[English] 日本語
 Yorodumi
Yorodumi- PDB-7tec: Structure of the Listeria monocytogenes GlnR-DNA complex to 3.45 ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7tec | ||||||
|---|---|---|---|---|---|---|---|
| Title | Structure of the Listeria monocytogenes GlnR-DNA complex to 3.45 Angstrom | ||||||
 Components Components |
| ||||||
 Keywords Keywords | DNA BINDING PROTEIN/DNA / GlnR / listeria / glutamine synthetase / repressor / transcription / glnRA / DNA BINDING PROTEIN / DNA BINDING PROTEIN-DNA complex | ||||||
| Function / homology |  Function and homology information Function and homology information | ||||||
| Biological species |  Listeria monocytogenes (bacteria) Listeria monocytogenes (bacteria)synthetic construct (others) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / MOLECULAR REPLACEMENT /  molecular replacement / Resolution: 3.45 Å molecular replacement / Resolution: 3.45 Å | ||||||
 Authors Authors | Schumacher, M.A. / Brennan, R.G. | ||||||
| Funding support |  United States, 1items United States, 1items
| ||||||
 Citation Citation |  Journal: Nat Commun / Year: 2022 Journal: Nat Commun / Year: 2022Title: Molecular dissection of the glutamine synthetase-GlnR nitrogen regulatory circuitry in Gram-positive bacteria. Authors: Brady A Travis / Jared V Peck / Raul Salinas / Brandon Dopkins / Nicholas Lent / Viet D Nguyen / Mario J Borgnia / Richard G Brennan / Maria A Schumacher /  Abstract: How bacteria sense and respond to nitrogen levels are central questions in microbial physiology. In Gram-positive bacteria, nitrogen homeostasis is controlled by an operon encoding glutamine ...How bacteria sense and respond to nitrogen levels are central questions in microbial physiology. In Gram-positive bacteria, nitrogen homeostasis is controlled by an operon encoding glutamine synthetase (GS), a dodecameric machine that assimilates ammonium into glutamine, and the GlnR repressor. GlnR detects nitrogen excess indirectly by binding glutamine-feedback-inhibited-GS (FBI-GS), which activates its transcription-repression function. The molecular mechanisms behind this regulatory circuitry, however, are unknown. Here we describe biochemical and structural analyses of GS and FBI-GS-GlnR complexes from pathogenic and non-pathogenic Gram-positive bacteria. The structures show FBI-GS binds the GlnR C-terminal domain within its active-site cavity, juxtaposing two GlnR monomers to form a DNA-binding-competent GlnR dimer. The FBI-GS-GlnR interaction stabilizes the inactive GS conformation. Strikingly, this interaction also favors a remarkable dodecamer to tetradecamer transition in some GS, breaking the paradigm that all bacterial GS are dodecamers. These data thus unveil unique structural mechanisms of transcription and enzymatic regulation. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7tec.cif.gz 7tec.cif.gz | 70.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7tec.ent.gz pdb7tec.ent.gz | 47.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7tec.json.gz 7tec.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/te/7tec https://data.pdbj.org/pub/pdb/validation_reports/te/7tec ftp://data.pdbj.org/pub/pdb/validation_reports/te/7tec ftp://data.pdbj.org/pub/pdb/validation_reports/te/7tec | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  7tdpC  7tdvC  7teaSC  7tenC  7tf6C  7tf7C  7tf9C  7tfaC  7tfbC  7tfcC  7tfdC  7tfeC S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 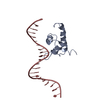
