[English] 日本語
 Yorodumi
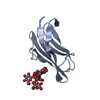
Yorodumi- PDB-4agj: Crystal structure of the capsid protein (110-267) from Aura virus... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4agj | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of the capsid protein (110-267) from Aura virus in complex with dioxane | ||||||
 Components Components | CAPSID PROTEIN Capsid Capsid | ||||||
 Keywords Keywords |  HYDROLASE / HYDROLASE /  VIRAL PROTEIN / VIRAL PROTEIN /  DIOXANE DIOXANE | ||||||
| Function / homology |  Function and homology information Function and homology information togavirin / T=4 icosahedral viral capsid / symbiont-mediated suppression of host toll-like receptor signaling pathway / host cell cytoplasm / symbiont entry into host cell / serine-type endopeptidase activity / fusion of virus membrane with host endosome membrane / host cell nucleus / virion attachment to host cell / host cell plasma membrane ... togavirin / T=4 icosahedral viral capsid / symbiont-mediated suppression of host toll-like receptor signaling pathway / host cell cytoplasm / symbiont entry into host cell / serine-type endopeptidase activity / fusion of virus membrane with host endosome membrane / host cell nucleus / virion attachment to host cell / host cell plasma membrane ... togavirin / T=4 icosahedral viral capsid / symbiont-mediated suppression of host toll-like receptor signaling pathway / host cell cytoplasm / symbiont entry into host cell / serine-type endopeptidase activity / fusion of virus membrane with host endosome membrane / host cell nucleus / virion attachment to host cell / host cell plasma membrane / virion membrane / structural molecule activity / togavirin / T=4 icosahedral viral capsid / symbiont-mediated suppression of host toll-like receptor signaling pathway / host cell cytoplasm / symbiont entry into host cell / serine-type endopeptidase activity / fusion of virus membrane with host endosome membrane / host cell nucleus / virion attachment to host cell / host cell plasma membrane / virion membrane / structural molecule activity /  proteolysis / proteolysis /  RNA binding / RNA binding /  membrane membraneSimilarity search - Function | ||||||
| Biological species |   AURA VIRUS AURA VIRUS | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 1.98 Å MOLECULAR REPLACEMENT / Resolution: 1.98 Å | ||||||
 Authors Authors | Aggarwal, M. / Kumar, P. / Tomar, S. | ||||||
 Citation Citation |  Journal: Plos One / Year: 2012 Journal: Plos One / Year: 2012Title: Crystal Structure of Aura Virus Capsid Protease and its Complex with Dioxane: New Insights Into Capsid-Glycoprotein Molecular Contacts. Authors: Aggarwal, M. / Tapas, S. / Preeti / Siwach, A. / Kumar, P. / Kuhn, R.J. / Tomar, S. #1: Journal: Acta Crystallogr.,Sect.F / Year: 2011 Title: Crystallization, High-Resolution Data Collection and Preliminary Crystallographic Analysis of Aura Virus Capsid Protease and its Complex with Dioxane. Authors: Aggarwal, M. / Dhindwal, S. / Pratap, S. / Kuhn, R.J. / Kumar, P. / Tomar, S. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4agj.cif.gz 4agj.cif.gz | 69.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4agj.ent.gz pdb4agj.ent.gz | 50.9 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4agj.json.gz 4agj.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ag/4agj https://data.pdbj.org/pub/pdb/validation_reports/ag/4agj ftp://data.pdbj.org/pub/pdb/validation_reports/ag/4agj ftp://data.pdbj.org/pub/pdb/validation_reports/ag/4agj | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  4agkC  1kxaS C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein |  Capsid / COAT PROTEIN / C Capsid / COAT PROTEIN / CMass: 17009.230 Da / Num. of mol.: 1 / Fragment: RESIDUES 110-267 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   AURA VIRUS / Production host: AURA VIRUS / Production host:   ESCHERICHIA COLI (E. coli) / Strain (production host): BL21(DE3) / Variant (production host): ROSETTA / References: UniProt: Q86925, ESCHERICHIA COLI (E. coli) / Strain (production host): BL21(DE3) / Variant (production host): ROSETTA / References: UniProt: Q86925,  togavirin togavirin |
|---|---|
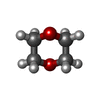
| #2: Chemical | ChemComp-DIO /  1,4-Dioxane 1,4-Dioxane |
| #3: Water | ChemComp-HOH /  Water Water |
| Sequence details | SOME RESIDUES ARE FOUND TO BE DIFFERENT IN OUR DATA |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 1.99 Å3/Da / Density % sol: 38.31 % / Description: NONE |
|---|---|
Crystal grow | Details: BIS-TRIS AND POLYETHYLENE GLYCOL MONOMETHYL ETHER 2000 |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: BRUKER AXS MICROSTAR-H / Wavelength: 1.5418 ROTATING ANODE / Type: BRUKER AXS MICROSTAR-H / Wavelength: 1.5418 |
| Detector | Type: MAR scanner 345 mm plate / Detector: IMAGE PLATE / Date: Dec 3, 2010 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 1.5418 Å / Relative weight: 1 : 1.5418 Å / Relative weight: 1 |
| Reflection | Resolution: 1.98→50 Å / Num. obs: 7989 / % possible obs: 83.9 % / Observed criterion σ(I): 2 / Redundancy: 3.1 % / Rmerge(I) obs: 0.05 / Net I/σ(I): 20.34 |
| Reflection shell | Resolution: 1.98→2.01 Å / Redundancy: 1.9 % / Rmerge(I) obs: 0.29 / Mean I/σ(I) obs: 2 / % possible all: 33.6 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 1KXA Resolution: 1.98→48.17 Å / Cor.coef. Fo:Fc: 0.964 / Cor.coef. Fo:Fc free: 0.922 / SU B: 9.617 / SU ML: 0.148 / Cross valid method: THROUGHOUT / ESU R: 0.268 / ESU R Free: 0.232 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 36.728 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.98→48.17 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller











 PDBj
PDBj