+検索条件
-Structure paper
| タイトル | Cryo-EM reveals a mechanism of USP1 inhibition through a cryptic binding site. |
|---|---|
| ジャーナル・号・ページ | Sci Adv, Vol. 8, Issue 39, Page eabq6353, Year 2022 |
| 掲載日 | 2022年9月30日 |
 著者 著者 | Martin L Rennie / Connor Arkinson / Viduth K Chaugule / Helen Walden /  |
| PubMed 要旨 | Repair of DNA damage is critical to genomic integrity and frequently disrupted in cancers. Ubiquitin-specific protease 1 (USP1), a nucleus-localized deubiquitinase, lies at the interface of multiple ...Repair of DNA damage is critical to genomic integrity and frequently disrupted in cancers. Ubiquitin-specific protease 1 (USP1), a nucleus-localized deubiquitinase, lies at the interface of multiple DNA repair pathways and is a promising drug target for certain cancers. Although multiple inhibitors of this enzyme, including one in phase 1 clinical trials, have been established, their binding mode is unknown. Here, we use cryo-electron microscopy to study an assembled enzyme-substrate-inhibitor complex of USP1 and the well-established inhibitor, ML323. Achieving 2.5-Å resolution, with and without ML323, we find an unusual binding mode in which the inhibitor disrupts part of the hydrophobic core of USP1. The consequent conformational changes in the secondary structure lead to subtle rearrangements in the active site that underlie the mechanism of inhibition. These structures provide a platform for structure-based drug design targeting USP1. |
 リンク リンク |  Sci Adv / Sci Adv /  PubMed:36170365 / PubMed:36170365 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 2.49 - 2.85 Å |
| 構造データ |  EMDB-14719: FANCD2 Middle and C-terminal domains with USP1-NTE (focused refinement) EMDB-14720, PDB-7zh3: EMDB-14721, PDB-7zh4: EMDB-14722, PDB-8a9k: EMDB-15284, PDB-8a9j: |
| 化合物 |  ChemComp-ZN:  ChemComp-HOH: 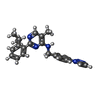 ChemComp-JDA: |
| 由来 |
|
 キーワード キーワード | HYDROLASE / Deubiquitinase / Complex / Enzyme-Substrate / Inhibitor |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について











 homo sapiens (ヒト)
homo sapiens (ヒト)