+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-12177 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | The H/ACA RNP lobe of human telomerase | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | H/ACA RNP / ribonucleoprotein / complex / RNA / RNA BINDING PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationtelomere formation via telomerase / box H/ACA scaRNP complex / box H/ACA telomerase RNP complex / telomerase RNA localization to Cajal body / protein localization to Cajal body / snoRNA guided rRNA pseudouridine synthesis / box H/ACA snoRNP complex / Isomerases; Intramolecular transferases; Transferring other groups / rRNA pseudouridine synthesis / box H/ACA sno(s)RNA 3'-end processing ...telomere formation via telomerase / box H/ACA scaRNP complex / box H/ACA telomerase RNP complex / telomerase RNA localization to Cajal body / protein localization to Cajal body / snoRNA guided rRNA pseudouridine synthesis / box H/ACA snoRNP complex / Isomerases; Intramolecular transferases; Transferring other groups / rRNA pseudouridine synthesis / box H/ACA sno(s)RNA 3'-end processing / pseudouridine synthesis / snRNA pseudouridine synthesis / enzyme-directed rRNA pseudouridine synthesis / telomerase RNA stabilization / Cajal body organization / box H/ACA snoRNA binding / telomerase activity / regulation of telomerase RNA localization to Cajal body / mRNA pseudouridine synthesis / pseudouridine synthase activity / scaRNA localization to Cajal body / positive regulation of establishment of protein localization to telomere / positive regulation of protein localization to Cajal body / positive regulation of telomerase RNA localization to Cajal body / sno(s)RNA-containing ribonucleoprotein complex / telomerase holoenzyme complex / telomerase RNA binding / rRNA modification in the nucleus and cytosol / positive regulation of double-strand break repair / positive regulation of double-strand break repair via nonhomologous end joining / Association of TriC/CCT with target proteins during biosynthesis / : / Telomere Extension By Telomerase / RNA folding / telomere maintenance via telomerase / positive regulation of double-strand break repair via homologous recombination / RNA processing / Cajal body / positive regulation of telomere maintenance via telomerase / positive regulation of DNA repair / maturation of LSU-rRNA / fibrillar center / rRNA processing / site of double-strand break / protein-folding chaperone binding / histone binding / cytosolic large ribosomal subunit / chromosome, telomeric region / nuclear body / DNA repair / ubiquitin protein ligase binding / protein-containing complex binding / nucleolus / RNA binding / nucleoplasm / identical protein binding / nucleus / cytosol / cytoplasm Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
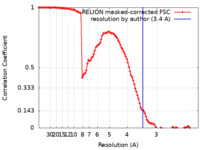
| Method | single particle reconstruction / cryo EM / Resolution: 3.4 Å | |||||||||
 Authors Authors | Nguyen THD / Ghanim GE | |||||||||
| Funding support |  United Kingdom, United Kingdom,  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2021 Journal: Nature / Year: 2021Title: Structure of human telomerase holoenzyme with bound telomeric DNA. Authors: George E Ghanim / Adam J Fountain / Anne-Marie M van Roon / Ramya Rangan / Rhiju Das / Kathleen Collins / Thi Hoang Duong Nguyen /   Abstract: Telomerase adds telomeric repeats at chromosome ends to compensate for the telomere loss that is caused by incomplete genome end replication. In humans, telomerase is upregulated during embryogenesis ...Telomerase adds telomeric repeats at chromosome ends to compensate for the telomere loss that is caused by incomplete genome end replication. In humans, telomerase is upregulated