[English] 日本語
 Yorodumi
Yorodumi- EMDB-5776: Structure of the capsaicin receptor, TRPV1, in complex with DkTx ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-5776 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of the capsaicin receptor, TRPV1, in complex with DkTx and RTX determined by single particle electron cryo-microscopy | |||||||||
 Map data Map data | Reconstruction of rat TRPV1 channel in complex with DkTx and RTX | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | TRPV1 channel / DkTx / RTX | |||||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of iodide transmembrane transport / positive regulation of membrane depolarization / negative regulation of establishment of blood-brain barrier / response to capsazepine / sensory perception of mechanical stimulus / peptide secretion / cellular response to temperature stimulus / positive regulation of sensory perception of pain / temperature-gated ion channel activity / detection of chemical stimulus involved in sensory perception of pain ...negative regulation of iodide transmembrane transport / positive regulation of membrane depolarization / negative regulation of establishment of blood-brain barrier / response to capsazepine / sensory perception of mechanical stimulus / peptide secretion / cellular response to temperature stimulus / positive regulation of sensory perception of pain / temperature-gated ion channel activity / detection of chemical stimulus involved in sensory perception of pain / positive regulation of renal sodium excretion / negative regulation of axon regeneration / TRP channels / positive regulation of cardiac muscle cell differentiation / smooth muscle contraction involved in micturition / fever generation / detection of temperature stimulus involved in thermoception / thermoception / urinary bladder smooth muscle contraction / response to pH / glutamate secretion / monoatomic cation transmembrane transporter activity / negative regulation of systemic arterial blood pressure / positive regulation of urine volume / excitatory extracellular ligand-gated monoatomic ion channel activity / dendritic spine membrane / negative regulation of heart rate / response to acidic pH / response to pain / diet induced thermogenesis / cellular response to alkaloid / temperature homeostasis / cellular response to cytokine stimulus / ion channel inhibitor activity / cellular response to ATP / intracellularly gated calcium channel activity / detection of temperature stimulus involved in sensory perception of pain / negative regulation of mitochondrial membrane potential / calcium ion import across plasma membrane / behavioral response to pain / positive regulation of vasoconstriction / sodium channel regulator activity / potassium channel regulator activity / monoatomic ion channel activity / ligand-gated monoatomic ion channel activity / monoatomic cation channel activity / cellular response to acidic pH / extracellular ligand-gated monoatomic ion channel activity / phosphatidylinositol binding / sensory perception of pain / axon terminus / positive regulation of excitatory postsynaptic potential / sarcoplasmic reticulum / lipid metabolic process / phosphoprotein binding / microglial cell activation / cellular response to nerve growth factor stimulus / response to peptide hormone / cellular response to growth factor stimulus / GABA-ergic synapse / cellular response to tumor necrosis factor / calcium ion transmembrane transport / calcium channel activity / positive regulation of nitric oxide biosynthetic process / calcium ion transport / transmembrane signaling receptor activity / cellular response to heat / sensory perception of taste / toxin activity / response to heat / positive regulation of cytosolic calcium ion concentration / monoatomic ion transmembrane transport / protein homotetramerization / calmodulin binding / postsynaptic membrane / neuron projection / positive regulation of apoptotic process / external side of plasma membrane / neuronal cell body / dendrite / negative regulation of transcription by RNA polymerase II / extracellular region / ATP binding / membrane / metal ion binding / identical protein binding / nucleus / plasma membrane Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.8 Å | |||||||||
 Authors Authors | Liao M / Cao E / Julius D / Cheng Y | |||||||||
