Entry Database : PDB / ID : 5mkxTitle 1.68A STRUCTURE PCAF BROMODOMAIN WITH 4-chloro-2-methyl-5-(methylamino)pyridazin-3(2H)-one Histone acetyltransferase KAT2B Keywords / / / / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Homo sapiens (human)Method / / Resolution : 1.68 Å Authors Chung, C.-W. Journal : J. Med. Chem. / Year : 2017Title : Discovery of a Potent, Cell Penetrant, and Selective p300/CBP-Associated Factor (PCAF)/General Control Nonderepressible 5 (GCN5) Bromodomain Chemical Probe.Authors: Humphreys, P.G. / Bamborough, P. / Chung, C.W. / Craggs, P.D. / Gordon, L. / Grandi, P. / Hayhow, T.G. / Hussain, J. / Jones, K.L. / Lindon, M. / Michon, A.M. / Renaux, J.F. / Suckling, C.J. ... Authors : Humphreys, P.G. / Bamborough, P. / Chung, C.W. / Craggs, P.D. / Gordon, L. / Grandi, P. / Hayhow, T.G. / Hussain, J. / Jones, K.L. / Lindon, M. / Michon, A.M. / Renaux, J.F. / Suckling, C.J. / Tough, D.F. / Prinjha, R.K. History Deposition Dec 5, 2016 Deposition site / Processing site Revision 1.0 Dec 20, 2017 Provider / Type Revision 1.1 Jan 24, 2018 Group / Category / citation_authorItem _citation.country / _citation.journal_abbrev ... _citation.country / _citation.journal_abbrev / _citation.journal_id_ASTM / _citation.journal_id_CSD / _citation.journal_id_ISSN / _citation.journal_volume / _citation.page_first / _citation.page_last / _citation.pdbx_database_id_DOI / _citation.pdbx_database_id_PubMed / _citation.title / _citation.year Revision 1.2 Jul 13, 2022 Group / Structure summary / Category / database_2Item / _database_2.pdbx_DOI / _database_2.pdbx_database_accessionRevision 1.3 Jun 19, 2024 Group / Category / chem_comp_bond
Show all Show less
 Yorodumi
Yorodumi Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information Homo sapiens (human)
Homo sapiens (human) X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  FOURIER SYNTHESIS / Resolution: 1.68 Å
FOURIER SYNTHESIS / Resolution: 1.68 Å  Authors
Authors Citation
Citation Journal: J. Med. Chem. / Year: 2017
Journal: J. Med. Chem. / Year: 2017 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 5mkx.cif.gz
5mkx.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb5mkx.ent.gz
pdb5mkx.ent.gz PDB format
PDB format 5mkx.json.gz
5mkx.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/mk/5mkx
https://data.pdbj.org/pub/pdb/validation_reports/mk/5mkx ftp://data.pdbj.org/pub/pdb/validation_reports/mk/5mkx
ftp://data.pdbj.org/pub/pdb/validation_reports/mk/5mkx Links
Links Assembly
Assembly


 Components
Components Homo sapiens (human) / Gene: KAT2B, PCAF / Production host:
Homo sapiens (human) / Gene: KAT2B, PCAF / Production host: 
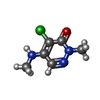







 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation ROTATING ANODE / Type: RIGAKU FR-E+ SUPERBRIGHT / Wavelength: 1.54178 Å
ROTATING ANODE / Type: RIGAKU FR-E+ SUPERBRIGHT / Wavelength: 1.54178 Å Processing
Processing FOURIER SYNTHESIS / Resolution: 1.68→32.65 Å / Cor.coef. Fo:Fc: 0.963 / Cor.coef. Fo:Fc free: 0.953 / SU B: 3.289 / SU ML: 0.063 / Cross valid method: THROUGHOUT / ESU R: 0.089 / ESU R Free: 0.088 / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
FOURIER SYNTHESIS / Resolution: 1.68→32.65 Å / Cor.coef. Fo:Fc: 0.963 / Cor.coef. Fo:Fc free: 0.953 / SU B: 3.289 / SU ML: 0.063 / Cross valid method: THROUGHOUT / ESU R: 0.089 / ESU R Free: 0.088 / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS Movie
Movie Controller
Controller















 PDBj
PDBj












