+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: EMDB / ID: EMD-6272 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Single particle cryo-EM structure of rotavirus VP6 at 2.6 Angstroms resolution | |||||||||
 マップデータ マップデータ | Unsharpened reconstruction of rotavirus VP6 | |||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | rotavirus / VP6 / cryo-EM / single particle | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報viral intermediate capsid / T=13 icosahedral viral capsid / host cell surface receptor binding / fusion of virus membrane with host plasma membrane / viral envelope / structural molecule activity / metal ion binding 類似検索 - 分子機能 | |||||||||
| 生物種 |  Bovine rotavirus strain UK/G6 (ウイルス) Bovine rotavirus strain UK/G6 (ウイルス) | |||||||||
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 2.6 Å | |||||||||
 データ登録者 データ登録者 | Grant T / Grigorieff N | |||||||||
 引用 引用 |  ジャーナル: Elife / 年: 2015 ジャーナル: Elife / 年: 2015タイトル: Measuring the optimal exposure for single particle cryo-EM using a 2.6 Å reconstruction of rotavirus VP6. 著者: Timothy Grant / Nikolaus Grigorieff /  要旨: Biological specimens suffer radiation damage when imaged in an electron microscope, ultimately limiting the attainable resolution. At a given resolution, an optimal exposure can be defined that ...Biological specimens suffer radiation damage when imaged in an electron microscope, ultimately limiting the attainable resolution. At a given resolution, an optimal exposure can be defined that maximizes the signal-to-noise ratio in the image. Using a 2.6 Å resolution single particle cryo-EM reconstruction of rotavirus VP6, determined from movies recorded with a total exposure of 100 electrons/Å(2), we obtained accurate measurements of optimal exposure values over a wide range of resolutions. At low and intermediate resolutions, our measured values are considerably higher than obtained previously for crystalline specimens, indicating that both images and movies should be collected with higher exposures than are generally used. We demonstrate a method of using our optimal exposure values to filter movie frames, yielding images with improved contrast that lead to higher resolution reconstructions. This 'high-exposure' technique should benefit cryo-EM work on all types of samples, especially those of relatively low-molecular mass. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| ムービー |
 ムービービューア ムービービューア |
|---|---|
| 構造ビューア | EMマップ:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| 添付画像 |
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_6272.map.gz emd_6272.map.gz | 4.4 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-6272-v30.xml emd-6272-v30.xml emd-6272.xml emd-6272.xml | 10.5 KB 10.5 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| 画像 |  emd_6272.png emd_6272.png emd_6272_1.png emd_6272_1.png | 180.9 KB 145 KB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-6272 http://ftp.pdbj.org/pub/emdb/structures/EMD-6272 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-6272 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-6272 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_6272_validation.pdf.gz emd_6272_validation.pdf.gz | 366.3 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_6272_full_validation.pdf.gz emd_6272_full_validation.pdf.gz | 365.8 KB | 表示 | |
| XML形式データ |  emd_6272_validation.xml.gz emd_6272_validation.xml.gz | 5.1 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-6272 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-6272 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-6272 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-6272 | HTTPS FTP |
-関連構造データ
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_6272.map.gz / 形式: CCP4 / 大きさ: 4.7 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_6272.map.gz / 形式: CCP4 / 大きさ: 4.7 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Unsharpened reconstruction of rotavirus VP6 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.023 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
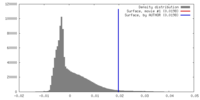
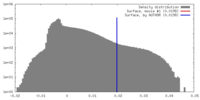
| 密度 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
CCP4マップ ヘッダ情報:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-添付データ
- 試料の構成要素
試料の構成要素
-全体 : 13-fold average of VP6 trimer from full rotavirus reconstruction
| 全体 | 名称: 13-fold average of VP6 trimer from full rotavirus reconstruction |
|---|---|
| 要素 |
|
-超分子 #1000: 13-fold average of VP6 trimer from full rotavirus reconstruction
| 超分子 | 名称: 13-fold average of VP6 trimer from full rotavirus reconstruction タイプ: sample / ID: 1000 / 集合状態: Trimer / Number unique components: 1 |
|---|---|
| 分子量 | 理論値: 41 KDa |
-分子 #1: Bovine rotavirus VP6
| 分子 | 名称: Bovine rotavirus VP6 / タイプ: protein_or_peptide / ID: 1 / Name.synonym: Intermediate capsid protein VP6 / コピー数: 3 / 集合状態: Trimer / 組換発現: Yes |
|---|---|
| 由来(天然) | 生物種:  Bovine rotavirus strain UK/G6 (ウイルス) / 別称: Rv a Bovine rotavirus strain UK/G6 (ウイルス) / 別称: Rv a |
| 分子量 | 理論値: 41 KDa |
| 組換発現 | 生物種:  Chlorocebus sabaeus (オナガザル) Chlorocebus sabaeus (オナガザル) |
| 配列 | UniProtKB: Intermediate capsid protein VP6 |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 濃度 | 2.5 mg/mL |
|---|---|
| グリッド | 詳細: C-Flat 1.2/1.3 |
| 凍結 | 凍結剤: ETHANE / チャンバー内湿度: 80 % / チャンバー内温度: 120 K / 装置: FEI VITROBOT MARK II / 手法: Blot for 4-6 seconds. |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| アライメント法 | Legacy - 非点収差: Corrected at 29,000 times magnification |
| 日付 | 2014年8月13日 |
| 撮影 | カテゴリ: CCD / フィルム・検出器のモデル: GATAN K2 (4k x 4k) / 実像数: 531 / 平均電子線量: 100 e/Å2 詳細: 130-frame movies, 0.1 seconds per frame, 100e/A2 total dose. Super resolution. |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 倍率(補正後): 48876 / 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / Cs: 2.7 mm / 最大 デフォーカス(公称値): 2.0 µm / 最小 デフォーカス(公称値): 0.4 µm / 倍率(公称値): 29000 |
| 試料ステージ | 試料ホルダーモデル: FEI TITAN KRIOS AUTOGRID HOLDER |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
- 画像解析
画像解析
| 詳細 | Additional 13-fold averaging was performed on top of the I symmetry. |
|---|---|
| CTF補正 | 詳細: Each Particle |
| 最終 再構成 | アルゴリズム: OTHER / 解像度のタイプ: BY AUTHOR / 解像度: 2.6 Å / 解像度の算出法: OTHER / ソフトウェア - 名称: TIGRIS, IMAGIC, FREALIGN 詳細: Final map is a 13-fold average of VP6 trimers from the asymmetric unit of the reconstruction of the whole capsid. Data at resolutions higher than 15A were not used for alignments. 使用した粒子像数: 4000 |
 ムービー
ムービー コントローラー
コントローラー



















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)