+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-11165 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Coxsackievirus B4 in complex with capsid binder compound 48 | |||||||||
 Map data Map data | Coxsackievirus B4 in complex with capsid binder compound 48 | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Enterovirus / Coxsackievirus B4 / Inhibitor / Capsid Binder / VIRUS | |||||||||
| Function / homology |  Function and homology information Function and homology informationsymbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of RIG-I activity / picornain 2A / symbiont-mediated suppression of host mRNA export from nucleus / symbiont genome entry into host cell via pore formation in plasma membrane / picornain 3C / T=pseudo3 icosahedral viral capsid / host cell cytoplasmic vesicle membrane / endocytosis involved in viral entry into host cell / nucleoside-triphosphate phosphatase / protein complex oligomerization ...symbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of RIG-I activity / picornain 2A / symbiont-mediated suppression of host mRNA export from nucleus / symbiont genome entry into host cell via pore formation in plasma membrane / picornain 3C / T=pseudo3 icosahedral viral capsid / host cell cytoplasmic vesicle membrane / endocytosis involved in viral entry into host cell / nucleoside-triphosphate phosphatase / protein complex oligomerization / monoatomic ion channel activity / symbiont-mediated suppression of host gene expression / DNA replication / RNA helicase activity / induction by virus of host autophagy / RNA-directed RNA polymerase / viral RNA genome replication / cysteine-type endopeptidase activity / RNA-dependent RNA polymerase activity / virus-mediated perturbation of host defense response / DNA-templated transcription / host cell nucleus / virion attachment to host cell / structural molecule activity / ATP hydrolysis activity / proteolysis / RNA binding / ATP binding / membrane / metal ion binding Similarity search - Function | |||||||||
| Biological species |  Coxsackievirus B4 (strain E2) Coxsackievirus B4 (strain E2) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.7 Å | |||||||||
 Authors Authors | Flatt JW / Domanska A | |||||||||
| Funding support |  Finland, 2 items Finland, 2 items
| |||||||||
 Citation Citation |  Journal: Commun Biol / Year: 2021 Journal: Commun Biol / Year: 2021Title: Identification of a conserved virion-stabilizing network inside the interprotomer pocket of enteroviruses. Authors: Justin W Flatt / Aušra Domanska / Alma L Seppälä / Sarah J Butcher /  Abstract: Enteroviruses pose a persistent and widespread threat to human physical health, with no specific treatments available. Small molecule capsid binders have the potential to be developed as antivirals ...Enteroviruses pose a persistent and widespread threat to human physical health, with no specific treatments available. Small molecule capsid binders have the potential to be developed as antivirals that prevent virus attachment and entry into host cells. To aid with broad-range drug development, we report here structures of coxsackieviruses B3 and B4 bound to different interprotomer-targeting capsid binders using single-particle cryo-EM. The EM density maps are beyond 3 Å resolution, providing detailed information about interactions in the ligand-binding pocket. Comparative analysis revealed the residues that form a conserved virion-stabilizing network at the interprotomer site, and showed the small molecule properties that allow anchoring in the pocket to inhibit virus disassembly. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_11165.map.gz emd_11165.map.gz | 59 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-11165-v30.xml emd-11165-v30.xml emd-11165.xml emd-11165.xml | 15.1 KB 15.1 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_11165.png emd_11165.png | 243 KB | ||
