+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6x93 | ||||||
|---|---|---|---|---|---|---|---|
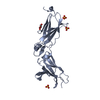
| Title | Interleukin-10 signaling complex with IL-10RA and IL-10RB | ||||||
 Components Components |
| ||||||
 Keywords Keywords | CYTOKINE / IL-10 / receptor / IL-10RA / IL-10RB / signaling | ||||||
| Function / homology |  Function and homology information Function and homology informationinterleukin-10 binding / negative regulation of cytokine activity / negative regulation of chronic inflammatory response to antigenic stimulus / interleukin-10 receptor binding / regulation of response to wounding / interleukin-10 receptor activity / negative regulation of interleukin-18 production / interleukin-28 receptor complex / negative regulation of myeloid dendritic cell activation / negative regulation of interferon-alpha production ...interleukin-10 binding / negative regulation of cytokine activity / negative regulation of chronic inflammatory response to antigenic stimulus / interleukin-10 receptor binding / regulation of response to wounding / interleukin-10 receptor activity / negative regulation of interleukin-18 production / interleukin-28 receptor complex / negative regulation of myeloid dendritic cell activation / negative regulation of interferon-alpha production / negative regulation of chemokine (C-C motif) ligand 5 production / positive regulation of cellular respiration / negative regulation of membrane protein ectodomain proteolysis / response to inactivity / positive regulation of plasma cell differentiation / positive regulation of B cell apoptotic process / chronic inflammatory response to antigenic stimulus / regulation of isotype switching / negative regulation of heterotypic cell-cell adhesion / type III interferon-mediated signaling pathway / negative regulation of cytokine production involved in immune response / negative regulation of MHC class II biosynthetic process / negative regulation of interleukin-1 production / interleukin-10-mediated signaling pathway / branching involved in labyrinthine layer morphogenesis / negative regulation of interleukin-12 production / intestinal epithelial structure maintenance / negative regulation of interleukin-8 production / response to carbon monoxide / negative regulation of nitric oxide biosynthetic process / endothelial cell apoptotic process / positive regulation of MHC class II biosynthetic process / positive regulation of macrophage activation / negative regulation of oxidative stress-induced neuron intrinsic apoptotic signaling pathway / negative regulation of mitotic cell cycle / leukocyte chemotaxis / positive regulation of heterotypic cell-cell adhesion / type 2 immune response / CD163 mediating an anti-inflammatory response / Other interleukin signaling / negative regulation of cytokine production / cellular response to hepatocyte growth factor stimulus / Interleukin-20 family signaling / ubiquitin-dependent endocytosis / positive regulation of immunoglobulin production / negative regulation of B cell proliferation / defense response to protozoan / positive regulation of sprouting angiogenesis / Interleukin-10 signaling / regulation of synapse organization / negative regulation of type II interferon production / negative regulation of interleukin-6 production / hemopoiesis / B cell proliferation / negative regulation of vascular associated smooth muscle cell proliferation / negative regulation of tumor necrosis factor production / coreceptor activity / positive regulation of vascular associated smooth muscle cell proliferation / negative regulation of T cell proliferation / Nuclear events stimulated by ALK signaling in cancer / positive regulation of cell cycle / positive regulation of endothelial cell proliferation / liver regeneration / Gene and protein expression by JAK-STAT signaling after Interleukin-12 stimulation / FCGR3A-mediated IL10 synthesis / B cell differentiation / response to glucocorticoid / cytokine activity / positive regulation of cytokine production / response to activity / negative regulation of autophagy / positive regulation of receptor signaling pathway via JAK-STAT / growth factor activity / cellular response to estradiol stimulus / response to molecule of bacterial origin / cellular response to virus / response to insulin / positive regulation of miRNA transcription / negative regulation of inflammatory response / cytokine-mediated signaling pathway / Signaling by ALK fusions and activated point mutants / cellular response to lipopolysaccharide / signaling receptor activity / regulation of gene expression / Interleukin-4 and Interleukin-13 signaling / response to lipopolysaccharide / defense response to virus / protein dimerization activity / defense response to bacterium / apical plasma membrane / cilium / immune response / response to xenobiotic stimulus / inflammatory response / negative regulation of cell population proliferation / negative regulation of apoptotic process / positive regulation of DNA-templated transcription / signal transduction / positive regulation of transcription by RNA polymerase II / : Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.5 Å | ||||||
 Authors Authors | Saxton, R.A. / Tsutsumi, N. / Gati, C. / Garcia, K.C. | ||||||
| Funding support |  United States, 1items United States, 1items
| ||||||
