[English] 日本語
 Yorodumi
Yorodumi- EMDB-14552: The barbed end complex of dynactin bound to BICDR1 and the cytopl... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | The barbed end complex of dynactin bound to BICDR1 and the cytoplasmic dynein tails (A2, B1, B2) | ||||||||||||
 Map data Map data | The barbed end complex of dynactin with BICDR-A and dynein tails (A2, B1, B2). 1.2445 A/pix. | ||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | Dynein / dynactin / cargo transport / activating adaptor / cytoskeleton / STRUCTURAL PROTEIN | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationGolgi to secretory granule transport / : / Factors involved in megakaryocyte development and platelet production / RHOF GTPase cycle / dynactin complex / Regulation of PLK1 Activity at G2/M Transition / Loss of Nlp from mitotic centrosomes / Loss of proteins required for interphase microtubule organization from the centrosome / Anchoring of the basal body to the plasma membrane / AURKA Activation by TPX2 ...Golgi to secretory granule transport / : / Factors involved in megakaryocyte development and platelet production / RHOF GTPase cycle / dynactin complex / Regulation of PLK1 Activity at G2/M Transition / Loss of Nlp from mitotic centrosomes / Loss of proteins required for interphase microtubule organization from the centrosome / Anchoring of the basal body to the plasma membrane / AURKA Activation by TPX2 / Recruitment of mitotic centrosome proteins and complexes / F-actin capping protein complex / WASH complex / dynein light chain binding / transport along microtubule / dynein heavy chain binding / positive regulation of intracellular transport / regulation of metaphase plate congression / positive regulation of spindle assembly / establishment of spindle localization / barbed-end actin filament capping / regulation of cell morphogenesis / vesicle transport along microtubule / retrograde axonal transport / COPI-independent Golgi-to-ER retrograde traffic / P-body assembly / HSP90 chaperone cycle for steroid hormone receptors (SHR) in the presence of ligand / MHC class II antigen presentation / minus-end-directed microtubule motor activity / centrosome localization / Recruitment of NuMA to mitotic centrosomes / dynein light intermediate chain binding / cytoplasmic dynein complex / microtubule motor activity / COPI-mediated anterograde transport / microtubule-based movement / nuclear migration / dynein intermediate chain binding / cortical cytoskeleton / dynactin binding / COPI-mediated anterograde transport / cytoplasmic microtubule / cytoplasmic microtubule organization / Amplification of signal from unattached kinetochores via a MAD2 inhibitory signal / axon cytoplasm / Loss of Nlp from mitotic centrosomes / Loss of proteins required for interphase microtubule organization from the centrosome / cytoskeleton organization / Recruitment of mitotic centrosome proteins and complexes / MHC class II antigen presentation / Recruitment of NuMA to mitotic centrosomes / Anchoring of the basal body to the plasma membrane / Mitotic Prometaphase / EML4 and NUDC in mitotic spindle formation / HSP90 chaperone cycle for steroid hormone receptors (SHR) in the presence of ligand / AURKA Activation by TPX2 / Resolution of Sister Chromatid Cohesion / stress granule assembly / regulation of mitotic spindle organization / mitotic spindle organization / sarcomere / filopodium / RHO GTPases Activate Formins / cellular response to nerve growth factor stimulus / kinetochore / microtubule cytoskeleton organization / small GTPase binding / HCMV Early Events / Aggrephagy / neuron projection development / azurophil granule lumen / actin filament binding / Separation of Sister Chromatids / Regulation of PLK1 Activity at G2/M Transition / late endosome / positive regulation of cold-induced thermogenesis / actin binding / actin cytoskeleton organization / cell cortex / vesicle / microtubule / cell division / Neutrophil degranulation / centrosome / RNA binding / extracellular exosome / extracellular region / ATP binding / membrane / identical protein binding / cytoplasm / cytosol Similarity search - Function | ||||||||||||
| Biological species |  Homo sapiens (human) / Homo sapiens (human) /   | ||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.3 Å | ||||||||||||
 Authors Authors | Chaaban S / Carter AP | ||||||||||||
| Funding support |  United Kingdom, European Union, 3 items United Kingdom, European Union, 3 items
| ||||||||||||