| Filedesc metadata |  emd-11165.cif.gz emd-11165.cif.gz | 6.1 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-11165 http://ftp.pdbj.org/pub/emdb/structures/EMD-11165 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-11165 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-11165 | HTTPS FTP |
-Validation report
| Summary document |  emd_11165_validation.pdf.gz emd_11165_validation.pdf.gz | 484.5 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_11165_full_validation.pdf.gz emd_11165_full_validation.pdf.gz | 484.1 KB | Display | |
| Data in XML |  emd_11165_validation.xml.gz emd_11165_validation.xml.gz | 7.3 KB | Display | |
| Data in CIF |  emd_11165_validation.cif.gz emd_11165_validation.cif.gz | 8.4 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-11165 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-11165 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-11165 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-11165 | HTTPS FTP |
-Related structure data
| Related structure data |  6zckMC  6zclC  6zmsC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | |
| EM raw data |  EMPIAR-10652 (Title: Identification of a conserved virion-stabilizing network inside the interprotomer pocket of enteroviruses EMPIAR-10652 (Title: Identification of a conserved virion-stabilizing network inside the interprotomer pocket of enterovirusesData size: 7.3 TB Data #1: Unaligned multiframe micrographs of CVB4 in complex with CP48 [micrographs - multiframe] Data #2: Aligned multi-frame micrographs dose weighted [micrographs - single frame] Data #3: Aligned non dose weighted micrographs [micrographs - single frame] Data #4: Un-aligned multiframe micrographs of CVB4 control [micrographs - multiframe]) |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_11165.map.gz / Format: CCP4 / Size: 282.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_11165.map.gz / Format: CCP4 / Size: 282.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Coxsackievirus B4 in complex with capsid binder compound 48 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.06 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Coxsackievirus B4 (strain E2)
| Entire | Name:  Coxsackievirus B4 (strain E2) Coxsackievirus B4 (strain E2) |
|---|---|
| Components |
|
-Supramolecule #1: Coxsackievirus B4 (strain E2)
| Supramolecule | Name: Coxsackievirus B4 (strain E2) / type: virus / ID: 1 / Parent: 0 / Macromolecule list: #1-#4 Details: Virus harvested in BGM cells and purified in CsCl gradient. NCBI-ID: 103905 / Sci species name: Coxsackievirus B4 (strain E2) / Virus type: VIRION / Virus isolate: STRAIN / Virus enveloped: No / Virus empty: No |
|---|---|
| Host (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Virus shell | Shell ID: 1 / Name: Icosahedron / Diameter: 300.0 Å / T number (triangulation number): 3 |
-Macromolecule #1: Capsid protein VP1
| Macromolecule | Name: Capsid protein VP1 / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Coxsackievirus B4 (strain E2) / Strain: E2 Coxsackievirus B4 (strain E2) / Strain: E2 |
| Molecular weight | Theoretical: 30.685498 KDa |
