[English] 日本語
 Yorodumi
Yorodumi- EMDB-21427: Single Particle Cryo-EM Structure of the Natively Isolated Sec61,... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-21427 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
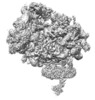
| Title | Single Particle Cryo-EM Structure of the Natively Isolated Sec61, TMCO1, Nicalin, TMEM147, and CCDC47 Containing Ribosome-Translocon Complex | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.4 Å | |||||||||
 Authors Authors | McGilvray PT / Anghel SA / Sundaram A / Trnka MJ / Zhong F / Hu H / Burlingame AL / Keenan RJ | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Elife / Year: 2020 Journal: Elife / Year: 2020Title: An ER translocon for multi-pass membrane protein biogenesis. Authors: Philip T McGilvray / S Andrei Anghel / Arunkumar Sundaram / Frank Zhong / Michael J Trnka / James R Fuller / Hong Hu / Alma L Burlingame / Robert J Keenan /  Abstract: Membrane proteins with multiple transmembrane domains play critical roles in cell physiology, but little is known about the machinery coordinating their biogenesis at the endoplasmic reticulum. Here ...Membrane proteins with multiple transmembrane domains play critical roles in cell physiology, but little is known about the machinery coordinating their biogenesis at the endoplasmic reticulum. Here we describe a ~ 360 kDa ribosome-associated complex comprising the core Sec61 channel and five accessory factors: TMCO1, CCDC47 and the Nicalin-TMEM147-NOMO complex. Cryo-electron microscopy reveals a large assembly at the ribosome exit tunnel organized around a central membrane cavity. Similar to protein-conducting channels that facilitate movement of transmembrane segments, cytosolic and luminal funnels in TMCO1 and TMEM147, respectively, suggest routes into the central membrane cavity. High-throughput mRNA sequencing shows selective translocon engagement with hundreds of different multi-pass membrane proteins. Consistent with a role in multi-pass membrane protein biogenesis, cells lacking different accessory components show reduced levels of one such client, the glutamate transporter EAAT1. These results identify a new human translocon and provide a molecular framework for understanding its role in multi-pass membrane protein biogenesis. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_21427.map.gz emd_21427.map.gz | 413.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-21427-v30.xml emd-21427-v30.xml emd-21427.xml emd-21427.xml | 63.9 KB 63.9 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_21427_fsc.xml emd_21427_fsc.xml | 17.6 KB | Display |  FSC data file FSC data file |
| Images |  emd_21427.png emd_21427.png | 69.8 KB | ||
| Others |  emd_21427_half_map_1.map.gz emd_21427_half_map_1.map.gz emd_21427_half_map_2.map.gz emd_21427_half_map_2.map.gz | 417.4 MB 415.7 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-21427 http://ftp.pdbj.org/pub/emdb/structures/EMD-21427 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-21427 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-21427 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_21427.map.gz / Format: CCP4 / Size: 476.8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_21427.map.gz / Format: CCP4 / Size: 476.8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.36 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Half map: #2
| File | emd_21427_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_21427_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
+Entire : Native complex of a translating ribosome and a novel translocon c...
+Supramolecule #1: Native complex of a translating ribosome and a novel translocon c...