during embryogenesis and in cancers, and mutations that compromise the function of telomerase result in disease. A previous structure of human telomerase at a resolution of 8 Å revealed a vertebrate-specific composition and architecture, comprising a catalytic core that is flexibly tethered to an H and ACA (hereafter, H/ACA) box ribonucleoprotein (RNP) lobe by telomerase RNA. High-resolution structural information is necessary to develop treatments that can effectively modulate telomerase activity as a therapeutic approach against cancers and disease. Here we used cryo-electron microscopy to determine the structure of human telomerase holoenzyme bound to telomeric DNA at sub-4 Å resolution, which reveals crucial DNA- and RNA-binding interfaces in the active site of telomerase as well as the locations of mutations that alter telomerase activity. We identified a histone H2A-H2B dimer within the holoenzyme that was bound to an essential telomerase RNA motif, which suggests a role for histones in the folding and function of telomerase RNA. Furthermore, this structure of a eukaryotic H/ACA RNP reveals the molecular recognition of conserved RNA and protein motifs, as well as interactions that are crucial for understanding the molecular pathology of many mutations that cause disease. Our findings provide the structural details of the assembly and active site of human telomerase, which paves the way for the development of therapeutic agents that target this enzyme. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_12177.map.gz emd_12177.map.gz | 95.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-12177-v30.xml emd-12177-v30.xml emd-12177.xml emd-12177.xml | 26 KB 26 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_12177_fsc.xml emd_12177_fsc.xml | 10.7 KB | Display |  FSC data file FSC data file |
| Images |  emd_12177.png emd_12177.png | 107.4 KB | ||
| Filedesc metadata |  emd-12177.cif.gz emd-12177.cif.gz | 7.8 KB | ||
| Others |  emd_12177_half_map_1.map.gz emd_12177_half_map_1.map.gz emd_12177_half_map_2.map.gz emd_12177_half_map_2.map.gz | 80.7 MB 80.7 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-12177 http://ftp.pdbj.org/pub/emdb/structures/EMD-12177 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12177 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12177 | HTTPS FTP |
-Related structure data
| Related structure data |  7bgbMC  7bg9C C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | |
| EM raw data |  EMPIAR-10732 (Title: Structure of human telomerase holoenzyme with bound telomeric DNA EMPIAR-10732 (Title: Structure of human telomerase holoenzyme with bound telomeric DNAData size: 11.2 TB Data #1: Unaligned multiframe micrographs of human telomerase holoenzyme bound to a telomeric DNA [micrographs - multiframe]) |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_12177.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_12177.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.11 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Half map: #1
| File | emd_12177_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
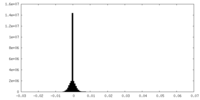
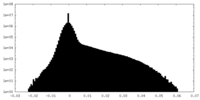
| Density Histograms |
-Half map: #2
| File | emd_12177_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
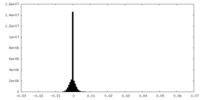
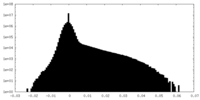
| Density Histograms |
- Sample components
Sample components
-Entire : Human telomerase H/ACA RNP lobe
| Entire | Name: Human telomerase H/ACA RNP lobe |
|---|---|
| Components |
|
-Supramolecule #1: Human telomerase H/ACA RNP lobe
