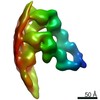
登録情報 データベース : EMDB / ID : EMD-12289タイトル Structure of the in situ actomyosin complex from the A-band of mouse psoas muscle sarcomere in the rigor state obtained by sub-tomogram averaging Averaged map of in situ actomyosin complex 複合体 : In situ actomyosin complex in the rigor state from mouse psoas muscle複合体 : actinタンパク質・ペプチド : Actin, alpha skeletal muscle複合体 : myosin double headタンパク質・ペプチド : Myosin-4タンパク質・ペプチド : Myosin light chain 1/3, skeletal muscle isoformタンパク質・ペプチド : Myosin regulatory light chain 2, skeletal muscle isoform複合体 : tropomyosinタンパク質・ペプチド : Tropomyosin alpha-1 chain / / / / 機能・相同性 分子機能 ドメイン・相同性 構成要素
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / 生物種 Mus musculus (ハツカネズミ)手法 / / 解像度 : 10.2 Å Wang Z / Grange M 資金援助 Organization Grant number 国 Max Planck Society Wellcome Trust 201543/Z/16/Z European Research Council (ERC) 856118 European Union Medical Research Council (MRC, United Kingdom) MR/R003106/1 European Molecular Biology Organization (EMBO) ALTF 693-2018 European Union
ジャーナル : Cell / 年 : 2021タイトル : The molecular basis for sarcomere organization in vertebrate skeletal muscle.著者 : Zhexin Wang / Michael Grange / Thorsten Wagner / Ay Lin Kho / Mathias Gautel / Stefan Raunser / 要旨 : Sarcomeres are force-generating and load-bearing devices of muscles. A precise molecular picture of how sarcomeres are built underpins understanding their role in health and disease. Here, we ... Sarcomeres are force-generating and load-bearing devices of muscles. A precise molecular picture of how sarcomeres are built underpins understanding their role in health and disease. Here, we determine the molecular architecture of native vertebrate skeletal sarcomeres by electron cryo-tomography. Our reconstruction reveals molecular details of the three-dimensional organization and interaction of actin and myosin in the A-band, I-band, and Z-disc and demonstrates that α-actinin cross-links antiparallel actin filaments by forming doublets with 6-nm spacing. Structures of myosin, tropomyosin, and actin at ~10 Å further reveal two conformations of the "double-head" myosin, where the flexible orientation of the lever arm and light chains enable myosin not only to interact with the same actin filament, but also to split between two actin filaments. Our results provide unexpected insights into the fundamental organization of vertebrate skeletal muscle and serve as a strong foundation for future investigations of muscle diseases. 履歴 登録 2021年2月4日 - ヘッダ(付随情報) 公開 2021年4月7日 - マップ公開 2021年4月7日 - 更新 2024年5月1日 - 現状 2024年5月1日 処理サイト : PDBe / 状態 : 公開
すべて表示 表示を減らす
 データを開く
データを開く 基本情報
基本情報 マップデータ
マップデータ 試料
試料 キーワード
キーワード 機能・相同性情報
機能・相同性情報
 データ登録者
データ登録者 ドイツ,
ドイツ,  英国, European Union, 5件
英国, European Union, 5件  引用
引用 ジャーナル: Cell / 年: 2021
ジャーナル: Cell / 年: 2021

 構造の表示
構造の表示 ムービービューア
ムービービューア SurfView
SurfView Molmil
Molmil Jmol/JSmol
Jmol/JSmol ダウンロードとリンク
ダウンロードとリンク emd_12289.map.gz
emd_12289.map.gz EMDBマップデータ形式
EMDBマップデータ形式 emd-12289-v30.xml
emd-12289-v30.xml emd-12289.xml
emd-12289.xml EMDBヘッダ
EMDBヘッダ emd_12289_fsc.xml
emd_12289_fsc.xml FSCデータファイル
FSCデータファイル emd_12289.png
emd_12289.png emd_12289_msk_1.map
emd_12289_msk_1.map マスクマップ
マスクマップ emd-12289.cif.gz
emd-12289.cif.gz emd_12289_half_map_1.map.gz
emd_12289_half_map_1.map.gz emd_12289_half_map_2.map.gz
emd_12289_half_map_2.map.gz http://ftp.pdbj.org/pub/emdb/structures/EMD-12289
http://ftp.pdbj.org/pub/emdb/structures/EMD-12289 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12289
ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12289 リンク
リンク EMDB (EBI/PDBe) /
EMDB (EBI/PDBe) /  EMDataResource
EMDataResource マップ
マップ ダウンロード / ファイル: emd_12289.map.gz / 形式: CCP4 / 大きさ: 30.5 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES)
ダウンロード / ファイル: emd_12289.map.gz / 形式: CCP4 / 大きさ: 30.5 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) emd_12289_msk_1.map
emd_12289_msk_1.map 試料の構成要素
試料の構成要素








 解析
解析 試料調製
試料調製 電子顕微鏡法
電子顕微鏡法 FIELD EMISSION GUN
FIELD EMISSION GUN
 画像解析
画像解析
 ムービー
ムービー コントローラー
コントローラー


















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)