登録情報 データベース : PDB / ID : 6jp8タイトル Rabbit Cav1.1-Bay K8644 Complex (Voltage-dependent L-type calcium channel subunit ...) x 2 (Voltage-dependent calcium channel ...) x 2 キーワード / 機能・相同性 分子機能 ドメイン・相同性 構成要素
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / 生物種 Oryctolagus cuniculus (ウサギ)手法 / / / 解像度 : 2.7 Å データ登録者 Zhao, Y. / Huang, G. / Wu, J. / Yan, N. 資金援助 組織 認可番号 国 Ministry of Science and Technology (China) 2016YFA0500402 Ministry of Science and Technology (China) 2015CB910101 National Natural Science Foundation of China 31621092 National Natural Science Foundation of China 31630017 National Natural Science Foundation of China 81861138009 National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) R01GM130762
ジャーナル : Cell / 年 : 2019タイトル : Molecular Basis for Ligand Modulation of a Mammalian Voltage-Gated Ca Channel.著者 : Yanyu Zhao / Gaoxingyu Huang / Jianping Wu / Qiurong Wu / Shuai Gao / Zhen Yan / Jianlin Lei / Nieng Yan / 要旨 : The L-type voltage-gated Ca (Ca) channels are modulated by various compounds exemplified by 1,4-dihydropyridines (DHP), benzothiazepines (BTZ), and phenylalkylamines (PAA), many of which have been ... The L-type voltage-gated Ca (Ca) channels are modulated by various compounds exemplified by 1,4-dihydropyridines (DHP), benzothiazepines (BTZ), and phenylalkylamines (PAA), many of which have been used for characterizing channel properties and for treatment of hypertension and other disorders. Here, we report the cryoelectron microscopy (cryo-EM) structures of Ca1.1 in complex with archetypal antagonistic drugs, nifedipine, diltiazem, and verapamil, at resolutions of 2.9 Å, 3.0 Å, and 2.7 Å, respectively, and with a DHP agonist Bay K 8644 at 2.8 Å. Diltiazem and verapamil traverse the central cavity of the pore domain, directly blocking ion permeation. Although nifedipine and Bay K 8644 occupy the same fenestration site at the interface of repeats III and IV, the coordination details support previous functional observations that Bay K 8644 is less favored in the inactivated state. These structures elucidate the modes of action of different Ca ligands and establish a framework for structure-guided drug discovery. 履歴 登録 2019年3月26日 登録サイト / 処理サイト 改定 1.0 2019年6月12日 Provider / タイプ 改定 1.1 2019年10月23日 Group / Database references / カテゴリ / citation_authorItem _citation.journal_abbrev / _citation.journal_id_CSD ... _citation.journal_abbrev / _citation.journal_id_CSD / _citation.journal_id_ISSN / _citation.journal_volume / _citation.page_first / _citation.page_last / _citation.pdbx_database_id_DOI / _citation.pdbx_database_id_PubMed / _citation.title / _citation.year 改定 1.2 2019年11月6日 Group / Other / カテゴリ / Item 改定 2.0 2020年7月29日 Group Advisory / Atomic model ... Advisory / Atomic model / Data collection / Derived calculations / Structure summary カテゴリ atom_site / chem_comp ... atom_site / chem_comp / em_entity_assembly / entity / pdbx_branch_scheme / pdbx_chem_comp_identifier / pdbx_entity_branch / pdbx_entity_branch_descriptor / pdbx_entity_branch_link / pdbx_entity_branch_list / pdbx_entity_nonpoly / pdbx_nonpoly_scheme / pdbx_struct_assembly_gen / pdbx_struct_conn_angle / pdbx_unobs_or_zero_occ_atoms / pdbx_validate_close_contact / struct_asym / struct_conn / struct_conn_type / struct_site / struct_site_gen Item _atom_site.B_iso_or_equiv / _atom_site.Cartn_x ... _atom_site.B_iso_or_equiv / _atom_site.Cartn_x / _atom_site.Cartn_y / _atom_site.Cartn_z / _atom_site.auth_asym_id / _atom_site.auth_atom_id / _atom_site.auth_comp_id / _atom_site.auth_seq_id / _atom_site.label_asym_id / _atom_site.label_atom_id / _atom_site.label_comp_id / _atom_site.label_entity_id / _atom_site.type_symbol / _chem_comp.name / _chem_comp.type / _em_entity_assembly.entity_id_list / _pdbx_struct_assembly_gen.asym_id_list / _pdbx_struct_conn_angle.ptnr2_label_asym_id / _pdbx_unobs_or_zero_occ_atoms.auth_asym_id / _pdbx_unobs_or_zero_occ_atoms.auth_seq_id / _pdbx_unobs_or_zero_occ_atoms.label_asym_id / _pdbx_validate_close_contact.auth_asym_id_1 / _pdbx_validate_close_contact.auth_asym_id_2 / _pdbx_validate_close_contact.auth_atom_id_1 / _pdbx_validate_close_contact.auth_atom_id_2 / _pdbx_validate_close_contact.auth_seq_id_1 / _pdbx_validate_close_contact.auth_seq_id_2 / _struct_conn.conn_type_id / _struct_conn.id / _struct_conn.pdbx_dist_value / _struct_conn.pdbx_leaving_atom_flag / _struct_conn.pdbx_role / _struct_conn.ptnr1_auth_asym_id / _struct_conn.ptnr1_auth_comp_id / _struct_conn.ptnr1_auth_seq_id / _struct_conn.ptnr1_label_asym_id / _struct_conn.ptnr1_label_atom_id / _struct_conn.ptnr1_label_comp_id / _struct_conn.ptnr1_label_seq_id / _struct_conn.ptnr2_auth_asym_id / _struct_conn.ptnr2_auth_comp_id / _struct_conn.ptnr2_auth_seq_id / _struct_conn.ptnr2_label_asym_id / _struct_conn.ptnr2_label_atom_id / _struct_conn.ptnr2_label_comp_id / _struct_conn_type.id 解説 / Provider / タイプ 改定 2.1 2022年3月23日 Group Author supporting evidence / Database references ... Author supporting evidence / Database references / Derived calculations / Structure summary カテゴリ chem_comp / database_2 ... chem_comp / database_2 / pdbx_audit_support / struct_conn Item _chem_comp.pdbx_synonyms / _database_2.pdbx_DOI ... _chem_comp.pdbx_synonyms / _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _pdbx_audit_support.funding_organization / _struct_conn.pdbx_leaving_atom_flag 改定 3.0 2023年11月15日 Group / Data collection / カテゴリ / chem_comp_atom / chem_comp_bond / Item / _atom_site.label_atom_id改定 3.1 2024年10月9日 Group / Structure summaryカテゴリ / pdbx_entry_details / pdbx_modification_featureItem / _pdbx_entry_details.has_protein_modification
すべて表示 表示を減らす
 データを開く
データを開く 基本情報
基本情報 要素
要素 キーワード
キーワード 機能・相同性情報
機能・相同性情報
 データ登録者
データ登録者 中国, 6件
中国, 6件  引用
引用 ジャーナル: Cell / 年: 2019
ジャーナル: Cell / 年: 2019

