+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-23385 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Thermotoga maritima Encapsulin Nanocompartment Pore Mutant S7G | ||||||||||||
 Map data Map data | |||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | Encapsulin / Nanocompartment / VIRUS LIKE PARTICLE | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationencapsulin nanocompartment / Hydrolases; Acting on peptide bonds (peptidases) / iron ion transport / peptidase activity / intracellular iron ion homeostasis / proteolysis Similarity search - Function | ||||||||||||
| Biological species |   Thermotoga maritima MSB8 (bacteria) / Thermotoga maritima MSB8 (bacteria) /   Thermotoga maritima (strain ATCC 43589 / MSB8 / DSM 3109 / JCM 10099) (bacteria) Thermotoga maritima (strain ATCC 43589 / MSB8 / DSM 3109 / JCM 10099) (bacteria) | ||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.53 Å | ||||||||||||
 Authors Authors | Andreas MP / Adamson L | ||||||||||||
| Funding support |  Australia, Australia,  United States, 3 items United States, 3 items
| ||||||||||||
 Citation Citation |  Journal: Sci Adv / Year: 2022 Journal: Sci Adv / Year: 2022Title: Pore structure controls stability and molecular flux in engineered protein cages. Authors: Lachlan S R Adamson / Nuren Tasneem / Michael P Andreas / William Close / Eric N Jenner / Taylor N Szyszka / Reginald Young / Li Chen Cheah / Alexander Norman / Hugo I MacDermott-Opeskin / ...Authors: Lachlan S R Adamson / Nuren Tasneem / Michael P Andreas / William Close / Eric N Jenner / Taylor N Szyszka / Reginald Young / Li Chen Cheah / Alexander Norman / Hugo I MacDermott-Opeskin / Megan L O'Mara / Frank Sainsbury / Tobias W Giessen / Yu Heng Lau /   Abstract: Protein cages are a common architectural motif used by living organisms to compartmentalize and control biochemical reactions. While engineered protein cages have featured in the construction of ...Protein cages are a common architectural motif used by living organisms to compartmentalize and control biochemical reactions. While engineered protein cages have featured in the construction of nanoreactors and synthetic organelles, relatively little is known about the underlying molecular parameters that govern stability and flux through their pores. In this work, we systematically designed 24 variants of the encapsulin cage, featuring pores of different sizes and charges. Twelve pore variants were successfully assembled and purified, including eight designs with exceptional thermal stability. While negatively charged mutations were better tolerated, we were able to form stable assemblies covering a full range of pore sizes and charges, as observed in seven new cryo-EM structures at 2.5- to 3.6-Å resolution. Molecular dynamics simulations and stopped-flow experiments revealed the importance of considering both pore size and charge, together with flexibility and rate-determining steps, when designing protein cages for controlling molecular flux. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_23385.map.gz emd_23385.map.gz | 112.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-23385-v30.xml emd-23385-v30.xml emd-23385.xml emd-23385.xml | 15.5 KB 15.5 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_23385.png emd_23385.png | 97.3 KB | ||
| Filedesc metadata |  emd-23385.cif.gz emd-23385.cif.gz | 6.1 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-23385 http://ftp.pdbj.org/pub/emdb/structures/EMD-23385 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-23385 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-23385 | HTTPS FTP |
-Related structure data
| Related structure data |  7litMC  7liiC  7lijC  7likC  7lilC  7limC  7lisC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_23385.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_23385.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.986 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Thermotoga maritima Encapsulin Nanocompartment Pore Mutant S7G
