+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: PDB / ID: 487d | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
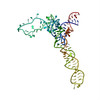
| タイトル | SEVEN RIBOSOMAL PROTEINS FITTED TO A CRYO-ELECTRON MICROSCOPIC MAP OF THE LARGE 50S SUBUNIT AT 7.5 ANGSTROMS RESOLUTION | |||||||||
 要素 要素 | (50S ribosomal protein ...) x 7 | |||||||||
 キーワード キーワード | RIBOSOME / LARGE RIBOSOMAL SUBUNIT / RIBOSOMAL PROTEIN / PROTEIN BIOSYNTHESIS / EM-RECONSTRUCTION / ATOMIC STRUCTURE / 3D ARRANGEMENT / FITTING | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報ribosomal large subunit assembly / response to radiation / regulation of translation / transferase activity / 5S rRNA binding / large ribosomal subunit rRNA binding / cytosolic large ribosomal subunit / cytoplasmic translation / tRNA binding / rRNA binding ...ribosomal large subunit assembly / response to radiation / regulation of translation / transferase activity / 5S rRNA binding / large ribosomal subunit rRNA binding / cytosolic large ribosomal subunit / cytoplasmic translation / tRNA binding / rRNA binding / negative regulation of translation / ribosome / structural constituent of ribosome / translation / ribonucleoprotein complex / cytoplasm / cytosol 類似検索 - 分子機能 | |||||||||
| 生物種 |   Thermus thermophilus (バクテリア) Thermus thermophilus (バクテリア)  Geobacillus stearothermophilus (バクテリア) Geobacillus stearothermophilus (バクテリア)  Thermotoga maritima (バクテリア) Thermotoga maritima (バクテリア) | |||||||||
| 手法 | 電子顕微鏡法 / 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 7.5 Å | |||||||||
 データ登録者 データ登録者 | Brimacombe, R. / Mueller, F. | |||||||||
 引用 引用 |  ジャーナル: J Mol Biol / 年: 2000 ジャーナル: J Mol Biol / 年: 2000タイトル: The 3D arrangement of the 23 S and 5 S rRNA in the Escherichia coli 50 S ribosomal subunit based on a cryo-electron microscopic reconstruction at 7.5 A resolution. 著者: F Mueller / I Sommer / P Baranov / R Matadeen / M Stoldt / J Wöhnert / M Görlach / M van Heel / R Brimacombe /  要旨: The Escherichia coli 23 S and 5 S rRNA molecules have been fitted helix by helix to a cryo-electron microscopic (EM) reconstruction of the 50 S ribosomal subunit, using an unfiltered version of the ...The Escherichia coli 23 S and 5 S rRNA molecules have been fitted helix by helix to a cryo-electron microscopic (EM) reconstruction of the 50 S ribosomal subunit, using an unfiltered version of the recently published 50 S reconstruction at 7.5 A resolution. At this resolution, the EM density shows a well-defined network of fine structural elements, in which the major and minor grooves of the rRNA helices can be discerned at many locations. The 3D folding of the rRNA molecules within this EM density is constrained by their well-established secondary structures, and further constraints are provided by intra and inter-rRNA crosslinking data, as well as by tertiary interactions and pseudoknots. RNA-protein cross-link and foot-print sites on the 23 S and 5 S rRNA were used to position the rRNA elements concerned in relation to the known arrangement of the ribosomal proteins as determined by immuno-electron microscopy. The published X-ray or NMR structures of seven 50 S ribosomal proteins or RNA-protein complexes were incorporated into the EM density. The 3D locations of cross-link and foot-print sites to the 23 S rRNA from tRNA bound to the ribosomal A, P or E sites were correlated with the positions of the tRNA molecules directly observed in earlier reconstructions of the 70 S ribosome at 