+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: PDB / ID: 7w0p | ||||||
|---|---|---|---|---|---|---|---|
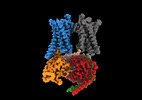
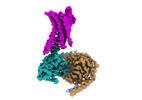
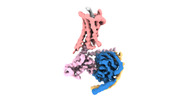
| タイトル | Cryo-EM structure of a GPCR-Gi complex with peptide | ||||||
 要素 要素 |
| ||||||
 キーワード キーワード | MEMBRANE PROTEIN / Complex / GPCR | ||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報mesendoderm migration / mesoderm migration involved in gastrulation / cell migration involved in mesendoderm migration / apelin receptor binding / positive regulation of G protein-coupled receptor internalization / apelin receptor activity / apelin receptor signaling pathway / regulation of gap junction assembly / positive regulation of inhibitory G protein-coupled receptor phosphorylation / vascular associated smooth muscle cell differentiation ...mesendoderm migration / mesoderm migration involved in gastrulation / cell migration involved in mesendoderm migration / apelin receptor binding / positive regulation of G protein-coupled receptor internalization / apelin receptor activity / apelin receptor signaling pathway / regulation of gap junction assembly / positive regulation of inhibitory G protein-coupled receptor phosphorylation / vascular associated smooth muscle cell differentiation / atrioventricular valve development / positive regulation of heart contraction / regulation of body fluid levels / venous blood vessel development / positive regulation of trophoblast cell migration / positive regulation of blood vessel endothelial cell proliferation involved in sprouting angiogenesis / endocardial cushion formation / placenta blood vessel development / negative regulation of cAMP-mediated signaling / endoderm development / C-C chemokine receptor activity / coronary vasculature development / C-C chemokine binding / adult heart development / embryonic heart tube development / vasculature development / aorta development / ventricular septum morphogenesis / blood vessel development / heart looping / Adenylate cyclase inhibitory pathway / positive regulation of protein localization to cell cortex / vasculogenesis / regulation of cAMP-mediated signaling / D2 dopamine receptor binding / G protein-coupled serotonin receptor binding / regulation of mitotic spindle organization / gastrulation / cellular response to forskolin / adenylate cyclase-inhibiting G protein-coupled receptor signaling pathway / Peptide ligand-binding receptors / neurogenesis / positive regulation of release of sequestered calcium ion into cytosol / cell chemotaxis / Regulation of insulin secretion / G protein-coupled receptor binding / G protein-coupled receptor activity / calcium-mediated signaling / electron transport chain / G-protein beta/gamma-subunit complex binding / Olfactory Signaling Pathway / Activation of the phototransduction cascade / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor / hormone activity / G-protein activation / brain development / G protein-coupled acetylcholine receptor signaling pathway / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / Prostacyclin signalling through prostacyclin receptor / Glucagon signaling in metabolic regulation / G beta:gamma signalling through CDC42 / response to peptide hormone / ADP signalling through P2Y purinoceptor 12 / G beta:gamma signalling through BTK / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / Sensory perception of sweet, bitter, and umami (glutamate) taste / photoreceptor disc membrane / Adrenaline,noradrenaline inhibits insulin secretion / Glucagon-type ligand receptors / Vasopressin regulates renal water homeostasis via Aquaporins / G alpha (z) signalling events / cellular response to catecholamine stimulus / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / ADORA2B mediated anti-inflammatory cytokines production / sensory perception of taste / ADP signalling through P2Y purinoceptor 1 / adenylate cyclase-activating dopamine receptor signaling pathway / G beta:gamma signalling through PI3Kgamma / cellular response to prostaglandin E stimulus / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / positive regulation of angiogenesis / GPER1 signaling / GDP binding / G-protein beta-subunit binding / Inactivation, recovery and regulation of the phototransduction cascade / heterotrimeric G-protein complex / G alpha (12/13) signalling events / extracellular vesicle / signaling receptor complex adaptor activity / Thrombin signalling through proteinase activated receptors (PARs) / GTPase binding / retina development in camera-type eye / signaling receptor activity / Ca2+ pathway / heart development / phospholipase C-activating G protein-coupled receptor signaling pathway 類似検索 - 分子機能 | ||||||
| 生物種 |  Homo sapiens (ヒト) Homo sapiens (ヒト) | ||||||
| 手法 | 電子顕微鏡法 / 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 3.16 Å | ||||||
 データ登録者 データ登録者 | Xu, F. / Yue, Y. / Liu, L.E. / Wu, L.J. / Hanson, M. | ||||||
| 資金援助 | 1件
| ||||||
 引用 引用 |  ジャーナル: Nat Struct Mol Biol / 年: 2022 ジャーナル: Nat Struct Mol Biol / 年: 2022タイトル: Structural insight into apelin receptor-G protein stoichiometry. 著者: Yang Yue / Lier Liu / Li-Jie Wu / Yiran Wu / Ling Wang / Fei Li / Junlin Liu / Gye-Won Han / Bo Chen / Xi Lin / Rebecca L Brouillette / Émile Breault / Jean-Michel Longpré / Songting Shi / ...著者: Yang Yue / Lier Liu / Li-Jie Wu / Yiran Wu / Ling Wang / Fei Li / Junlin Liu / Gye-Won Han / Bo Chen / Xi Lin / Rebecca L Brouillette / Émile Breault / Jean-Michel Longpré / Songting Shi / Hui Lei / Philippe Sarret / Raymond C Stevens / Michael A Hanson / Fei Xu /    要旨: The technique of cryogenic-electron microscopy (cryo-EM) has revolutionized the field of membrane protein structure and function with a focus on the dominantly observed molecular species. This report ...The technique of cryogenic-electron microscopy (cryo-EM) has revolutionized the field of membrane protein structure and function with a focus on the dominantly observed molecular species. This report describes the structural characterization of a fully active human apelin receptor (APJR) complexed with heterotrimeric G protein observed in both 2:1 and 1:1 stoichiometric ratios. We use cryo-EM single-particle analysis to determine the structural details of both species from the same sample preparation. Protein preparations, in the presence of the endogenous peptide ligand ELA or a synthetic small molecule, both demonstrate these mixed stoichiometric states. Structural differences in G protein engagement between dimeric and monomeric APJR suggest a role for the stoichiometry of G protein-coupled receptor- (GPCR-)G protein coupling on downstream signaling and receptor pharmacology. Furthermore, a small, hydrophobic dimer interface provides a starting framework for additional class A GPCR dimerization studies. Together, these findings uncover a mechanism of versatile regulation through oligomerization by which GPCRs can modulate their signaling. | ||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 構造ビューア | 分子:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- ダウンロードとリンク
ダウンロードとリンク
- ダウンロード
ダウンロード
| PDBx/mmCIF形式 |  7w0p.cif.gz 7w0p.cif.gz | 234 KB | 表示 |  PDBx/mmCIF形式 PDBx/mmCIF形式 |
|---|---|---|---|---|
| PDB形式 |  pdb7w0p.ent.gz pdb7w0p.ent.gz | 183 KB | 表示 |  PDB形式 PDB形式 |
| PDBx/mmJSON形式 |  7w0p.json.gz 7w0p.json.gz | ツリー表示 |  PDBx/mmJSON形式 PDBx/mmJSON形式 | |
| その他 |  その他のダウンロード その他のダウンロード |
-検証レポート
| 文書・要旨 |  7w0p_validation.pdf.gz 7w0p_validation.pdf.gz | 1 MB | 表示 |  wwPDB検証レポート wwPDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  7w0p_full_validation.pdf.gz 7w0p_full_validation.pdf.gz | 1 MB | 表示 | |
| XML形式データ |  7w0p_validation.xml.gz 7w0p_validation.xml.gz | 42.8 KB | 表示 | |
| CIF形式データ |  7w0p_validation.cif.gz 7w0p_validation.cif.gz | 62.7 KB | 表示 | |
| アーカイブディレクトリ |  https://data.pdbj.org/pub/pdb/validation_reports/w0/7w0p https://data.pdbj.org/pub/pdb/validation_reports/w0/7w0p ftp://data.pdbj.org/pub/pdb/validation_reports/w0/7w0p ftp://data.pdbj.org/pub/pdb/validation_reports/w0/7w0p | HTTPS FTP |
-関連構造データ
| 関連構造データ |  32247MC  7susC  7w0lC  7w0mC  7w0nC  7w0oC M: このデータのモデリングに利用したマップデータ C: 同じ文献を引用 ( |
|---|---|
| 類似構造データ | 類似検索 - 機能・相同性  F&H 検索 F&H 検索 |
- リンク
リンク
- 集合体
集合体
| 登録構造単位 | 
|
|---|---|
| 1 |
|
- 要素
要素
-Guanine nucleotide-binding protein ... , 3種, 3分子 ABC
| #1: タンパク質 | 分子量: 40559.160 Da / 分子数: 1 / 由来タイプ: 組換発現 / 由来: (組換発現)  Homo sapiens (ヒト) / 遺伝子: GNAI1 / 発現宿主: Homo sapiens (ヒト) / 遺伝子: GNAI1 / 発現宿主:  Trichoplusia ni (イラクサキンウワバ) / 参照: UniProt: P63096 Trichoplusia ni (イラクサキンウワバ) / 参照: UniProt: P63096 |
|---|---|
| #2: タンパク質 | 分子量: 37416.930 Da / 分子数: 1 / 由来タイプ: 組換発現 / 由来: (組換発現)  Homo sapiens (ヒト) / 遺伝子: GNB1 / 発現宿主: Homo sapiens (ヒト) / 遺伝子: GNB1 / 発現宿主:  Trichoplusia ni (イラクサキンウワバ) / 参照: UniProt: P62873 Trichoplusia ni (イラクサキンウワバ) / 参照: UniProt: P62873 |
| #3: タンパク質 | 分子量: 7861.143 Da / 分子数: 1 / 由来タイプ: 組換発現 / 由来: (組換発現)  Homo sapiens (ヒト) / 遺伝子: GNG2 / 発現宿主: Homo sapiens (ヒト) / 遺伝子: GNG2 / 発現宿主:  Trichoplusia ni (イラクサキンウワバ) / 参照: UniProt: P59768 Trichoplusia ni (イラクサキンウワバ) / 参照: UniProt: P59768 |
-タンパク質 / 抗体 / タンパク質・ペプチド , 3種, 3分子 RSD
| #4: タンパク質 | 分子量: 54236.480 Da / 分子数: 1 / 由来タイプ: 組換発現 詳細: Fusion protein of Soluble cytochrome b562, linker and Apelin receptor 由来: (組換発現)   Homo sapiens (ヒト) Homo sapiens (ヒト)遺伝子: cybC, APLNR, AGTRL1, APJ / 発現宿主:  Trichoplusia ni (イラクサキンウワバ) / 参照: UniProt: P0ABE7, UniProt: P35414 Trichoplusia ni (イラクサキンウワバ) / 参照: UniProt: P0ABE7, UniProt: P35414 |
|---|---|
| #5: 抗体 | 分子量: 31870.629 Da / 分子数: 1 / 由来タイプ: 組換発現 / 由来: (組換発現)  Homo sapiens (ヒト) / 発現宿主: Homo sapiens (ヒト) / 発現宿主:  Trichoplusia ni (イラクサキンウワバ) Trichoplusia ni (イラクサキンウワバ) |
| #6: タンパク質・ペプチド | 分子量: 3980.873 Da / 分子数: 1 / 由来タイプ: 合成 / 由来: (合成)  Homo sapiens (ヒト) / 参照: UniProt: P0DMC3 Homo sapiens (ヒト) / 参照: UniProt: P0DMC3 |
-実験情報
-実験
| 実験 | 手法: 電子顕微鏡法 |
|---|---|
| EM実験 | 試料の集合状態: PARTICLE / 3次元再構成法: 単粒子再構成法 |
- 試料調製
試料調製
| 構成要素 | 名称: GPCR-Gi Complex / タイプ: COMPLEX / Entity ID: all / 由来: RECOMBINANT |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 由来(組換発現) | 生物種:  Trichoplusia ni (イラクサキンウワバ) Trichoplusia ni (イラクサキンウワバ) |
| 緩衝液 | pH: 7.5 |
| 試料 | 包埋: NO / シャドウイング: NO / 染色: NO / 凍結: YES |
| 急速凍結 | 凍結剤: ETHANE |
- 電子顕微鏡撮影
電子顕微鏡撮影
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
|---|---|
| 顕微鏡 | モデル: FEI TITAN KRIOS |
| 電子銃 | 電子線源:  FIELD EMISSION GUN / 加速電圧: 300 kV / 照射モード: FLOOD BEAM FIELD EMISSION GUN / 加速電圧: 300 kV / 照射モード: FLOOD BEAM |
| 電子レンズ | モード: BRIGHT FIELD |
| 撮影 | 電子線照射量: 60 e/Å2 フィルム・検出器のモデル: GATAN K3 BIOQUANTUM (6k x 4k) |
- 解析
解析
| CTF補正 | タイプ: NONE |
|---|---|
| 3次元再構成 | 解像度: 3.16 Å / 解像度の算出法: FSC 0.143 CUT-OFF / 粒子像の数: 63197 / 対称性のタイプ: POINT |
 ムービー
ムービー コントローラー
コントローラー




 PDBj
PDBj


