| Supramolecule | Name: Human telomerase H/ACA RNP lobe / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#6 Details: The hTR H/ACA motif, which adopts a double-hairpin structure hinged by the H-box and flanked by the ACA box, scaffolds the assembly of two copies each of dyskerin, NHP2, NOP10 and GAR1, one ...Details: The hTR H/ACA motif, which adopts a double-hairpin structure hinged by the H-box and flanked by the ACA box, scaffolds the assembly of two copies each of dyskerin, NHP2, NOP10 and GAR1, one on each hairpin. TCAB1 binds the CAB box. |
|---|
-Supramolecule #2: ribonucleoprotein and telomerase complex
| Supramolecule | Name: ribonucleoprotein and telomerase complex / type: complex / ID: 2 / Parent: 1 / Details: 1,2,3,5,6 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Supramolecule #3: RNA
| Supramolecule | Name: RNA / type: complex / ID: 3 / Parent: 1 / Details: 4 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Telomerase Cajal body protein 1
| Macromolecule | Name: Telomerase Cajal body protein 1 / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / Organ: Kidney Homo sapiens (human) / Organ: Kidney |
| Molecular weight | Theoretical: 59.35707 KDa |
| Sequence | String: MKTLETQPLA PDCCPSDQDP APAHPSPHAS PMNKNADSEL MPPPPERGDP PRLSPDPVAG SAVSQELREG DPVSLSTPLE TEFGSPSEL SPRIEEQELS ENTSLPAEEA NGSLSEEEAN GPELGSGKAM EDTSGEPAAE DEGDTAWNYS FSQLPRFLSG S WSEFSTQP ...String: MKTLETQPLA PDCCPSDQDP APAHPSPHAS PMNKNADSEL MPPPPERGDP PRLSPDPVAG SAVSQELREG DPVSLSTPLE TEFGSPSEL SPRIEEQELS ENTSLPAEEA NGSLSEEEAN GPELGSGKAM EDTSGEPAAE DEGDTAWNYS FSQLPRFLSG S WSEFSTQP ENFLKGCKWA PDGSCILTNS ADNILRIYNL PPELYHEGEQ VEYAEMVPVL RMVEGDTIYD YCWYSLMSSA QP DTSYVAS SSRENPIHIW DAFTGELRAS FRAYNHLDEL TAAHSLCFSP DGSQLFCGFN RTVRVFSTAR PGRDCEVRAT FAK KQGQSG IISCIAFSPA QPLYACGSYG RSLGLYAWDD GSPLALLGGH QGGITHLCFH PDGNRFFSGA RKDAELLCWD LRQS GYPLW SLGREVTTNQ RIYFDLDPTG QFLVSGSTSG AVSVWDTDGP GNDGKPEPVL SFLPQKDCTN GVSLHPSLPL LATAS GQRV FPEPTESGDE GEELGLPLLS TRHVHLECRL QLWWCGGAPD SSIPDDHQGE KGQGGTEGGV GELI UniProtKB: Telomerase Cajal body protein 1 |
-Macromolecule #2: H/ACA ribonucleoprotein complex subunit DKC1
| Macromolecule | Name: H/ACA ribonucleoprotein complex subunit DKC1 / type: protein_or_peptide / ID: 2 / Number of copies: 2 / Enantiomer: LEVO EC number: Isomerases; Intramolecular transferases; Transferring other groups |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / Organ: Kidney Homo sapiens (human) / Organ: Kidney |
| Molecular weight | Theoretical: 57.779211 KDa |
| Sequence | String: MADAEVIILP KKHKKKKERK SLPEEDVAEI QHAEEFLIKP ESKVAKLDTS QWPLLLKNFD KLNVRTTHYT PLACGSNPLK REIGDYIRT GFINLDKPSN PSSHEVVAWI RRILRVEKTG HSGTLDPKVT GCLIVCIERA TRLVKSQQSA GKEYVGIVRL H NAIEGGTQ ...String: MADAEVIILP KKHKKKKERK SLPEEDVAEI QHAEEFLIKP ESKVAKLDTS QWPLLLKNFD KLNVRTTHYT PLACGSNPLK REIGDYIRT GFINLDKPSN PSSHEVVAWI RRILRVEKTG HSGTLDPKVT GCLIVCIERA TRLVKSQQSA GKEYVGIVRL H NAIEGGTQ LSRALETLTG ALFQRPPLIA AVKRQLRVRT IYESKMIEYD PERRLGIFWV SCEAGTYIRT LCVHLGLLLG VG GQMQELR RVRSGVMSEK DHMVTMHDVL DAQWLYDNHK DESYLRRVVY PLEKLLTSHK RLVMKDSAVN AICYGAKIML PGV LRYEDG IEVNQEIVVI TTKGEAICMA IALMTTAVIS TCDHGIVAKI KRVIMERDTY PRKWGLGPKA SQKKLMIKQG LLDK HGKPT DSTPATWKQE YVDYSESAKK EVVAEVVKAP QVVAEAAKTA KRKRESESES DETPPAAPQL IKKEKKKSKK DKKAK AGLE SGAEPGDGDS DTTKKKKKKK KAKEVELVSE UniProtKB: H/ACA ribonucleoprotein complex subunit DKC1 |
-Macromolecule #3: H/ACA ribonucleoprotein complex subunit 3
| Macromolecule | Name: H/ACA ribonucleoprotein complex subunit 3 / type: protein_or_peptide / ID: 3 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / Organ: Kidney Homo sapiens (human) / Organ: Kidney |
| Molecular weight | Theoretical: 7.719989 KDa |
| Sequence | String: MFLQYYLNEQ GDRVYTLKKF DPMGQQTCSA HPARFSPDDK YSRHRITIKK RFKVLMTQQP RPVL UniProtKB: H/ACA ribonucleoprotein complex subunit 3 |
