+Search query
-Structure paper
| Title | CSN5i-3 is an orthosteric molecular glue inhibitor of COP9 signalosome. |
|---|---|
| Journal, issue, pages | Nature, Vol. 652, Issue 8112, Page 1375-1383, Year 2026 |
| Publish date | Feb 11, 2026 |
 Authors Authors | Huigang Shi / Xiaorong Wang / Clinton Yu / Haibin Mao / Fenglong Jiao / Merav Braitbard / Ben Shor / Zhongsheng Zhang / Thomas R Hinds / Shiyun Cao / Erkang Fan / Dina Schneidman-Duhovny / Lan Huang / Ning Zheng /   |
| PubMed Abstract | Orthosteric inhibitors block enzyme active sites and prevent substrates from binding. Enhancing their specificity through substrate dependence seems inherently unlikely, as their mechanism hinges on ...Orthosteric inhibitors block enzyme active sites and prevent substrates from binding. Enhancing their specificity through substrate dependence seems inherently unlikely, as their mechanism hinges on direct competition rather than selective recognition. Here we show that a molecular glue mechanism unexpectedly imparts substrate-dependent potency to CSN5i-3, an orthosteric inhibitor of the COP9 signalosome (CSN). We first confirm that CSN5i-3 inhibits CSN, which catalyses NEDD8 (N8) deconjugation from the cullin-RING ubiquitin ligases, by occupying the active site of its catalytic subunit, CSN5, and directly competing with the iso-peptide bond substrate. Notably, the orthosteric inhibitor binds free CSN with only micromolar affinity, yet achieves nanomolar potency in blocking its deneddylase activity. Cryogenic electron microscopy structures of the enzyme-substrate-inhibitor complex reveal that active site-engaged CSN5i-3 occludes the substrate iso-peptide linkage while simultaneously extending an N8-binding exosite of CSN5, acting as a molecular glue to cement the N8-CSN5 interaction. The cooperativity of this trimolecular CSN5i-3-N8-CSN5 assembly, in turn, sequesters CSN5i-3 at its binding site, conferring high potency to the orthosteric inhibitor despite its low affinity for the free enzyme. Together, our findings highlight the modest affinity requirements of molecule glues for individual target proteins and establish orthosteric molecular glue inhibitors as a new class of substrate-dependent enzyme antagonists. |
 External links External links |  Nature / Nature /  PubMed:41673158 / PubMed:41673158 /  PubMed Central PubMed Central |
| Methods | EM (single particle) |
| Resolution | 2.96 - 4.6 Å |
| Structure data | EMDB-47532, PDB-9e5z: EMDB-47660, PDB-9e77:  EMDB-47663: Cryo-EM structure of COP9 signalosome deneddylation state with cullin-5  EMDB-47665: Focused map of COP9 signalosome deneddylation state with cullin-5 EMDB-47698, PDB-9e81:  EMDB-47699: Focused map of COP9 signalosome deneddylation state with cullin-4A  EMDB-47701: Cryo-EM structure of COP9 signalosome deneddylation complex with cullin-2  EMDB-47702: Focused map of COP9 signalosome deneddylation complex with cullin-2  EMDB-47729: Cryo-EM structure of CSN-N8CUL1 complex with CSN5i-3  EMDB-47767: Focused map of CSN-N8CUL1 in complex with CSN5i-3  EMDB-47776: Cryo-EM structure of COP9 signalosome deneddylation complex with cullin-3 EMDB-47976, PDB-9efm: EMDB-47977, PDB-9efq: EMDB-47981, PDB-9efv: EMDB-47983, PDB-9eg1:  EMDB-47985: Focused map of COP9 signalosome deneddylation complex with neddylated cullin-3 EMDB-47986, PDB-9eg8: EMDB-47990, PDB-9egl: EMDB-71639, PDB-9ph4: |
| Chemicals |  ChemComp-ZN: 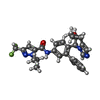 ChemComp-6LT:  PDB-1ch3: |
| Source |
|
 Keywords Keywords | METAL BINDING PROTEIN / COP9 / COP9 signalosome / signalosome / deneddylation / CSN5 / metalloprotease / SIGNALING PROTEIN / NEDD8 / CSN5i-3 / N8CUL1 / CSN / cullin-2 / CSN-N8CUL2 / CUL2 / othosteric molecular glue / N8CUL5 / CUL5 / N8CUL4A / CUL4A / deneddylation complex / CSN5i-1a |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers























 homo sapiens (human)
homo sapiens (human)