 構造の表示
構造の表示 ムービービューア
ムービービューア Molmil
Molmil Jmol/JSmol
Jmol/JSmol ダウンロードとリンク
ダウンロードとリンク ダウンロード
ダウンロード 6jp8.cif.gz
6jp8.cif.gz PDBx/mmCIF形式
PDBx/mmCIF形式 pdb6jp8.ent.gz
pdb6jp8.ent.gz PDB形式
PDB形式 6jp8.json.gz
6jp8.json.gz PDBx/mmJSON形式
PDBx/mmJSON形式 その他のダウンロード
その他のダウンロード 6jp8_validation.pdf.gz
6jp8_validation.pdf.gz wwPDB検証レポート
wwPDB検証レポート 6jp8_full_validation.pdf.gz
6jp8_full_validation.pdf.gz 6jp8_validation.xml.gz
6jp8_validation.xml.gz 6jp8_validation.cif.gz
6jp8_validation.cif.gz https://data.pdbj.org/pub/pdb/validation_reports/jp/6jp8
https://data.pdbj.org/pub/pdb/validation_reports/jp/6jp8 ftp://data.pdbj.org/pub/pdb/validation_reports/jp/6jp8
ftp://data.pdbj.org/pub/pdb/validation_reports/jp/6jp8 リンク
リンク 集合体
集合体
 要素
要素





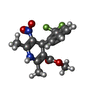

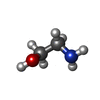


 試料調製
試料調製
 Homo sapiens (ヒト)
Homo sapiens (ヒト) 電子顕微鏡撮影
電子顕微鏡撮影
 FIELD EMISSION GUN / 加速電圧: 300 kV / 照射モード: FLOOD BEAM
FIELD EMISSION GUN / 加速電圧: 300 kV / 照射モード: FLOOD BEAM 解析
解析 ムービー
ムービー コントローラー
コントローラー

















 PDBj
PDBj