-Macromolecule #5: H/ACA ribonucleoprotein complex subunit 1
| Macromolecule | Name: H/ACA ribonucleoprotein complex subunit 1 / type: protein_or_peptide / ID: 5 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / Organ: Kidney Homo sapiens (human) / Organ: Kidney |
| Molecular weight | Theoretical: 22.387963 KDa |
| Sequence | String: MSFRGGGRGG FNRGGGGGGF NRGGSSNHFR GGGGGGGGGN FRGGGRGGFG RGGGRGGFNK GQDQGPPERV VLLGEFLHPC EDDIVCKCT TDENKVPYFN APVYLENKEQ IGKVDEIFGQ LRDFYFSVKL SENMKASSFK KLQKFYIDPY KLLPLQRFLP R PPGEKGPP ...String: MSFRGGGRGG FNRGGGGGGF NRGGSSNHFR GGGGGGGGGN FRGGGRGGFG RGGGRGGFNK GQDQGPPERV VLLGEFLHPC EDDIVCKCT TDENKVPYFN APVYLENKEQ IGKVDEIFGQ LRDFYFSVKL SENMKASSFK KLQKFYIDPY KLLPLQRFLP R PPGEKGPP RGGGRGGRGG GRGGGGRGGG RGGGFRGGRG GGGGGFRGGR GGGFRGRGH UniProtKB: H/ACA ribonucleoprotein complex subunit 1 |
-Macromolecule #6: H/ACA ribonucleoprotein complex subunit 2
| Macromolecule | Name: H/ACA ribonucleoprotein complex subunit 2 / type: protein_or_peptide / ID: 6 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / Organ: Kidney Homo sapiens (human) / Organ: Kidney |
| Molecular weight | Theoretical: 17.22607 KDa |
| Sequence | String: MTKIKADPDG PEAQAEACSG ERTYQELLVN QNPIAQPLAS RRLTRKLYKC IKKAVKQKQI RRGVKEVQKF VNKGEKGIMV LAGDTLPIE VYCHLPVMCE DRNLPYVYIP SKTDLGAAAG SKRPTCVIMV KPHEEYQEAY DECLEEVQSL PLPL UniProtKB: H/ACA ribonucleoprotein complex subunit 2 |
-Macromolecule #4: RNA (92-MER)
| Macromolecule | Name: RNA (92-MER) / type: rna / ID: 4 / Number of copies: 1 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / Organ: Kidney Homo sapiens (human) / Organ: Kidney |
| Molecular weight | Theoretical: 145.477797 KDa |
| Sequence | String: GGGUUGCGGA GGGUGGGCCU GGGAGGGGUG GUGGCCAUUU UUUGUCUAAC CCUAACUGAG AAGGGCGUAG GCGCCGUGCU UUUGCUCCC CGCGCGCUGU UUUUCUCGCU GACUUUCAGC GGGCGGAAAA GCCUCGGCCU GCCGCCUUCC ACCGUUCAUU C UAGAGCAA ...String: GGGUUGCGGA GGGUGGGCCU GGGAGGGGUG GUGGCCAUUU UUUGUCUAAC CCUAACUGAG AAGGGCGUAG GCGCCGUGCU UUUGCUCCC CGCGCGCUGU UUUUCUCGCU GACUUUCAGC GGGCGGAAAA GCCUCGGCCU GCCGCCUUCC ACCGUUCAUU C UAGAGCAA ACAAAAAAUG UCAGCUGCUG GCCCGUUCGC CCCUCCCGGG GACCUGCGGC GGGUCGCCUG CCCAGCCCCC GA ACCCCGC CUGGAGGCCG CGGUCGGCCC GGGGCUUCUC CGGAGGCACC CACUGCCACC GCGAAGAGUU GGGCUCUGUC AGC CGCGGG UCUCUCGGGG GCGAGGGCGA GGUUCAGGCC UUUCAGGCCG CAGGAAGAGG AACGGAGCGA GUCCCCGCGC GCGG CGCGA UUCCCUGAGC UGUGGGACGU GCACCCAGGA CUCGGCUCAC ACAUGC |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 Component:
| |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Grid | Model: C-flat / Material: COPPER / Mesh: 400 / Support film - Material: CARBON / Support film - topology: CONTINUOUS / Support film - Film thickness: 5 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 12 sec. / Pretreatment - Atmosphere: AIR | |||||||||||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV / Details: Blot for 4-5 seconds before plunging. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Temperature | Min: 78.0 K |
| Specialist optics | Energy filter - Name: GIF Quantum LS / Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Digitization - Dimensions - Width: 5760 pixel / Digitization - Dimensions - Height: 4092 pixel / Number grids imaged: 2 / Number real images: 43639 / Average exposure time: 1.0 sec. / Average electron dose: 47.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 70.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 3.5 µm / Nominal defocus min: 1.2 µm / Nominal magnification: 81000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: RECIPROCAL / Protocol: AB INITIO MODEL / Overall B value: 104 |
|---|---|
| Output model |  PDB-7bgb: |
 Movie
Movie Controller
Controller


























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)