+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7uwk | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of the higher-order IL-25-IL-17RB complex | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Components Components |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Keywords Keywords | CYTOKINE / Receptor complex / IL-17E / IL-25 / IL-17RB | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationinterleukin-17E receptor binding / interleukin-17 receptor activity / eosinophil differentiation / Interleukin-17 signaling / response to fungus / positive regulation of interleukin-5 production / positive regulation of interleukin-13 production / response to nematode / inflammatory response to antigenic stimulus / cytokine receptor activity ...interleukin-17E receptor binding / interleukin-17 receptor activity / eosinophil differentiation / Interleukin-17 signaling / response to fungus / positive regulation of interleukin-5 production / positive regulation of interleukin-13 production / response to nematode / inflammatory response to antigenic stimulus / cytokine receptor activity / cytokine activity / defense response / regulation of cell growth / positive regulation of inflammatory response / cell surface / positive regulation of transcription by RNA polymerase II / : / extracellular region / membrane / plasma membrane / cytoplasm Similarity search - Function | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 4.4 Å | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Authors Authors | Wilson, S.C. / Caveney, N.A. / Jude, K.M. / Garcia, K.C. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Funding support |  United States, 1items United States, 1items
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Citation Citation |  Journal: Nature / Year: 2022 Journal: Nature / Year: 2022Title: Organizing structural principles of the IL-17 ligand-receptor axis. Authors: Steven C Wilson / Nathanael A Caveney / Michelle Yen / Christoph Pollmann / Xinyu Xiang / Kevin M Jude / Maximillian Hafer / Naotaka Tsutsumi / Jacob Piehler / K Christopher Garcia /   Abstract: The IL-17 family of cytokines and receptors have central roles in host defence against infection and development of inflammatory diseases. The compositions and structures of functional IL-17 family ...The IL-17 family of cytokines and receptors have central roles in host defence against infection and development of inflammatory diseases. The compositions and structures of functional IL-17 family ligand-receptor signalling assemblies remain unclear. IL-17E (also known as IL-25) is a key regulator of type 2 immune responses and driver of inflammatory diseases, such as allergic asthma, and requires both IL-17 receptor A (IL-17RA) and IL-17RB to elicit functional responses. Here we studied IL-25-IL-17RB binary and IL-25-IL-17RB-IL-17RA ternary complexes using a combination of cryo-electron microscopy, single-molecule imaging and cell-based signalling approaches. The IL-25-IL-17RB-IL-17RA ternary signalling assembly is a C2-symmetric complex in which the IL-25-IL-17RB homodimer is flanked by two 'wing-like' IL-17RA co-receptors through a 'tip-to-tip' geometry that is the key receptor-receptor interaction required for initiation of signal transduction. IL-25 interacts solely with IL-17RB to allosterically promote the formation of the IL-17RB-IL-17RA tip-to-tip interface. The resulting large separation between the receptors at the membrane-proximal level may reflect proximity constraints imposed by the intracellular domains for signalling. Cryo-electron microscopy structures of IL-17A-IL-17RA and IL-17A-IL-17RA-IL-17RC complexes reveal that this tip-to-tip architecture is a key organizing principle of the IL-17 receptor family. Furthermore, these studies reveal dual actions for IL-17RA sharing among IL-17 cytokine complexes, by either directly engaging IL-17 cytokines or alternatively functioning as a co-receptor. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7uwk.cif.gz 7uwk.cif.gz | 370.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7uwk.ent.gz pdb7uwk.ent.gz | 297.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7uwk.json.gz 7uwk.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/uw/7uwk https://data.pdbj.org/pub/pdb/validation_reports/uw/7uwk ftp://data.pdbj.org/pub/pdb/validation_reports/uw/7uwk ftp://data.pdbj.org/pub/pdb/validation_reports/uw/7uwk | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  26834MC  7uwjC  7uwlC  7uwmC  7uwnC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 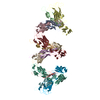
|
|---|---|
| 1 |
|
- Components
Components
| #1: Protein | Mass: 21490.201 Da / Num. of mol.: 6 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: IL25, IL17E, UNQ3120/PRO10272 / Production host: Homo sapiens (human) / Gene: IL25, IL17E, UNQ3120/PRO10272 / Production host:  Homo sapiens (human) / References: UniProt: Q9H293 Homo sapiens (human) / References: UniProt: Q9H293#2: Protein | Mass: 33649.098 Da / Num. of mol.: 6 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: IL17RB, CRL4, EVI27, IL17BR, UNQ2501/PRO19612 / Production host: Homo sapiens (human) / Gene: IL17RB, CRL4, EVI27, IL17BR, UNQ2501/PRO19612 / Production host:  Homo sapiens (human) / References: UniProt: Q9NRM6 Homo sapiens (human) / References: UniProt: Q9NRM6Has protein modification | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: higher-order IL-25-IL-17RB complex / Type: COMPLEX / Entity ID: all / Source: RECOMBINANT |
|---|---|
| Molecular weight | Experimental value: NO |
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Source (recombinant) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Buffer solution | pH: 7.4 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: TFS KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: OTHER FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: OTHER |
| Electron lens | Mode: OTHER / Nominal defocus max: 2200 nm / Nominal defocus min: 800 nm |
| Image recording | Electron dose: 53 e/Å2 / Film or detector model: GATAN K3 (6k x 4k) |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.19.2_4158: / Classification: refinement | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software | Name: PHENIX / Category: model refinement | ||||||||||||||||||||||||
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||
| 3D reconstruction | Resolution: 4.4 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 297300 / Symmetry type: POINT | ||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller







 PDBj
PDBj