 Citation Citation |  Journal: Nature / Year: 2022 Journal: Nature / Year: 2022Title: Structure of dynein-dynactin on microtubules shows tandem adaptor binding. Authors: Sami Chaaban / Andrew P Carter /  Abstract: Cytoplasmic dynein is a microtubule motor that is activated by its cofactor dynactin and a coiled-coil cargo adaptor. Up to two dynein dimers can be recruited per dynactin, and interactions between ...Cytoplasmic dynein is a microtubule motor that is activated by its cofactor dynactin and a coiled-coil cargo adaptor. Up to two dynein dimers can be recruited per dynactin, and interactions between them affect their combined motile behaviour. Different coiled-coil adaptors are linked to different cargos, and some share motifs known to contact sites on dynein and dynactin. There is limited structural information on how the resulting complex interacts with microtubules and how adaptors are recruited. Here we develop a cryo-electron microscopy processing pipeline to solve the high-resolution structure of dynein-dynactin and the adaptor BICDR1 bound to microtubules. This reveals the asymmetric interactions between neighbouring dynein motor domains and how they relate to motile behaviour. We found that two adaptors occupy the complex. Both adaptors make similar interactions with the dyneins but diverge in their contacts with each other and dynactin. Our structure has implications for the stability and stoichiometry of motor recruitment by cargos. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_14552.map.gz emd_14552.map.gz | 1.4 GB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-14552-v30.xml emd-14552-v30.xml emd-14552.xml emd-14552.xml | 36.4 KB 36.4 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_14552_fsc.xml emd_14552_fsc.xml | 19.7 KB | Display |  FSC data file FSC data file |
| Images |  emd_14552.png emd_14552.png | 105.2 KB | ||
| Masks |  emd_14552_msk_1.map emd_14552_msk_1.map | 1.7 GB |  Mask map Mask map | |
| Filedesc metadata |  emd-14552.cif.gz emd-14552.cif.gz | 11.8 KB | ||
| Others |  emd_14552_additional_1.map.gz emd_14552_additional_1.map.gz emd_14552_half_map_1.map.gz emd_14552_half_map_1.map.gz emd_14552_half_map_2.map.gz emd_14552_half_map_2.map.gz | 1.4 GB 1.4 GB 1.4 GB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-14552 http://ftp.pdbj.org/pub/emdb/structures/EMD-14552 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-14552 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-14552 | HTTPS FTP |
-Related structure data
| Related structure data |  7z8iMC  7z8fC  7z8gC  7z8hC  7z8jC  7z8kC  7z8lC  7z8mC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_14552.map.gz / Format: CCP4 / Size: 1.7 GB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_14552.map.gz / Format: CCP4 / Size: 1.7 GB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | The barbed end complex of dynactin with BICDR-A and dynein tails (A2, B1, B2). 1.2445 A/pix. | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.2445 Å | ||||||||||||||||||||||||||||||||||||
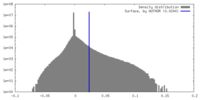
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_14552_msk_1.map emd_14552_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
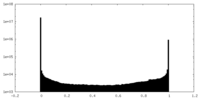
| Density Histograms |
-Additional map: The barbed end complex of dynactin with BICDR-A...
| File | emd_14552_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | The barbed end complex of dynactin with BICDR-A and dynein tails (A2, B1, B2). Unsharpenned map fit to the consensus map rescaled to 1.2445 A/pix. | ||||||||||||
| Projections & Slices |
| ||||||||||||
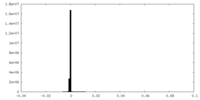
| Density Histograms |
-Half map: Half map 1 of the barbed end complex...
| File | emd_14552_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 1 of the barbed end complex of dynactin with BICDR-A and dynein tails (A2, B1, B2) after fitting to the consensus map. | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half map 2 of the barbed end complex...
| File | emd_14552_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 2 of the barbed end complex of dynactin with BICDR-A and dynein tails (A2, B1, B2) after fitting to the consensus map. | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
+Entire : Complex of dynein, dynactin, and BICDR1 bound to microtubules
+Supramolecule #1: Complex of dynein, dynactin, and BICDR1 bound to microtubules
+Supramolecule #2: Dynein, cytoplasmic 1
+Supramolecule #3: Dynactin
+Supramolecule #4: BICDR1
+Macromolecule #1: ARP1 actin related protein 1 homolog A
+Macromolecule #2: Capping protein (Actin filament) muscle Z-line, alpha 1
+Macromolecule #3: F-actin capping protein beta subunit
+Macromolecule #4: BICD family-like cargo adapter 1
+Macromolecule #5: Cytoplasmic dynein 1 heavy chain 1
+Macromolecule #6: Cytoplasmic dynein 1 intermediate chain 2
+Macromolecule #7: Cytoplasmic dynein 1 light intermediate chain 2
+Macromolecule #8: ADENOSINE-5'-DIPHOSPHATE
+Macromolecule #9: MAGNESIUM ION
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.2 Component:
| |||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 293.15 K / Instrument: FEI VITROBOT MARK IV / Details: 20 second incubation. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Quantum LS / Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Digitization - Dimensions - Width: 5760 pixel / Digitization - Dimensions - Height: 4092 pixel / Number grids imaged: 14 / Number real images: 88715 / Average exposure time: 3.0 sec. / Average electron dose: 53.0 e/Å2 Details: Images were collected in movie-mode and fractionated into 53 movie frames |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 50.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 4.0 µm / Nominal defocus min: 1.2 µm / Nominal magnification: 81000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: FLEXIBLE FIT |
|---|---|
| Output model |  PDB-7z8i: |
 Movie
Movie Controller
Controller

































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)