13 A or 20 A. Similarly, the positions of cross-link sites within the peptidyl transferase ring of the 23 S rRNA from the aminoacyl residue of tRNA were correlated with the locations of the CCA ends of the A and P site tRNA. Sites on the 23 S rRNA that are cross-linked to the N termini of peptides of different lengths were all found to lie within or close to the internal tunnel connecting the peptidyl transferase region with the presumed peptide exit site on the solvent side of the 50 S subunit. The post-transcriptionally modified bases in the 23 S rRNA form a cluster close to the peptidyl transferase area. The minimum conserved core elements of the secondary structure of the 23 S rRNA form a compact block within the 3D structure and, conversely, the points corresponding to the locations of expansion segments in 28 S rRNA all lie on the outside of the structure. #1:  ジャーナル: Cell(Cambridge,Mass.) / 年: 1999 ジャーナル: Cell(Cambridge,Mass.) / 年: 1999タイトル: A Detailed View of a Ribosomal Active Site: The Structure of the Gtpase Center at 2.6 Angstroms Resolution. 著者: Wimberly, B.T. / Guymon, R. / Mc Cutcheon, J.P. / White, S. / Ramakrishnan, V. #2:  ジャーナル: Embo J. / 年: 1999 ジャーナル: Embo J. / 年: 1999タイトル: The NMR Structure of the 5S Rrna E-Domain-Protein C25 Complex Shows Pre-Formed and Induced Recognition. 著者: Stoldt, M. / Woehnert, J. / Ohlenschlaeger, O. / Goerlach, M. / Brown, L.R. #3:  ジャーナル: Embo J. / 年: 1999 ジャーナル: Embo J. / 年: 1999タイトル: The Three-Dimensional Structure of the RNA-Binding Domain of Ribosomal Protein L2; a Protein at the Peptidyl Transferase Center of the Ribosome. 著者: Nakagawa, A. / Nakashima, T. / Taniguchi, M. / Hosaka, H. / Kimura, M. / Tanaka, I. #4:  ジャーナル: J.Mol.Biol. / 年: 1996 ジャーナル: J.Mol.Biol. / 年: 1996タイトル: Ribosomal Protein L9: A Structure Determination by the Combined Use of X-Ray Crystallography and NMR Spectroscopy. 著者: Hoffman, D.W. / Cameron, C.S. / Davies, C. / White, S.W. / Ramakrishnan, V. #5:  ジャーナル: Embo J. / 年: 1996 ジャーナル: Embo J. / 年: 1996タイトル: Crystal Structure of the RNA Binding Ribosomal Protein L1 from Thermus Thermophilus. 著者: Nikonov, S. / Nevskaya, N. / Eliseikina, I. / Fomenkova, N. / Nikulin, A. / Ossina, N. / Garber, M. / Jonsson, B.H. / Briand, C. / Al-Karadaghi, S. / Svensson, A. / Aevarsson, A. / Liljas, A. #6:  ジャーナル: Structure / 年: 1996 ジャーナル: Structure / 年: 1996タイトル: The Crystal Structure of Ribosomal Protein L14 Reveals an Important Organizational Component of the Translational Apparatus. 著者: Davies, C. / White, S.W. / Ramakrishnan, V. #7:  ジャーナル: Embo J. / 年: 1993 ジャーナル: Embo J. / 年: 1993タイトル: Ribosomal Protein L6: Structural Evidence of Gene Duplication from a Primitive RNA Binding Proetin. 著者: Golden, B.L. / Ramakrishnan, V. / White, S.W. #8:  ジャーナル: Structure / 年: 1999 ジャーナル: Structure / 年: 1999タイトル: The Escherichia coli large ribosomal subunit at 7.5 A resolution 著者: Matadeen, R. / Patwardhan, A. / Gowen, B. / Orlova, E.V. / Pape, T. / Cuff, M. / Mueller, F. / Brimacombe, R. / van Heel, M. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| ムービー |
 ムービービューア ムービービューア |
|---|---|
| 構造ビューア | 分子:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- ダウンロードとリンク
ダウンロードとリンク
- ダウンロード
ダウンロード
| PDBx/mmCIF形式 |  487d.cif.gz 487d.cif.gz | 224 KB | 表示 |  PDBx/mmCIF形式 PDBx/mmCIF形式 |
|---|---|---|---|---|
| PDB形式 |  pdb487d.ent.gz pdb487d.ent.gz | 176.7 KB | 表示 |  PDB形式 PDB形式 |
| PDBx/mmJSON形式 |  487d.json.gz 487d.json.gz | ツリー表示 |  PDBx/mmJSON形式 PDBx/mmJSON形式 | |
