.pdf.gz | 289.6 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_12367_full_validation.pdf.gz emd_12367_full_validation.pdf.gz | 288.8 KB | Display | |
| Data in XML |  emd_12367_validation.xml.gz emd_12367_validation.xml.gz | 14 KB | Display | |
| Data in CIF |  emd_12367_validation.cif.gz emd_12367_validation.cif.gz | 18.6 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12367 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12367 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12367 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12367 | HTTPS FTP |
-Related structure data
| Related structure data |  7niwMC  7niuC  7nivC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_12367.map.gz / Format: CCP4 / Size: 343 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_12367.map.gz / Format: CCP4 / Size: 343 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Nanodisc reconstituted human ABCB4 in complex with 4B1-Fab (posaconazole-bound, inward-open conformation) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.66 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
+Entire : Nanodisc reconstituted human ABCB4 in complex with 4B1-Fab (posac...
+Supramolecule #1: Nanodisc reconstituted human ABCB4 in complex with 4B1-Fab (posac...
+Supramolecule #2: phosphatidylcholine translocator ABCB4
+Supramolecule #3: 4B1 Fab-fragment: light chain
+Supramolecule #4: 4B1 Fab-fragment: heavy chain
+Macromolecule #1: Phosphatidylcholine translocator ABCB4
+Macromolecule #2: 4B1 Fab-fragment light chain
+Macromolecule #3: 4B1 Fab-fragment heavy chain
+Macromolecule #4: CHOLESTEROL
+Macromolecule #5: POSACONAZOLE
+Macromolecule #6: 1,2-DILINOLEOYL-SN-GLYCERO-3-PHOSPHOCHOLINE
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.48 mg/mL |
|---|---|
| Buffer | pH: 7.4 |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 80.4 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 100.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller