[English] 日本語
 Yorodumi
Yorodumi- PDB-8fpi: Co-structure of the Respiratory Syncytial Virus RNA-dependent RNA... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8fpi | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Co-structure of the Respiratory Syncytial Virus RNA-dependent RNA polymerase with MRK-1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Components Components |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Keywords Keywords | REPLICATION / RNA-BINDING PROTEIN / RSV / RDRP / RNA-DEPENDENT RNA POLYMERASE / PRNTASE / POLYRIBONUCLEOTIDYL TRANSFERASE / RNA CAPPING / VIRAL REPLICATION / VIRAL PROTEIN | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationRespiratory syncytial virus genome transcription / Translation of respiratory syncytial virus mRNAs / GDP polyribonucleotidyltransferase / negative stranded viral RNA replication / Respiratory syncytial virus genome replication / RSV-host interactions / Assembly and release of respiratory syncytial virus (RSV) virions / Maturation of hRSV A proteins / Respiratory syncytial virus (RSV) attachment and entry / Hydrolases; Acting on acid anhydrides; In phosphorus-containing anhydrides ...Respiratory syncytial virus genome transcription / Translation of respiratory syncytial virus mRNAs / GDP polyribonucleotidyltransferase / negative stranded viral RNA replication / Respiratory syncytial virus genome replication / RSV-host interactions / Assembly and release of respiratory syncytial virus (RSV) virions / Maturation of hRSV A proteins / Respiratory syncytial virus (RSV) attachment and entry / Hydrolases; Acting on acid anhydrides; In phosphorus-containing anhydrides / viral life cycle / virion component / symbiont-mediated suppression of host NF-kappaB cascade / host cell cytoplasm / mRNA 5'-cap (guanine-N7-)-methyltransferase activity / RNA-directed RNA polymerase / RNA-directed RNA polymerase activity / GTPase activity / ATP binding / metal ion binding Similarity search - Function | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological species |  Human respiratory syncytial virus A2 Human respiratory syncytial virus A2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 2.52 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Authors Authors | Fischmann, T.O. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Funding support |  United States, 1items United States, 1items
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Citation Citation |  Journal: Commun Biol / Year: 2023 Journal: Commun Biol / Year: 2023Title: Conserved allosteric inhibitory site on the respiratory syncytial virus and human metapneumovirus RNA-dependent RNA polymerases. Authors: Victoria A Kleiner / Thierry O Fischmann / John A Howe / Douglas C Beshore / Michael J Eddins / Yan Hou / Todd Mayhood / Daniel Klein / Debbie D Nahas / Bob J Lucas / He Xi / Edward Murray / ...Authors: Victoria A Kleiner / Thierry O Fischmann / John A Howe / Douglas C Beshore / Michael J Eddins / Yan Hou / Todd Mayhood / Daniel Klein / Debbie D Nahas / Bob J Lucas / He Xi / Edward Murray / Daphne Y Ma / Krista Getty / Rachel Fearns /  Abstract: Respiratory syncytial virus (RSV) and human metapneumovirus (HMPV) are related RNA viruses responsible for severe respiratory infections and resulting disease in infants, elderly, and ...Respiratory syncytial virus (RSV) and human metapneumovirus (HMPV) are related RNA viruses responsible for severe respiratory infections and resulting disease in infants, elderly, and immunocompromised adults. Therapeutic small molecule inhibitors that bind to the RSV polymerase and inhibit viral replication are being developed, but their binding sites and molecular mechanisms of action remain largely unknown. Here we report a conserved allosteric inhibitory site identified on the L polymerase proteins of RSV and HMPV that can be targeted by a dual-specificity, non-nucleoside inhibitor, termed MRK-1. Cryo-EM structures of the inhibitor in complexes with truncated RSV and full-length HMPV polymerase proteins provide a structural understanding of how MRK-1 is active against both viruses. Functional analyses indicate that MRK-1 inhibits conformational changes necessary for the polymerase to engage in RNA synthesis initiation and to transition into an elongation mode. Competition studies reveal that the MRK-1 binding pocket is distinct from that of a capping inhibitor with an overlapping resistance profile, suggesting that the polymerase conformation bound by MRK-1 may be distinct from that involved in mRNA capping. These findings should facilitate optimization of dual RSV and HMPV replication inhibitors and provide insights into the molecular mechanisms underlying their polymerase activities. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8fpi.cif.gz 8fpi.cif.gz | 569.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8fpi.ent.gz pdb8fpi.ent.gz | 458 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8fpi.json.gz 8fpi.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/fp/8fpi https://data.pdbj.org/pub/pdb/validation_reports/fp/8fpi ftp://data.pdbj.org/pub/pdb/validation_reports/fp/8fpi ftp://data.pdbj.org/pub/pdb/validation_reports/fp/8fpi | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  29365MC  8fpjC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
| #1: Protein | Mass: 173267.922 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Human respiratory syncytial virus A2 / Cell line (production host): Sf9 / Production host: Human respiratory syncytial virus A2 / Cell line (production host): Sf9 / Production host:  References: UniProt: P28887, RNA-directed RNA polymerase, Hydrolases; Acting on acid anhydrides; In phosphorus-containing anhydrides, GDP polyribonucleotidyltransferase, NNS virus cap methyltransferase | ||||||
|---|---|---|---|---|---|---|---|
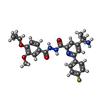
| #2: Protein | Mass: 29062.895 Da / Num. of mol.: 4 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Human respiratory syncytial virus A2 / Cell line (production host): Sf9 / Production host: Human respiratory syncytial virus A2 / Cell line (production host): Sf9 / Production host:  #3: Chemical | ChemComp-Y6L / | Has ligand of interest | Y | Has protein modification | N | |
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: RESPIRATORY SYNCYTIAL VIRUS POLYMERASE (L) PROTEIN BOUND BY THE TETRAMERIC PHOSPHOPROTEIN (P) AND COMPLEXED WITH MRK-1 Type: COMPLEX / Entity ID: #2 / Source: RECOMBINANT |
|---|---|
| Molecular weight | Experimental value: NO |
| Source (natural) | Organism:  Human orthopneumovirus Human orthopneumovirus |
| Source (recombinant) | Organism:  |
| Buffer solution | pH: 7.4 |
| Specimen | Conc.: 0.6 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal defocus max: 2200 nm / Nominal defocus min: 800 nm |
| Image recording | Electron dose: 34.5 e/Å2 / Detector mode: COUNTING / Film or detector model: FEI FALCON III (4k x 4k) |
- Processing
Processing
| EM software | Name: PHENIX / Category: model refinement |
|---|---|
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION |
| Symmetry | Point symmetry: C1 (asymmetric) |
| 3D reconstruction | Resolution: 2.52 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 589457 / Num. of class averages: 13 / Symmetry type: POINT |
| Atomic model building | Space: REAL |
| Refinement | Highest resolution: 2.52 Å |
 Movie
Movie Controller
Controller



 PDBj
PDBj