[English] 日本語
 Yorodumi
Yorodumi- PDB-6c26: The Cryo-EM structure of a eukaryotic oligosaccharyl transferase ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6c26 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | The Cryo-EM structure of a eukaryotic oligosaccharyl transferase complex | |||||||||
 Components Components | (Dolichyl-diphosphooligosaccharide--protein glycosyltransferase subunit ...) x 8 | |||||||||
 Keywords Keywords | TRANSFERASE / complex / glycosylation | |||||||||
| Function / homology |  Function and homology information Function and homology informationMiscellaneous transport and binding events / oligosaccharyltransferase I complex / protein O-linked mannosylation / oligosaccharyltransferase complex / dolichyl-diphosphooligosaccharide-protein glycotransferase / dolichyl-diphosphooligosaccharide-protein glycotransferase activity / protein N-linked glycosylation via asparagine / protein N-linked glycosylation / glycosyltransferase activity / protein glycosylation ...Miscellaneous transport and binding events / oligosaccharyltransferase I complex / protein O-linked mannosylation / oligosaccharyltransferase complex / dolichyl-diphosphooligosaccharide-protein glycotransferase / dolichyl-diphosphooligosaccharide-protein glycotransferase activity / protein N-linked glycosylation via asparagine / protein N-linked glycosylation / glycosyltransferase activity / protein glycosylation / protein-disulfide reductase activity / Neutrophil degranulation / post-translational protein modification / protein-macromolecule adaptor activity / nuclear envelope / protein-containing complex assembly / endoplasmic reticulum membrane / endoplasmic reticulum / mitochondrion / membrane / metal ion binding Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.5 Å | |||||||||
 Authors Authors | Bai, L. / Li, H. | |||||||||
| Funding support |  United States, 1items United States, 1items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2018 Journal: Nature / Year: 2018Title: The atomic structure of a eukaryotic oligosaccharyltransferase complex. Authors: Lin Bai / Tong Wang / Gongpu Zhao / Amanda Kovach / Huilin Li /  Abstract: N-glycosylation is a ubiquitous modification of eukaryotic secretory and membrane-bound proteins; about 90% of glycoproteins are N-glycosylated. The reaction is catalysed by an eight-protein ...N-glycosylation is a ubiquitous modification of eukaryotic secretory and membrane-bound proteins; about 90% of glycoproteins are N-glycosylated. The reaction is catalysed by an eight-protein oligosaccharyltransferase (OST) complex that is embedded in the endoplasmic reticulum membrane. Our understanding of eukaryotic protein N-glycosylation has been limited owing to the lack of high-resolution structures. Here we report a 3.5 Å resolution cryo-electron microscopy structure of the Saccharomyces cerevisiae OST complex, revealing the structures of subunits Ost1-Ost5, Stt3, Wbp1 and Swp1. We found that seven phospholipids mediate many of the inter-subunit interactions, and an Stt3 N-glycan mediates interactions with Wbp1 and Swp1 in the lumen. Ost3 was found to mediate the OST-Sec61 translocon interface, funnelling the acceptor peptide towards the OST catalytic site as the nascent peptide emerges from the translocon. The structure provides insights into co-translational protein N-glycosylation, and may facilitate the development of small-molecule inhibitors that target this process. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6c26.cif.gz 6c26.cif.gz | 392.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6c26.ent.gz pdb6c26.ent.gz | 317.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6c26.json.gz 6c26.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  6c26_validation.pdf.gz 6c26_validation.pdf.gz | 1.2 MB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  6c26_full_validation.pdf.gz 6c26_full_validation.pdf.gz | 1.3 MB | Display | |
| Data in XML |  6c26_validation.xml.gz 6c26_validation.xml.gz | 65.4 KB | Display | |
| Data in CIF |  6c26_validation.cif.gz 6c26_validation.cif.gz | 94.4 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/c2/6c26 https://data.pdbj.org/pub/pdb/validation_reports/c2/6c26 ftp://data.pdbj.org/pub/pdb/validation_reports/c2/6c26 ftp://data.pdbj.org/pub/pdb/validation_reports/c2/6c26 | HTTPS FTP |
-Related structure data
| Related structure data |  7336MC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Dolichyl-diphosphooligosaccharide--protein glycosyltransferase subunit ... , 8 types, 8 molecules A15423CB
| #1: Protein | Mass: 81604.539 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Strain: ATCC 204508 / S288c References: UniProt: P39007, dolichyl-diphosphooligosaccharide-protein glycotransferase |
|---|---|
| #2: Protein | Mass: 54116.477 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Strain: ATCC 204508 / S288c References: UniProt: P41543, dolichyl-diphosphooligosaccharide-protein glycotransferase |
| #3: Protein | Mass: 9525.090 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Strain: ATCC 204508 / S288c References: UniProt: Q92316, dolichyl-diphosphooligosaccharide-protein glycotransferase |
| #4: Protein/peptide | Mass: 3986.696 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Strain: ATCC 204508 / S288c References: UniProt: Q99380, dolichyl-diphosphooligosaccharide-protein glycotransferase |
| #5: Protein | Mass: 14712.531 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Strain: ATCC 204508 / S288c References: UniProt: P46964, dolichyl-diphosphooligosaccharide-protein glycotransferase |
| #6: Protein | Mass: 39518.160 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Strain: ATCC 204508 / S288c References: UniProt: P48439, dolichyl-diphosphooligosaccharide-protein glycotransferase |
| #7: Protein | Mass: 31682.832 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Strain: ATCC 204508 / S288c References: UniProt: Q02795, dolichyl-diphosphooligosaccharide-protein glycotransferase |
| #8: Protein | Mass: 49444.438 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Strain: ATCC 204508 / S288c References: UniProt: P33767, dolichyl-diphosphooligosaccharide-protein glycotransferase |
-Sugars , 3 types, 3 molecules 
| #9: Polysaccharide | beta-D-mannopyranose-(1-2)-beta-D-mannopyranose-(1-2)-beta-D-mannopyranose-(1-2)-[beta-D- ...beta-D-mannopyranose-(1-2)-beta-D-mannopyranose-(1-2)-beta-D-mannopyranose-(1-2)-[beta-D-mannopyranose-(1-6)]beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose Source method: isolated from a genetically manipulated source |
|---|---|
| #10: Polysaccharide | beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta- ...beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose Source method: isolated from a genetically manipulated source |
| #12: Sugar | ChemComp-NAG / |
-Non-polymers , 1 types, 8 molecules 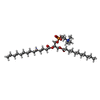
| #11: Chemical | ChemComp-EGY / ( |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: oligosaccharyl transferase complex / Type: COMPLEX / Entity ID: #1-#8 / Source: NATURAL |
|---|---|
| Source (natural) | Organism:  |
| Buffer solution | pH: 7.4 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD |
| Image recording | Electron dose: 2 e/Å2 / Film or detector model: GATAN K2 SUMMIT (4k x 4k) |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.11.1_2575: / Classification: refinement | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software |
| ||||||||||||||||||||||||||||||
| CTF correction | Type: NONE | ||||||||||||||||||||||||||||||
| Particle selection | Num. of particles selected: 823255 | ||||||||||||||||||||||||||||||
| Symmetry | Point symmetry: C1 (asymmetric) | ||||||||||||||||||||||||||||||
| 3D reconstruction | Resolution: 3.5 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 282202 / Symmetry type: POINT | ||||||||||||||||||||||||||||||
| Atomic model building | Protocol: FLEXIBLE FIT | ||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller









 PDBj
PDBj