 Citation Citation |  Journal: Science / Year: 2021 Journal: Science / Year: 2021Title: Structure-based decoupling of the pro- and anti-inflammatory functions of interleukin-10. Authors: Robert A Saxton / Naotaka Tsutsumi / Leon L Su / Gita C Abhiraman / Kritika Mohan / Lukas T Henneberg / Nanda G Aduri / Cornelius Gati / K Christopher Garcia /  Abstract: Interleukin-10 (IL-10) is an immunoregulatory cytokine with both anti-inflammatory and immunostimulatory properties and is frequently dysregulated in disease. We used a structure-based approach to ...Interleukin-10 (IL-10) is an immunoregulatory cytokine with both anti-inflammatory and immunostimulatory properties and is frequently dysregulated in disease. We used a structure-based approach to deconvolute IL-10 pleiotropy by determining the structure of the IL-10 receptor (IL-10R) complex by cryo-electron microscopy at a resolution of 3.5 angstroms. The hexameric structure shows how IL-10 and IL-10Rα form a composite surface to engage the shared signaling receptor IL-10Rβ, enabling the design of partial agonists. IL-10 variants with a range of IL-10Rβ binding strengths uncovered substantial differences in response thresholds across immune cell populations, providing a means of manipulating IL-10 cell type selectivity. Some variants displayed myeloid-biased activity by suppressing macrophage activation without stimulating inflammatory CD8 T cells, thereby uncoupling the major opposing functions of IL-10. These results provide a mechanistic blueprint for tuning the pleiotropic actions of IL-10. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6x93.cif.gz 6x93.cif.gz | 181.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6x93.ent.gz pdb6x93.ent.gz | 132.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6x93.json.gz 6x93.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/x9/6x93 https://data.pdbj.org/pub/pdb/validation_reports/x9/6x93 ftp://data.pdbj.org/pub/pdb/validation_reports/x9/6x93 ftp://data.pdbj.org/pub/pdb/validation_reports/x9/6x93 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  22098MC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | |
| EM raw data |  EMPIAR-10557 (Title: Interleukin-10 signaling complex with IL-10RA and IL-10RB EMPIAR-10557 (Title: Interleukin-10 signaling complex with IL-10RA and IL-10RBData size: 5.4 TB Data #1: Unaligned dark-subtracted TIFF movies with a gain reference for the 3D reconstruction of EMD-22098. [micrographs - multiframe]) |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
| #1: Protein | Mass: 18778.543 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: IL10 / Production host: Homo sapiens (human) / Gene: IL10 / Production host:  Homo sapiens (human) / References: UniProt: P22301 Homo sapiens (human) / References: UniProt: P22301#2: Protein | Mass: 24422.391 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: IL10RA, IL10R / Production host: Homo sapiens (human) / Gene: IL10RA, IL10R / Production host:  Homo sapiens (human) / References: UniProt: Q13651 Homo sapiens (human) / References: UniProt: Q13651#3: Protein | Mass: 23569.334 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: IL10RB, CRFB4, D21S58, D21S66 / Production host: Homo sapiens (human) / Gene: IL10RB, CRFB4, D21S58, D21S66 / Production host:  Trichoplusia ni (cabbage looper) / References: UniProt: Q08334 Trichoplusia ni (cabbage looper) / References: UniProt: Q08334Has protein modification | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component |
| ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular weight | Value: 0.13 MDa / Experimental value: NO | ||||||||||||||||||||||||
| Source (natural) |
| ||||||||||||||||||||||||
| Source (recombinant) |
| ||||||||||||||||||||||||
| Buffer solution | pH: 7.2 | ||||||||||||||||||||||||
| Buffer component |
| ||||||||||||||||||||||||
| Specimen | Conc.: 10 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES | ||||||||||||||||||||||||
| Specimen support | Grid material: GOLD / Grid mesh size: 300 divisions/in. / Grid type: Quantifoil R1.2/1.3 | ||||||||||||||||||||||||
| Vitrification | Instrument: LEICA EM GP / Cryogen name: ETHANE / Humidity: 95 % / Chamber temperature: 293 K / Details: 5s blotting |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal magnification: 81000 X / Nominal defocus max: -2000 nm / Nominal defocus min: -800 nm / Cs: 2.7 mm / C2 aperture diameter: 100 µm / Alignment procedure: COMA FREE |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Electron dose: 50 e/Å2 / Film or detector model: GATAN K3 (6k x 4k) / Num. of grids imaged: 1 / Num. of real images: 9413 |
| EM imaging optics | Energyfilter name: GIF Bioquantum / Energyfilter slit width: 20 eV |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software |
| ||||||||||||||||||||||||||||||
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||||||||
| Particle selection | Num. of particles selected: 6701298 | ||||||||||||||||||||||||||||||
| Symmetry | Point symmetry: C1 (asymmetric) | ||||||||||||||||||||||||||||||
| 3D reconstruction | Resolution: 3.5 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 86725 / Symmetry type: POINT | ||||||||||||||||||||||||||||||
| Atomic model building | Protocol: FLEXIBLE FIT / Space: REAL | ||||||||||||||||||||||||||||||
| Atomic model building |
| ||||||||||||||||||||||||||||||
| Refinement | Cross valid method: NONE Stereochemistry target values: GeoStd + Monomer Library + CDL v1.2 | ||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 76.12 Å2 | ||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller











 PDBj
PDBj