[English] 日本語
 Yorodumi
Yorodumi- PDB-1zgb: Crystal Structure of Torpedo Californica Acetylcholinesterase in ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1zgb | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal Structure of Torpedo Californica Acetylcholinesterase in Complex With an (R)-Tacrine(10)-Hupyridone Inhibitor. | ||||||
 Components Components | Acetylcholinesterase | ||||||
 Keywords Keywords | HYDROLASE / SERINE-HYDROLASE / PROTEIN-INHIBITOR COMPLEX / ENANTIOMERIC SELECTIVITY / Israel Structural Proteomics Center / ISPC / Structural Genomics | ||||||
| Function / homology |  Function and homology information Function and homology informationacetylcholine catabolic process in synaptic cleft / acetylcholinesterase / choline metabolic process / acetylcholinesterase activity / side of membrane / synaptic cleft / synapse / : / plasma membrane Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 2.3 Å MOLECULAR REPLACEMENT / Resolution: 2.3 Å | ||||||
 Authors Authors | Haviv, H. / Wong, D.M. / Greenblatt, H.M. / Carlier, P.R. / Pang, Y.P. / Silman, I. / Sussman, J.L. / Israel Structural Proteomics Center (ISPC) | ||||||
 Citation Citation |  Journal: J.Am.Chem.Soc. / Year: 2005 Journal: J.Am.Chem.Soc. / Year: 2005Title: Crystal Packing Mediates Enantioselective Ligand Recognition at the Peripheral Site of Acetylcholinesterase Authors: Haviv, H. / Wong, D.M. / Greenblatt, H.M. / Carlier, P.R. / Pang, Y.P. / Silman, I. / Sussman, J.L. #1: Journal: BIOORG.MED.CHEM.LETT. / Year: 1999 Title: Potent, easily synthesized huperzine A-tacrine hybrid acetylcholinesterase inhibitors Authors: Carlier, P.R. / Du, D.-M. / Han, Y.-F. / Liu, J. / Pang, Y.-P. #2:  Journal: J.Am.Chem.Soc. / Year: 2003 Journal: J.Am.Chem.Soc. / Year: 2003Title: Acetylcholinesterase Complexed with Bivalent Ligands Related to Huperzine A: Experimental Evidence for Species-Dependent Protein-Ligand Complementarity Authors: Wong, D.M. / Greenblatt, H.M. / Dvir, H. / Carlier, P.R. / Han, Y.-F. / Pang, Y.-P. / Silman, I. / Sussman, J.L. #3:  Journal: NAT.STRUCT.BIOL. / Year: 1997 Journal: NAT.STRUCT.BIOL. / Year: 1997Title: Structure of acetylcholinesterase complexed with the nootropic alkaloid, (-)-huperzine A Authors: Raves, M.L. / Harel, M. / Pang, Y.-P. / Silman, I. / Kozikowski, A.P. / Sussman, J.L. #4: Journal: Science / Year: 1991 Title: Atomic structure of acetylcholinesterase from Torpedo californica: a prototypic acetylcholine-binding protein Authors: Sussman, J.L. / Harel, M. / Frolow, F. / Oefner, C. / Goldman, A. / Toker, L. / Silman, I. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1zgb.cif.gz 1zgb.cif.gz | 123.9 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1zgb.ent.gz pdb1zgb.ent.gz | 94.5 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1zgb.json.gz 1zgb.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/zg/1zgb https://data.pdbj.org/pub/pdb/validation_reports/zg/1zgb ftp://data.pdbj.org/pub/pdb/validation_reports/zg/1zgb ftp://data.pdbj.org/pub/pdb/validation_reports/zg/1zgb | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 61325.090 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P04058, acetylcholinesterase | ||||||
|---|---|---|---|---|---|---|---|
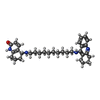
| #2: Sugar | | #3: Chemical | ChemComp-A1E / ( | #4: Water | ChemComp-HOH / | Has protein modification | Y | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 4.07 Å3/Da / Density % sol: 69.7 % |
|---|---|
| Crystal grow | Temperature: 277 K Details: PEG 200, pH 5.8, temperature 277K, VAPOR DIFFUSION, HANGING DROP. PROTEIN WAS CRYSTALLISED FROM 28% V/V PEG 200 0.5 M MES PH 5.8 AT 277K, SEEDING WITH TRIGONAL MICROCRYSTALS; THEN SOAKED IN ...Details: PEG 200, pH 5.8, temperature 277K, VAPOR DIFFUSION, HANGING DROP. PROTEIN WAS CRYSTALLISED FROM 28% V/V PEG 200 0.5 M MES PH 5.8 AT 277K, SEEDING WITH TRIGONAL MICROCRYSTALS; THEN SOAKED IN MOTHER LIQUOR (40% V/V PEG 200 IN 0.1 M MES BUFFER, PH 5.8) CONTAINING 2 MM (RS)-(+/-)- TACRINE(10)-HUPYRIDONE ((5RS)-(+/-)-5-{[10-(1,2,3,4-TETRAHYDROACRIDIN-9-YLAMINO) DECYL] AMINO}-5,6,7,8-TETRAHYDRO-QUINOLIN-2(1H)-ONE) BIS-OXALATE FOR 17 HOURS. |
-Data collection
| Diffraction | Mean temperature: 120 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU RUH3R / Wavelength: 1.5418 ROTATING ANODE / Type: RIGAKU RUH3R / Wavelength: 1.5418 |
| Detector | Type: RIGAKU RAXIS IV / Detector: IMAGE PLATE / Date: Sep 7, 2000 / Details: OSMIC BLUE CONFOCAL MIRRORS |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.5418 Å / Relative weight: 1 |
| Reflection | Resolution: 2.3→30 Å / Num. obs: 44250 / % possible obs: 98.5 % / Redundancy: 1.59 % / Biso Wilson estimate: 28.8 Å2 / Rmerge(I) obs: 0.068 / Net I/σ(I): 21.9 |
| Reflection shell | Resolution: 2.3→2.38 Å / Redundancy: 10 % / Rmerge(I) obs: 0.367 / Mean I/σ(I) obs: 5.16 / % possible all: 99.7 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT / Resolution: 2.3→29.19 Å / Rfactor Rfree error: 0.005 / Data cutoff high absF: 1962219.64 / Data cutoff low absF: 0 / Isotropic thermal model: RESTRAINED / Cross valid method: THROUGHOUT / σ(F): 0 MOLECULAR REPLACEMENT / Resolution: 2.3→29.19 Å / Rfactor Rfree error: 0.005 / Data cutoff high absF: 1962219.64 / Data cutoff low absF: 0 / Isotropic thermal model: RESTRAINED / Cross valid method: THROUGHOUT / σ(F): 0
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Solvent model: FLAT MODEL / Bsol: 35.49 Å2 / ksol: 0.35 e/Å3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 39.3 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine analyze |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.3→29.19 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.3→2.38 Å / Rfactor Rfree error: 0.014 / Total num. of bins used: 6
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Xplor file |
|
 Movie
Movie Controller
Controller













 PDBj
PDBj