[English] 日本語
 Yorodumi
Yorodumi- PDB-5w7v: CryoEM structure of the segment, DLIIKGISVHI, assembled into a tr... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5w7v | ||||||
|---|---|---|---|---|---|---|---|
| Title | CryoEM structure of the segment, DLIIKGISVHI, assembled into a triple-helical fibril | ||||||
 Components Components | TAR DNA-binding protein 43 | ||||||
 Keywords Keywords | PROTEIN FIBRIL / Amyloid / steric zipper | ||||||
| Function / homology |  Function and homology information Function and homology informationnuclear inner membrane organization / interchromatin granule / perichromatin fibrils / negative regulation by host of viral transcription / 3'-UTR-mediated mRNA destabilization / 3'-UTR-mediated mRNA stabilization / intracellular non-membrane-bounded organelle / pre-mRNA intronic binding / response to endoplasmic reticulum stress / RNA splicing ...nuclear inner membrane organization / interchromatin granule / perichromatin fibrils / negative regulation by host of viral transcription / 3'-UTR-mediated mRNA destabilization / 3'-UTR-mediated mRNA stabilization / intracellular non-membrane-bounded organelle / pre-mRNA intronic binding / response to endoplasmic reticulum stress / RNA splicing / molecular condensate scaffold activity / negative regulation of protein phosphorylation / mRNA 3'-UTR binding / regulation of circadian rhythm / regulation of protein stability / positive regulation of insulin secretion / cytoplasmic stress granule / positive regulation of protein import into nucleus / mRNA processing / rhythmic process / double-stranded DNA binding / regulation of gene expression / regulation of apoptotic process / amyloid fibril formation / regulation of cell cycle / nuclear speck / RNA polymerase II cis-regulatory region sequence-specific DNA binding / negative regulation of gene expression / lipid binding / chromatin / mitochondrion / DNA binding / RNA binding / nucleoplasm / identical protein binding / nucleus Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method | ELECTRON MICROSCOPY / helical reconstruction / cryo EM / Resolution: 3.8 Å | ||||||
 Authors Authors | Guenther, E.L. / Ge, P. / Eisenberg, D.S. | ||||||
| Funding support |  United States, 1items United States, 1items
| ||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2018 Journal: Nat Struct Mol Biol / Year: 2018Title: Atomic-level evidence for packing and positional amyloid polymorphism by segment from TDP-43 RRM2. Authors: Elizabeth L Guenther / Peng Ge / Hamilton Trinh / Michael R Sawaya / Duilio Cascio / David R Boyer / Tamir Gonen / Z Hong Zhou / David S Eisenberg /  Abstract: Proteins in the fibrous amyloid state are a major hallmark of neurodegenerative disease. Understanding the multiple conformations, or polymorphs, of amyloid proteins at the molecular level is a ...Proteins in the fibrous amyloid state are a major hallmark of neurodegenerative disease. Understanding the multiple conformations, or polymorphs, of amyloid proteins at the molecular level is a challenge of amyloid research. Here, we detail the wide range of polymorphs formed by a segment of human TAR DNA-binding protein 43 (TDP-43) as a model for the polymorphic capabilities of pathological amyloid aggregation. Using X-ray diffraction, microelectron diffraction (MicroED) and single-particle cryo-EM, we show that the DLIIKGISVHI segment from the second RNA-recognition motif (RRM2) forms an array of amyloid polymorphs. These associations include seven distinct interfaces displaying five different symmetry classes of steric zippers. Additionally, we find that this segment can adopt three different backbone conformations that contribute to its polymorphic capabilities. The polymorphic nature of this segment illustrates at the molecular level how amyloid proteins can form diverse fibril structures. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5w7v.cif.gz 5w7v.cif.gz | 24.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5w7v.ent.gz pdb5w7v.ent.gz | 16.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5w7v.json.gz 5w7v.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  5w7v_validation.pdf.gz 5w7v_validation.pdf.gz | 987.8 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  5w7v_full_validation.pdf.gz 5w7v_full_validation.pdf.gz | 987.5 KB | Display | |
| Data in XML |  5w7v_validation.xml.gz 5w7v_validation.xml.gz | 9.6 KB | Display | |
| Data in CIF |  5w7v_validation.cif.gz 5w7v_validation.cif.gz | 12.6 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/w7/5w7v https://data.pdbj.org/pub/pdb/validation_reports/w7/5w7v ftp://data.pdbj.org/pub/pdb/validation_reports/w7/5w7v ftp://data.pdbj.org/pub/pdb/validation_reports/w7/5w7v | HTTPS FTP |
-Related structure data
| Related structure data |  8781MC  8765C 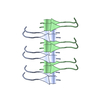 5w50C  5w52C M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 | x 30
|
| 2 |
|
| 3 | 
|
| Symmetry | Helical symmetry: (Circular symmetry: 1 / Dyad axis: no / N subunits divisor: 1 / Num. of operations: 30 / Rise per n subunits: 1.598 Å / Rotation per n subunits: -120.441 °) |
- Components
Components
| #1: Protein/peptide | Mass: 1209.479 Da / Num. of mol.: 9 / Fragment: RRM2 peptide (UNP residues 247-257) / Source method: obtained synthetically / Source: (synth.)  Homo sapiens (human) / References: UniProt: Q13148 Homo sapiens (human) / References: UniProt: Q13148 |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: HELICAL ARRAY / 3D reconstruction method: helical reconstruction |
- Sample preparation
Sample preparation
| Component | Name: DLIIKGISVHI fibril / Type: COMPLEX / Entity ID: all / Source: NATURAL |
|---|---|
| Molecular weight | Experimental value: NO |
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Buffer solution | pH: 7 |
| Buffer component | Name: Water / Formula: H2O |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Specimen support | Grid material: COPPER / Grid mesh size: 200 divisions/in. / Grid type: Quantifoil R1.2/1.3 |
| Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE / Humidity: 100 % / Chamber temperature: 295 K |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Cs: 2.7 mm / C2 aperture diameter: 70 µm / Alignment procedure: COMA FREE |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Average exposure time: 0.2 sec. / Electron dose: 1.2 e/Å2 / Detector mode: COUNTING / Film or detector model: GATAN K2 QUANTUM (4k x 4k) / Num. of grids imaged: 1 / Num. of real images: 610 |
| EM imaging optics | Energyfilter name: GIF Quantum |
| Image scans | Movie frames/image: 50 / Used frames/image: 4-20 |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.10.1_2155: / Classification: refinement | |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software |
| |||||||||||||||||||||||||
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | |||||||||||||||||||||||||
| Helical symmerty | Angular rotation/subunit: -120.441 ° / Axial rise/subunit: 1.598 Å / Axial symmetry: C1 | |||||||||||||||||||||||||
| 3D reconstruction | Resolution: 3.8 Å / Resolution method: FSC 0.5 CUT-OFF / Num. of particles: 18818 / Algorithm: FOURIER SPACE / Symmetry type: HELICAL | |||||||||||||||||||||||||
| Atomic model building | Protocol: AB INITIO MODEL / Space: REAL | |||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller








 PDBj
PDBj
