[English] 日本語
 Yorodumi
Yorodumi- EMDB-4370: Unique features of mammalian mitochondrial translation initiation... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-4370 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Unique features of mammalian mitochondrial translation initiation revealed by cryo-EM. This file contains the 39S ribosomal subunit. | ||||||||||||
 Map data Map data | |||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | translation initiation / initiation factor IF2 / mitochondria / membrane targeting / RIBOSOME | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationMitochondrial translation elongation / Mitochondrial translation termination / mitochondrial translational initiation / mitochondrial transcription / translation factor activity, RNA binding / mitochondrial translational elongation / mitochondrial translational termination / ribonuclease III activity / microprocessor complex / translation release factor activity, codon nonspecific ...Mitochondrial translation elongation / Mitochondrial translation termination / mitochondrial translational initiation / mitochondrial transcription / translation factor activity, RNA binding / mitochondrial translational elongation / mitochondrial translational termination / ribonuclease III activity / microprocessor complex / translation release factor activity, codon nonspecific / Mitochondrial translation initiation / Mitochondrial protein degradation / mitochondrial large ribosomal subunit / ribosome disassembly / peptidyl-tRNA hydrolase / regulation of translational initiation / aminoacyl-tRNA hydrolase activity / mitochondrial ribosome / organelle membrane / mitochondrial translation / ribosomal small subunit binding / RNA processing / translation initiation factor activity / rescue of stalled ribosome / large ribosomal subunit / double-stranded RNA binding / cell junction / cytosolic large ribosomal subunit / nuclear body / rRNA binding / ribosome / structural constituent of ribosome / ribonucleoprotein complex / translation / protein domain specific binding / nucleotide binding / GTPase activity / synapse / GTP binding / positive regulation of DNA-templated transcription / mitochondrion / RNA binding / nucleoplasm / nucleus / plasma membrane / cytosol / cytoplasm Similarity search - Function | ||||||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | ||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.2 Å | ||||||||||||
 Authors Authors | Kummer E / Leibundgut M | ||||||||||||
| Funding support |  Switzerland, 3 items Switzerland, 3 items
| ||||||||||||
 Citation Citation |  Journal: Nature / Year: 2018 Journal: Nature / Year: 2018Title: Unique features of mammalian mitochondrial translation initiation revealed by cryo-EM. Authors: Eva Kummer / Marc Leibundgut / Oliver Rackham / Richard G Lee / Daniel Boehringer / Aleksandra Filipovska / Nenad Ban /   Abstract: Mitochondria maintain their own specialized protein synthesis machinery, which in mammals is used exclusively for the synthesis of the membrane proteins responsible for oxidative phosphorylation. The ...Mitochondria maintain their own specialized protein synthesis machinery, which in mammals is used exclusively for the synthesis of the membrane proteins responsible for oxidative phosphorylation. The initiation of protein synthesis in mitochondria differs substantially from bacterial or cytosolic translation systems. Mitochondrial translation initiation lacks initiation factor 1, which is essential in all other translation systems from bacteria to mammals. Furthermore, only one type of methionyl transfer RNA (tRNA) is used for both initiation and elongation, necessitating that the initiation factor specifically recognizes the formylated version of tRNA (fMet-tRNA). Lastly, most mitochondrial mRNAs do not possess 5' leader sequences to promote mRNA binding to the ribosome. There is currently little mechanistic insight into mammalian mitochondrial translation initiation, and it is not clear how mRNA engagement, initiator-tRNA recruitment and start-codon selection occur. Here we determine the cryo-EM structure of the complete translation initiation complex from mammalian mitochondria at 3.2 Å. We describe the function of an additional domain insertion that is present in the mammalian mitochondrial initiation factor 2 (mtIF2). By closing the decoding centre, this insertion stabilizes the binding of leaderless mRNAs and induces conformational changes in the rRNA nucleotides involved in decoding. We identify unique features of mtIF2 that are required for specific recognition of fMet-tRNA and regulation of its GTPase activity. Finally, we observe that the ribosomal tunnel in the initiating ribosome is blocked by insertion of the N-terminal portion of mitochondrial protein mL45, which becomes exposed as the ribosome switches to elongation mode and may have an additional role in targeting of mitochondrial ribosomes to the protein-conducting pore in the inner mitochondrial membrane. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_4370.map.gz emd_4370.map.gz | 15.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-4370-v30.xml emd-4370-v30.xml emd-4370.xml emd-4370.xml | 88.4 KB 88.4 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_4370.png emd_4370.png | 149.9 KB | ||
| Masks |  emd_4370_msk_1.map emd_4370_msk_1.map | 84.6 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-4370.cif.gz emd-4370.cif.gz | 17.2 KB | ||