| Sequence | String: MGRVADTIAR GPSNSEQIPA LTAVETGHTS QVDPSDTMQT RHVHNYHSRS ESSIENFLCR SACVIYIKYS SAESNNLKRY AEWVINTRQ VAQLRRKMEM FTYIRCDMEL TFVITSHQEM STATNSDVPV QTHQIMYVPP GGPVPTSVND YVWQTSTNPS I FWTEGNAP ...String: MGRVADTIAR GPSNSEQIPA LTAVETGHTS QVDPSDTMQT RHVHNYHSRS ESSIENFLCR SACVIYIKYS SAESNNLKRY AEWVINTRQ VAQLRRKMEM FTYIRCDMEL TFVITSHQEM STATNSDVPV QTHQIMYVPP GGPVPTSVND YVWQTSTNPS I FWTEGNAP PRMSIPFMSI GNAYTMFYDG WSNFSRDGIY GYNSLNNMGT IYARHVNDSS PGGLTSTIRI YFKPKHVKAY VP RPPRLCQ YKKAKNVNFD VEAVTTERAS LVTT UniProtKB: Genome polyprotein |
-Macromolecule #2: Capsid protein VP2
| Macromolecule | Name: Capsid protein VP2 / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Coxsackievirus B4 (strain E2) / Strain: E2 Coxsackievirus B4 (strain E2) / Strain: E2 |
| Molecular weight | Theoretical: 27.70807 KDa |
| Sequence | String: SDRVRSITLG NSTITTQECA NVVVGYGVWP DYLSDEEATA EDQPTQPDVA TCRFYTLNSV KWEMQSAGWW WKFPDALSEM GLFGQNMQY HYLGRSGYTI HVQCNASKFH QGCLLVVCVP EAEMGCTNAE NAPTYGDLCG GETAKQFEQN AVTGETAVQT A VCNAGMGV ...String: SDRVRSITLG NSTITTQECA NVVVGYGVWP DYLSDEEATA EDQPTQPDVA TCRFYTLNSV KWEMQSAGWW WKFPDALSEM GLFGQNMQY HYLGRSGYTI HVQCNASKFH QGCLLVVCVP EAEMGCTNAE NAPTYGDLCG GETAKQFEQN AVTGETAVQT A VCNAGMGV GVGNLTIYPH QWINLRTNNS ATIVMPYINS VPMDNMFRHN NFTLMIIPFA PLDYVTGASS YIPITVTVAP MS AEYNGLR LAGHQ UniProtKB: Genome polyprotein |
-Macromolecule #3: Capsid protein VP3
| Macromolecule | Name: Capsid protein VP3 / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Coxsackievirus B4 (strain E2) / Strain: E2 Coxsackievirus B4 (strain E2) / Strain: E2 |
| Molecular weight | Theoretical: 26.44416 KDa |
| Sequence | String: GLPTMLTPGS TQFLTSDDFQ SPSAMPQFDV TPEMNIPGQV RNLMEIAEVD SVVPINNLQA NLKTMEAYRV QVRSTDEMGG QIFGFPLQP GASSVLQRTL LGEILNYYTH WSGSLKLTFV FCGSAMATGK FLLAYSPPGA GAPDSRKNAM LGTHVIWDVG L QSSCVLCV ...String: GLPTMLTPGS TQFLTSDDFQ SPSAMPQFDV TPEMNIPGQV RNLMEIAEVD SVVPINNLQA NLKTMEAYRV QVRSTDEMGG QIFGFPLQP GASSVLQRTL LGEILNYYTH WSGSLKLTFV FCGSAMATGK FLLAYSPPGA GAPDSRKNAM LGTHVIWDVG L QSSCVLCV PWISQTHYRY VVDDKYTASG FISCWYQTNV IVPAEAQKSC YIMCFVSACN DFSVRMLRDT QFIKQDTFYQ UniProtKB: Genome polyprotein |
-Macromolecule #4: Capsid protein VP4
| Macromolecule | Name: Capsid protein VP4 / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Coxsackievirus B4 (strain E2) / Strain: E2 Coxsackievirus B4 (strain E2) / Strain: E2 |
| Molecular weight | Theoretical: 7.499235 KDa |
| Sequence | String: MGAQVSTQKT GAHETSLSAS GNSIIHYTNI NYYKDAASNS ANRQDFTQDP SKFTEPVKDV MIKSLPALN UniProtKB: Genome polyprotein |
-Macromolecule #5: 4-[(6-propoxynaphthalen-2-yl)sulfonylamino]benzoic acid
| Macromolecule | Name: 4-[(6-propoxynaphthalen-2-yl)sulfonylamino]benzoic acid type: ligand / ID: 5 / Number of copies: 1 / Formula: QFW |
|---|---|
| Molecular weight | Theoretical: 385.434 Da |
| Chemical component information | 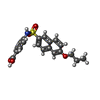 ChemComp-QFW: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7 Component:
| |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Grid | Model: PELCO Ultrathin Carbon with Lacey Carbon / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 45 sec. / Details: Ted Pella product No. 01824 | |||||||||||||||
| Vitrification | Cryogen name: ETHANE / Instrument: HOMEMADE PLUNGER | |||||||||||||||
| Details | purified virus mixed with compound 48, incubated at room temperature for 30 minutes before plunging. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Average electron dose: 47.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 3.0 µm / Nominal defocus min: 0.6 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: INSILICO MODEL |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 2.7 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: RELION (ver. 3) / Number images used: 13252 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller
















