[English] 日本語
 Yorodumi
Yorodumi- PDB-6o48: Wild-type HIV-1 protease in complex with a substrate analog CA-p2 -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6o48 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Wild-type HIV-1 protease in complex with a substrate analog CA-p2 | ||||||
 Components Components | HIV-1 protease | ||||||
 Keywords Keywords | HYDROLASE / VIRAL PROTEIN / HIV PROTEASE | ||||||
| Function / homology |  Function and homology information Function and homology informationHIV-1 retropepsin / symbiont-mediated activation of host apoptosis / retroviral ribonuclease H / exoribonuclease H / exoribonuclease H activity / host multivesicular body / DNA integration / viral genome integration into host DNA / RNA-directed DNA polymerase / establishment of integrated proviral latency ...HIV-1 retropepsin / symbiont-mediated activation of host apoptosis / retroviral ribonuclease H / exoribonuclease H / exoribonuclease H activity / host multivesicular body / DNA integration / viral genome integration into host DNA / RNA-directed DNA polymerase / establishment of integrated proviral latency / viral penetration into host nucleus / RNA stem-loop binding / symbiont-mediated suppression of host gene expression / RNA-directed DNA polymerase activity / host cell / RNA-DNA hybrid ribonuclease activity / Transferases; Transferring phosphorus-containing groups; Nucleotidyltransferases / viral nucleocapsid / aspartic-type endopeptidase activity / DNA recombination / DNA-directed DNA polymerase / Hydrolases; Acting on ester bonds / DNA-directed DNA polymerase activity / symbiont entry into host cell / lipid binding / host cell nucleus / host cell plasma membrane / structural molecule activity / virion membrane / proteolysis / DNA binding / zinc ion binding / membrane Similarity search - Function | ||||||
| Biological species |   Human immunodeficiency virus 1 Human immunodeficiency virus 1 | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.46 Å MOLECULAR REPLACEMENT / Resolution: 1.46 Å | ||||||
 Authors Authors | Wang, Y.-F. / Liu, F. / Weber, I.T. | ||||||
| Funding support |  United States, 1items United States, 1items
| ||||||
 Citation Citation |  Journal: Acs Omega / Year: 2019 Journal: Acs Omega / Year: 2019Title: Highly Drug-Resistant HIV-1 Protease Mutant PRS17 Shows Enhanced Binding to Substrate Analogues. Authors: Agniswamy, J. / Kneller, D.W. / Brothers, R. / Wang, Y.F. / Harrison, R.W. / Weber, I.T. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6o48.cif.gz 6o48.cif.gz | 103.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6o48.ent.gz pdb6o48.ent.gz | 76.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6o48.json.gz 6o48.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  6o48_validation.pdf.gz 6o48_validation.pdf.gz | 698.4 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  6o48_full_validation.pdf.gz 6o48_full_validation.pdf.gz | 701.5 KB | Display | |
| Data in XML |  6o48_validation.xml.gz 6o48_validation.xml.gz | 12.1 KB | Display | |
| Data in CIF |  6o48_validation.cif.gz 6o48_validation.cif.gz | 16.1 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/o4/6o48 https://data.pdbj.org/pub/pdb/validation_reports/o4/6o48 ftp://data.pdbj.org/pub/pdb/validation_reports/o4/6o48 ftp://data.pdbj.org/pub/pdb/validation_reports/o4/6o48 | HTTPS FTP |
-Related structure data
| Related structure data |  6o54C  6o57C  6o5aC  6o5xC  2aodS C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 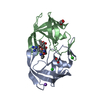
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 2 molecules AB
| #1: Protein | Mass: 10740.677 Da / Num. of mol.: 2 / Mutation: Q7K, L33I, L63I, C67A, C95A Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Human immunodeficiency virus 1 / Gene: pol / Production host: Human immunodeficiency virus 1 / Gene: pol / Production host:  |
|---|
-Non-polymers , 6 types, 114 molecules 









| #2: Chemical | ChemComp-NA / | ||||||||
|---|---|---|---|---|---|---|---|---|---|
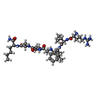
| #3: Chemical | | #4: Chemical | ChemComp-FMT / | #5: Chemical | ChemComp-0Q4 / |   Type: peptide-like, Peptide-like / Class: Inhibitor / Mass: 833.053 Da / Num. of mol.: 1 / Source method: obtained synthetically / Formula: C40H70N11O8 Type: peptide-like, Peptide-like / Class: Inhibitor / Mass: 833.053 Da / Num. of mol.: 1 / Source method: obtained synthetically / Formula: C40H70N11O8References: N-[(2R)-2-({N~5~-[amino(iminio)methyl]-L-ornithyl-L-valyl}amino)-4-methylpentyl]-L-phenylalanyl-L-alpha-glutamyl- L-alanyl-L-norleucinamide #6: Chemical | ChemComp-GOL / | #7: Water | ChemComp-HOH / | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.73 Å3/Da / Density % sol: 54.96 % |
|---|---|
| Crystal grow | Temperature: 298 K / Method: vapor diffusion, hanging drop / pH: 4.8 / Details: 1M sodium chloride and 0.1M sodium acetate |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  APS APS  / Beamline: 22-ID / Wavelength: 1 Å / Beamline: 22-ID / Wavelength: 1 Å |
| Detector | Type: MAR CCD 165 mm / Detector: CCD / Date: Oct 7, 2003 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1 Å / Relative weight: 1 |
| Reflection | Resolution: 1.46→50 Å / Num. obs: 38064 / % possible obs: 91.9 % / Redundancy: 4.1 % / Biso Wilson estimate: 16 Å2 / Rmerge(I) obs: 0.09 / Net I/σ(I): 11 |
| Reflection shell | Resolution: 1.46→1.51 Å / Redundancy: 1.7 % / Rmerge(I) obs: 0.438 / Mean I/σ(I) obs: 2 / Num. unique obs: 2696 / % possible all: 66.5 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 2AOD Resolution: 1.46→50 Å / Cross valid method: FREE R-VALUE / σ(F): 0 Details: ANISOTROPIC REFINEMENT REDUCED FREE R (NO CUTOFF) BY ?
| |||||||||||||||||||||||||||||||||
| Refine analyze | Num. disordered residues: 19 / Occupancy sum hydrogen: 0 / Occupancy sum non hydrogen: 1680.25 | |||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.46→50 Å
| |||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller





















 PDBj
PDBj