| その他 |  その他のダウンロード その他のダウンロード |
-検証レポート
| 文書・要旨 |  487d_validation.pdf.gz 487d_validation.pdf.gz | 396.8 KB | 表示 |  wwPDB検証レポート wwPDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  487d_full_validation.pdf.gz 487d_full_validation.pdf.gz | 439.5 KB | 表示 | |
| XML形式データ |  487d_validation.xml.gz 487d_validation.xml.gz | 25.7 KB | 表示 | |
| CIF形式データ |  487d_validation.cif.gz 487d_validation.cif.gz | 38.8 KB | 表示 | |
| アーカイブディレクトリ |  https://data.pdbj.org/pub/pdb/validation_reports/87/487d https://data.pdbj.org/pub/pdb/validation_reports/87/487d ftp://data.pdbj.org/pub/pdb/validation_reports/87/487d ftp://data.pdbj.org/pub/pdb/validation_reports/87/487d | HTTPS FTP |
-関連構造データ
- リンク
リンク
- 集合体
集合体
| 登録構造単位 | 
|
|---|---|
| 1 |
|
- 要素
要素
-50S ribosomal protein ... , 7種, 7分子 HIJKLMN
| #1: タンパク質 | 分子量: 24331.074 Da / 分子数: 1 / 由来タイプ: 天然 / 由来: (天然)   Thermus thermophilus (バクテリア) / 参照: UniProt: P27150, UniProt: Q5SLP7*PLUS Thermus thermophilus (バクテリア) / 参照: UniProt: P27150, UniProt: Q5SLP7*PLUS |
|---|---|
| #2: タンパク質 | 分子量: 14759.666 Da / 分子数: 1 / 由来タイプ: 天然 由来: (天然)   Geobacillus stearothermophilus (バクテリア) Geobacillus stearothermophilus (バクテリア)参照: UniProt: P04257 |
| #3: タンパク質 | 分子量: 17811.447 Da / 分子数: 1 / 由来タイプ: 天然 由来: (天然)   Geobacillus stearothermophilus (バクテリア) Geobacillus stearothermophilus (バクテリア)参照: UniProt: P02391 |
| #4: タンパク質 | 分子量: 16341.037 Da / 分子数: 1 / 由来タイプ: 天然 由来: (天然)   Geobacillus stearothermophilus (バクテリア) Geobacillus stearothermophilus (バクテリア)参照: UniProt: P02417 |
| #5: タンパク質 | 分子量: 14294.913 Da / 分子数: 1 / 由来タイプ: 天然 / 由来: (天然)   Thermotoga maritima (バクテリア) / 参照: UniProt: P29395 Thermotoga maritima (バクテリア) / 参照: UniProt: P29395 |
| #6: タンパク質 | 分子量: 13369.613 Da / 分子数: 1 / 由来タイプ: 天然 由来: (天然)   Geobacillus stearothermophilus (バクテリア) Geobacillus stearothermophilus (バクテリア)参照: UniProt: P04450 |
| #7: タンパク質 | 分子量: 10713.465 Da / 分子数: 1 / 由来タイプ: 天然 / 由来: (天然)  |
-実験情報
-実験
| 実験 | 手法: 電子顕微鏡法 |
|---|---|
| EM実験 | 試料の集合状態: PARTICLE / 3次元再構成法: 単粒子再構成法 |
- 試料調製
試料調製
| 構成要素 | 名称: LARGE 50S RIBOSOMAL SUBUNIT / タイプ: RIBOSOME |
|---|---|
| 試料 | 包埋: NO / シャドウイング: NO / 染色: NO / 凍結: YES |
| 結晶 | 解説: THE CRYST1 AND SCALE RECORDS ARE MEANINGLESS. |
- 電子顕微鏡撮影
電子顕微鏡撮影
| 顕微鏡 | モデル: FEI/PHILIPS CM200FEG / 詳細: from Structure 1999 citation |
|---|---|
| 電子銃 | 電子線源:  FIELD EMISSION GUN / 加速電圧: 200 kV / 照射モード: FLOOD BEAM FIELD EMISSION GUN / 加速電圧: 200 kV / 照射モード: FLOOD BEAM |
| 電子レンズ | モード: BRIGHT FIELD / 倍率(公称値): 38000 X / 最大 デフォーカス(公称値): 2000 nm / 最小 デフォーカス(公称値): 1000 nm |
| 試料ホルダ | 試料ホルダーモデル: GATAN LIQUID NITROGEN |
| 撮影 | フィルム・検出器のモデル: GENERIC FILM |
| 画像スキャン | Scanner model: IMAGE SCIENCE PATCHWORK DENSITOMETER |
- 解析
解析
| 対称性 | 点対称性: C1 (非対称) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 3次元再構成 | 解像度: 7.5 Å / 解像度の算出法: FSC 3 SIGMA CUT-OFF / 粒子像の数: 16000 / 対称性のタイプ: POINT | ||||||||||||
| 原子モデル構築 | 空間: REAL 詳細: DETAILS--OTHER REFINEMENT REMARKS- CRYO-EM RECONSTRUCTION THIS FILE HAS BEEN GENERATED BY THE USE OF ALL RELEVANT BIOCHEMICAL CONSTRAINTS AND THE CONSTRAINTS GIVEN BY THE ELECTRON DENSITY ...詳細: DETAILS--OTHER REFINEMENT REMARKS- CRYO-EM RECONSTRUCTION THIS FILE HAS BEEN GENERATED BY THE USE OF ALL RELEVANT BIOCHEMICAL CONSTRAINTS AND THE CONSTRAINTS GIVEN BY THE ELECTRON DENSITY CONTOUR OF THE RIBOSOME, WHICH WAS DERIVED FROM THE CRYO-ELECTRON MICROSCOPIC RECONSTRUCTION. | ||||||||||||
| 原子モデル構築 |
| ||||||||||||
| 精密化 | 最高解像度: 7.5 Å | ||||||||||||
| 精密化ステップ | サイクル: LAST / 最高解像度: 7.5 Å
|
 ムービー
ムービー コントローラー
コントローラー






 PDBj
PDBj