 Citation Citation |  Journal: Nature / Year: 2013 Journal: Nature / Year: 2013Title: TRPV1 structures in distinct conformations reveal activation mechanisms. Authors: Erhu Cao / Maofu Liao / Yifan Cheng / David Julius /  Abstract: Transient receptor potential (TRP) channels are polymodal signal detectors that respond to a wide range of physical and chemical stimuli. Elucidating how these channels integrate and convert ...Transient receptor potential (TRP) channels are polymodal signal detectors that respond to a wide range of physical and chemical stimuli. Elucidating how these channels integrate and convert physiological signals into channel opening is essential to understanding how they regulate cell excitability under normal and pathophysiological conditions. Here we exploit pharmacological probes (a peptide toxin and small vanilloid agonists) to determine structures of two activated states of the capsaicin receptor, TRPV1. A domain (consisting of transmembrane segments 1-4) that moves during activation of voltage-gated channels remains stationary in TRPV1, highlighting differences in gating mechanisms for these structurally related channel superfamilies. TRPV1 opening is associated with major structural rearrangements in the outer pore, including the pore helix and selectivity filter, as well as pronounced dilation of a hydrophobic constriction at the lower gate, suggesting a dual gating mechanism. Allosteric coupling between upper and lower gates may account for rich physiological modulation exhibited by TRPV1 and other TRP channels. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_5776.map.gz emd_5776.map.gz | 45.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-5776-v30.xml emd-5776-v30.xml emd-5776.xml emd-5776.xml | 10.1 KB 10.1 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_5776_1.jpg emd_5776_1.jpg | 133.3 KB | ||
| Others |  emd_5776_additional_1.map.gz emd_5776_additional_1.map.gz | 58.6 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-5776 http://ftp.pdbj.org/pub/emdb/structures/EMD-5776 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-5776 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-5776 | HTTPS FTP |
-Related structure data
| Related structure data |  3j5qMC  5777C  3j5rC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_5776.map.gz / Format: CCP4 / Size: 62.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_5776.map.gz / Format: CCP4 / Size: 62.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Reconstruction of rat TRPV1 channel in complex with DkTx and RTX | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.2156 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
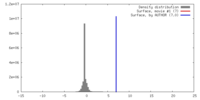
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Supplemental map: emd 5776 additional 1.map
| File | emd_5776_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
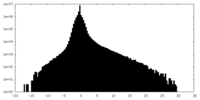
| Density Histograms |
- Sample components
Sample components
-Entire : Rat TRPV1 in complex with DkTx and resiniferatoxin
| Entire | Name: Rat TRPV1 in complex with DkTx and resiniferatoxin |
|---|---|
| Components |
|
-Supramolecule #1000: Rat TRPV1 in complex with DkTx and resiniferatoxin
| Supramolecule | Name: Rat TRPV1 in complex with DkTx and resiniferatoxin / type: sample / ID: 1000 / Details: The sample was monodisperse / Oligomeric state: tetramer / Number unique components: 1 |
|---|---|
| Molecular weight | Experimental: 300 KDa / Theoretical: 300 KDa |
-Macromolecule #1: TRPV1
| Macromolecule | Name: TRPV1 / type: protein_or_peptide / ID: 1 Details: Functional minimal construct containing residues 110-603 and 627-764. Number of copies: 1 / Oligomeric state: Tetramer / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Experimental: 300 KDa / Theoretical: 300 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) / Recombinant cell: HEK293S GnTI / Recombinant plasmid: pFastBac1 Homo sapiens (human) / Recombinant cell: HEK293S GnTI / Recombinant plasmid: pFastBac1 |
| Sequence | UniProtKB: Transient receptor potential cation channel subfamily V member 1 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.3 mg/mL |
|---|---|
| Buffer | pH: 7.4 / Details: 150 mM NaCl, 20 mM HEPES, 2 mM TCEP |
| Grid | Details: 400 mesh Quantifoil grid |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 90 % / Chamber temperature: 120 K / Instrument: FEI VITROBOT MARK III / Method: Blot for 6 sec |
- Electron microscopy
Electron microscopy
| Microscope | FEI POLARA 300 |
|---|---|
| Details | Gatan K2 Summit operated in super-resolution counting mode; image recorded with dose fractionation method. |
| Date | Jan 1, 2013 |
| Image recording | Category: CCD / Film or detector model: GATAN K2 (4k x 4k) / Number real images: 900 / Average electron dose: 21 e/Å2 Details: Every image is the average of 30 frames recorded using the K2 Summit. The final reconstruction was calculated from images averaged from frames #3-#16. |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated magnification: 31000 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.0 mm / Nominal defocus max: 3.0 µm / Nominal defocus min: 1.5 µm / Nominal magnification: 31000 |
| Sample stage | Specimen holder: Cooled to Liquid Nitrogen temperature / Specimen holder model: OTHER |
| Experimental equipment |  Model: Tecnai Polara / Image courtesy: FEI Company |
- Image processing
Image processing
| Details | 3D classification, refinement, and reconstruction were performed using RELION. |
|---|---|
| CTF correction | Details: Each particle |
| Final reconstruction | Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 3.8 Å / Resolution method: OTHER / Software - Name: RELION / Number images used: 36158 |
 Movie
Movie Controller
Controller















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)