| Entire | Name: Thermotoga maritima Encapsulin Nanocompartment Pore Mutant S7G |
|---|---|
| Components |
|
-Supramolecule #1: Thermotoga maritima Encapsulin Nanocompartment Pore Mutant S7G
| Supramolecule | Name: Thermotoga maritima Encapsulin Nanocompartment Pore Mutant S7G type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 Details: Thermotoga maratima encapsulin pore mutant with K182G, L190G, and deletion of amino acids K182-P189 |
|---|---|
| Source (natural) | Organism:   Thermotoga maritima MSB8 (bacteria) Thermotoga maritima MSB8 (bacteria) |
| Molecular weight | Theoretical: 1.796591 MDa |
-Macromolecule #1: Maritimacin
| Macromolecule | Name: Maritimacin / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO / EC number: Hydrolases; Acting on peptide bonds (peptidases) |
|---|---|
| Source (natural) | Organism:   Thermotoga maritima (strain ATCC 43589 / MSB8 / DSM 3109 / JCM 10099) (bacteria) Thermotoga maritima (strain ATCC 43589 / MSB8 / DSM 3109 / JCM 10099) (bacteria)Strain: ATCC 43589 / MSB8 / DSM 3109 / JCM 10099 |
| Molecular weight | Theoretical: 29.603766 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MEFLKRSFAP LTEKQWQEID NRAREIFKTQ LYGRKFVDVE GPYGWEYAAH PLGEVEVLSD ENEVVKWGLR KSLPLIELRA TFTLDLWEL DNLERGKPNV DLSSLEETVR KVAEFEDEVI FRGCEKSGVK GLLSFEERKI ECGSTPKDLL EAIVRALSIF S KDGIEGPY ...String: MEFLKRSFAP LTEKQWQEID NRAREIFKTQ LYGRKFVDVE GPYGWEYAAH PLGEVEVLSD ENEVVKWGLR KSLPLIELRA TFTLDLWEL DNLERGKPNV DLSSLEETVR KVAEFEDEVI FRGCEKSGVK GLLSFEERKI ECGSTPKDLL EAIVRALSIF S KDGIEGPY TLVINTDRWI NFLGGEKRVE ECLRGGKIIT TPRIEDALVV SERGGDFKLI LGQDLSIGYE DREKDAVRLF IT ETFTFQV VNPEALILLK F UniProtKB: Type 1 encapsulin shell protein |
-Macromolecule #2: RIBOFLAVIN
| Macromolecule | Name: RIBOFLAVIN / type: ligand / ID: 2 / Number of copies: 1 / Formula: RBF |
|---|---|
| Molecular weight | Theoretical: 376.364 Da |
| Chemical component information | 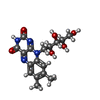 ChemComp-RBF: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 2.0 mg/mL | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 8 Component:
| |||||||||
| Grid | Model: C-flat-2/2 / Support film - Material: CARBON / Support film - topology: HOLEY | |||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 95 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV / Details: blot force 0, wait time 30 seconds. |
- Electron microscopy
Electron microscopy
| Microscope | TFS TALOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON III (4k x 4k) / Average exposure time: 47.84 sec. / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: -2.5 µm / Nominal defocus min: -1.0 µm |
| Sample stage | Cooling holder cryogen: NITROGEN |
- Image processing
Image processing
| Startup model | Type of model: PDB ENTRY PDB model - PDB ID: Details: Initial map for refinements made from PDB entry 3DKT |
|---|---|
| Final reconstruction | Applied symmetry - Point group: I (icosahedral) / Resolution.type: BY AUTHOR / Resolution: 2.53 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: cryoSPARC (ver. 2.15.0) / Number images used: 90905 |
| Initial angle assignment | Type: ANGULAR RECONSTITUTION / Software - Name: cryoSPARC (ver. 2.15.0) |
| Final angle assignment | Type: ANGULAR RECONSTITUTION / Software - Name: cryoSPARC (ver. 2.15.0) |
-Atomic model buiding 1
| Initial model | PDB ID: Chain - Chain ID: A / Chain - Residue range: 1-265 / Chain - Source name: PDB / Chain - Initial model type: experimental model |
|---|---|
| Refinement | Space: REAL / Protocol: FLEXIBLE FIT / Overall B value: 61.68 |
| Output model |  PDB-7lit: |
 Movie
Movie Controller
Controller



















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)






