+Supramolecule #2: 60S Subunit
+Supramolecule #3: 40S Subunit
+Supramolecule #4: Translocon
+Supramolecule #5: Sec61 Complex
+Supramolecule #6: TMCO1
+Supramolecule #7: NOMO-Nicalin-TMEM147 Complex
+Supramolecule #8: CCDC47
+Supramolecule #9: 60S acidic ribosomal protein P0
+Supramolecule #10: 60S acidic ribosomal protein P1
+Supramolecule #11: 60S acidic ribosomal protein P2
+Supramolecule #12: 60S ribosomal protein L10
+Supramolecule #13: 60S ribosomal protein L10a
+Supramolecule #14: 60S ribosomal protein L11
+Supramolecule #15: 60S ribosomal protein L12
+Supramolecule #16: 60S ribosomal protein L13
+Supramolecule #17: 60S ribosomal protein L13a
+Supramolecule #18: 60S ribosomal protein L14
+Supramolecule #19: 60S ribosomal protein L15
+Supramolecule #20: 60S ribosomal protein L17
+Supramolecule #21: 60S ribosomal protein L18
+Supramolecule #22: 60S ribosomal protein L18a
+Supramolecule #23: 60S ribosomal protein L19
+Supramolecule #24: 60S ribosomal protein L21
+Supramolecule #25: 60S ribosomal protein L22
+Supramolecule #26: 60S ribosomal protein L23
+Supramolecule #27: 60S ribosomal protein L23a
+Supramolecule #28: 60S ribosomal protein L24
+Supramolecule #29: 60S ribosomal protein L26
+Supramolecule #30: 60S ribosomal protein L27
+Supramolecule #31: 60S ribosomal protein L27a
+Supramolecule #32: 60S ribosomal protein L28
+Supramolecule #33: 60S ribosomal protein L3
+Supramolecule #34: 60S ribosomal protein L30
+Supramolecule #35: 60S ribosomal protein L31
+Supramolecule #36: 60S ribosomal protein L32
+Supramolecule #37: 60S ribosomal protein L34
+Supramolecule #38: 60S ribosomal protein L35
+Supramolecule #39: 60S ribosomal protein L35a
+Supramolecule #40: 60S ribosomal protein L36
+Supramolecule #41: 60S ribosomal protein L36a
+Supramolecule #42: 60S ribosomal protein L37a
+Supramolecule #43: 60S ribosomal protein L38
+Supramolecule #44: 60S ribosomal protein L39
+Supramolecule #45: 60S ribosomal protein L4
+Supramolecule #46: 60S ribosomal protein L5
+Supramolecule #47: 60S ribosomal protein L6
+Supramolecule #48: 60S ribosomal protein L7
+Supramolecule #49: 60S ribosomal protein L7a
+Supramolecule #50: 60S ribosomal protein L8
+Supramolecule #51: 60S ribosomal protein L9
+Supramolecule #52: 40S ribosomal protein S10
+Supramolecule #53: 40S ribosomal protein S11
+Supramolecule #54: 40S ribosomal protein S12
+Supramolecule #55: 40S ribosomal protein S13
+Supramolecule #56: 40S ribosomal protein S14
+Supramolecule #57: 40S ribosomal protein S15
+Supramolecule #58: 40S ribosomal protein S15a
+Supramolecule #59: 40S ribosomal protein S16
+Supramolecule #60: 40S ribosomal protein S17
+Supramolecule #61: 40S ribosomal protein S18
+Supramolecule #62: 40S ribosomal protein S19
+Supramolecule #63: 40S ribosomal protein S2
+Supramolecule #64: 40S ribosomal protein S21
+Supramolecule #65: 40S ribosomal protein S23
+Supramolecule #66: 40S ribosomal protein S24
+Supramolecule #67: 40S ribosomal protein S25
+Supramolecule #68: 40S ribosomal protein S26
+Supramolecule #69: 40S ribosomal protein S27
+Supramolecule #70: 40S ribosomal protein S27-like
+Supramolecule #71: 40S ribosomal protein S28
+Supramolecule #72: 40S ribosomal protein S29
+Supramolecule #73: 40S ribosomal protein S3
+Supramolecule #74: 40S ribosomal protein S30
+Supramolecule #75: 40S ribosomal protein S3a
+Supramolecule #76: 40S ribosomal protein S4, X isoform
+Supramolecule #77: 40S ribosomal protein S5
+Supramolecule #78: 40S ribosomal protein S6
+Supramolecule #79: 40S ribosomal protein S7
+Supramolecule #80: 40S ribosomal protein S8
+Supramolecule #81: 40S ribosomal protein S9
+Supramolecule #82: 40S ribosomal protein SA
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.4 mg/mL | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.4 Component:
| |||||||||||||||
| Grid | Model: C-flat-1.2/1.3 / Material: COPPER / Mesh: 200 / Support film - Material: CARBON / Support film - topology: CONTINUOUS / Support film - Film thickness: 2.0 nm / Pretreatment - Type: GLOW DISCHARGE | |||||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 298 K / Instrument: FEI VITROBOT MARK IV Details: Two filter papers were added to each arm, 2.5 microliters of sample were added to grids, and 0.5 second of drain time was allowed before vitrification.. | |||||||||||||||
| Details | Sample was well-dispersed on a thin (~2nm) carbon film. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Detector mode: COUNTING / Number grids imaged: 1 / Number real images: 5562 / Average exposure time: 3.8 sec. / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller













 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)







