| Others |  emd_4370_half_map_1.map.gz emd_4370_half_map_1.map.gz emd_4370_half_map_2.map.gz emd_4370_half_map_2.map.gz | 71.8 MB 71.8 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-4370 http://ftp.pdbj.org/pub/emdb/structures/EMD-4370 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-4370 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-4370 | HTTPS FTP |
-Related structure data
| Related structure data |  6gb2MC  4368C  4369C  6gawC  6gazC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_4370.map.gz / Format: CCP4 / Size: 84.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_4370.map.gz / Format: CCP4 / Size: 84.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 1.39 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Mask #1
| File |  emd_4370_msk_1.map emd_4370_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
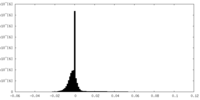
| Density Histograms |
-Half map: #1
| File | emd_4370_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_4370_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
+Entire : mammalian mitochondrial translation initiation complex
+Supramolecule #1: mammalian mitochondrial translation initiation complex
+Supramolecule #2: mammalian mitochondrial ribosome
+Supramolecule #3: Translation initiation factor IF-2, mitochondrial
+Supramolecule #4: tRNA
+Macromolecule #1: Mitochondrial ribosomal protein L12
+Macromolecule #2: Mitochondrial ribosomal protein L27
+Macromolecule #3: Mitochondrial ribosomal protein L28
+Macromolecule #4: Mitochondrial ribosomal protein L47
+Macromolecule #5: 'Mitochondrial ribosomal protein L30
+Macromolecule #6: 'Mitochondrial ribosomal protein L55
+Macromolecule #7: Mitochondrial ribosomal protein L32
+Macromolecule #8: Mitochondrial ribosomal protein L33
+Macromolecule #9: Mitochondrial ribosomal protein L34
+Macromolecule #10: Mitochondrial ribosomal protein L35
+Macromolecule #11: Ribosomal protein
+Macromolecule #14: Translation initiation factor IF-2, mitochondrial
+Macromolecule #15: Mitochondrial ribosomal protein L2
+Macromolecule #16: ICT1
+Macromolecule #17: Mitochondrial ribosomal protein L4
+Macromolecule #18: Mitochondrial ribosomal protein L9
+Macromolecule #19: Mitochondrial ribosomal protein L10
+Macromolecule #20: Mitochondrial ribosomal protein L11
+Macromolecule #21: Mitochondrial ribosomal protein L13
+Macromolecule #22: Mitochondrial ribosomal protein L14
+Macromolecule #23: Mitochondrial ribosomal protein L15
+Macromolecule #24: Mitochondrial ribosomal protein L16
+Macromolecule #25: Mitochondrial ribosomal protein L17
+Macromolecule #26: Mitochondrial ribosomal protein L18
+Macromolecule #27: Mitochondrial ribosomal protein L19
+Macromolecule #28: Mitochondrial ribosomal protein L20
+Macromolecule #29: Mitochondrial ribosomal protein L21
+Macromolecule #30: Mitochondrial ribosomal protein L22
+Macromolecule #31: Mitochondrial ribosomal protein L23
+Macromolecule #32: Mitochondrial ribosomal protein L24
+Macromolecule #33: Mitochondrial ribosomal protein L37
+Macromolecule #34: Mitochondrial ribosomal protein L38
+Macromolecule #35: Mitochondrial ribosomal protein L39
+Macromolecule #36: Mitochondrial ribosomal protein L40
+Macromolecule #37: Mitochondrial ribosomal protein L41
+Macromolecule #38: Mitochondrial ribosomal protein L42
+Macromolecule #39: Mitochondrial ribosomal protein L43
+Macromolecule #40: Mitochondrial ribosomal protein L44
+Macromolecule #41: Mitochondrial ribosomal protein L45
+Macromolecule #42: Mitochondrial ribosomal protein L46
+Macromolecule #43: Mitochondrial ribosomal protein L48
+Macromolecule #44: Mrpl34
+Macromolecule #45: Mitochondrial ribosomal protein L50
+Macromolecule #46: Mitochondrial ribosomal protein L51
+Macromolecule #47: Mitochondrial ribosomal protein L52
+Macromolecule #48: mL53, MRPL53
+Macromolecule #49: Uncharacterized protein
+Macromolecule #50: Mitochondrial ribosomal protein L57
+Macromolecule #51: Mitochondrial ribosomal protein L58
+Macromolecule #52: 'Mitochondrial ribosomal protein L59
+Macromolecule #53: mL65, MRPS30
+Macromolecule #54: Mitochondrial ribosomal protein S18A
+Macromolecule #55: unassigned secondary structure elements
+Macromolecule #12: 16S ribosomal RNA, mitochondrial
+Macromolecule #13: CP tRNAPhe, mitochondrial
+Macromolecule #56: P-site fMet-tRNAMet, mitochondrial
+Macromolecule #57: MAGNESIUM ION
+Macromolecule #58: ZINC ION
+Macromolecule #59: GUANOSINE-5'-MONOPHOSPHATE
+Macromolecule #60: SPERMINE
+Macromolecule #61: 5'-GUANOSINE-DIPHOSPHATE-MONOTHIOPHOSPHATE
+Macromolecule #62: SODIUM ION
+Macromolecule #63: N-FORMYLMETHIONINE
+Macromolecule #64: water
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.171 mg/mL |
|---|---|
| Buffer | pH: 7.6 |
| Grid | Model: Quantifoil R2/2 / Material: COPPER / Support film - Material: CARBON / Support film - topology: CONTINUOUS / Pretreatment - Type: GLOW DISCHARGE |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277.15 K / Instrument: FEI VITROBOT MARK IV |
| Details | contains 55S mitochondrial ribosome, mitochondrial initiation factor 2, mitochondrial formyl-Met-tRNAMet and MT-CO3 mRNA |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON III (4k x 4k) / Detector mode: INTEGRATING / Digitization - Dimensions - Width: 4096 pixel / Digitization - Dimensions - Height: 4096 pixel / Number real images: 13936 / Average exposure time: 1.4 sec. / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: RECIPROCAL / Protocol: OTHER / Overall B value: 56.9 |
|---|---|
| Output model |  PDB-6gb2: |
 Movie
Movie Controller
Controller
































 Z
Z Y
Y X
X