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 14638.857 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Listeria monocytogenes (bacteria) Listeria monocytogenes (bacteria)Gene: glnR, A8L61_07010, ABZ57_05085, AF006_07180, AF817_01990, AMC55_04605, APD94_02400, ART25_07755, ARV28_05490, ARV77_10415, B1N06_02425, B1N38_06025, B1N40_09865, B1N52_08095, B1N70_08585, B1N71_ ...Gene: glnR, A8L61_07010, ABZ57_05085, AF006_07180, AF817_01990, AMC55_04605, APD94_02400, ART25_07755, ARV28_05490, ARV77_10415, B1N06_02425, B1N38_06025, B1N40_09865, B1N52_08095, B1N70_08585, B1N71_01820, B1O10_02055, B1O25_08310, B2H14_10505, B4X79_13085, B4Y29_01290, B4Y40_11845, B4Y47_05380, B4Y49_02050, B4Y56_02055, B4Y57_02055, B5K54_13065, B6112_07615, B6O07_10740, BB997_08390, BCZ19_06080, BW273_06570, C6S26_02850, CW834_10150, CW845_07630, CW895_11495, CX098_14230, D4164_01995, D4271_02955, D4900_04675, D4920_02385, D4947_02360, D4C60_03330, D4D22_02955, D4D89_02780, D4U00_01965, D4U23_10015, D5M63_02050, D5N24_04355, D6P18_02820, D7104_08235, DCK28_08790, DCT16_08455, E0I39_02830, E1V33_00335, E1W43_05380, E1W56_05615, E3W32_09810, E5F58_10230, E5H26_02070, EPC87_01440, EX365_07695, EXZ73_06750, EYY39_05995, F3O35_05110, F3R75_05795, F6436_09515, F6515_11560, FA835_14895, FC284_14445, FJU19_02820, FL871_07620, FLQ97_14195, FLR03_05055, FLR11_13675, FNX31_12240, FNX40_10035, FORC68_1320, FR217_04555, FV747_02470, G3O21_002452, G3R95_000630, GEK29_02375, GFK29_08620, GH165_00575, GHH22_03975, GHM39_05980, GIG92_01790, GIH49_01260, GJW51_07325, GNG71_06855, GON91_01810, GT011_02015, GXB45_00580, GYN46_14485, GYO86_08555, GYP27_02015, GYS19_13395, GYU05_01010, GYU24_02035, GYX95_04495, GYY14_11060, GYZ23_12950, GYZ33_11570, GYZ61_07200, GZI09_15320, GZK40_07615, GZM52_07550, GZN68_09475, HP506_001182, HQN34_002361, I6I37_04555, I8J47_00192, IP987_001477, JKV73_01561, JKV74_07850, JKV76_01310, JKV77_01646, JKX60_08595, JKX61_06825, KV70_04590, KW30_04605, LmNIHS28_00654, M643_00970, QD52_08715, R019_12835, UI29_08755, UP23_04390 Production host:  |
|---|---|
| #2: DNA chain | Mass: 6447.185 Da / Num. of mol.: 1 / Source method: obtained synthetically / Source: (synth.) synthetic construct (others) |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 1.97 Å3/Da / Density % sol: 37.54 % |
|---|---|
| Crystal grow | Temperature: 275 K / Method: vapor diffusion, hanging drop Details: 50 mM sodium cacodylate pH 6.5, 35% MPD, 10 mM MgCl2 |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ALS ALS  / Beamline: 5.0.2 / Wavelength: 1.09 Å / Beamline: 5.0.2 / Wavelength: 1.09 Å |
| Detector | Type: DECTRIS PILATUS 6M / Detector: PIXEL / Date: Feb 12, 2021 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.09 Å / Relative weight: 1 |
| Reflection | Resolution: 3.45→37.51 Å / Num. obs: 2384 / % possible obs: 96.8 % / Redundancy: 4.1 % / CC1/2: 0.998 / Rpim(I) all: 0.038 / Rsym value: 0.064 / Net I/σ(I): 8.9 |
| Reflection shell | Resolution: 3.45→4.34 Å / Mean I/σ(I) obs: 2 / Num. unique obs: 237 / CC1/2: 0.71 / Rpim(I) all: 0.24 / Rsym value: 0.42 |
-Phasing
| Phasing | Method:  molecular replacement molecular replacement |
|---|
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 7TEA Resolution: 3.45→37.51 Å / SU ML: 0.34 / Cross valid method: THROUGHOUT / σ(F): 1.36 / Phase error: 23.48 / Stereochemistry target values: ML
| ||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | ||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 165.47 Å2 / Biso mean: 64.3464 Å2 / Biso min: 20 Å2 | ||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: final / Resolution: 3.45→37.51 Å
| ||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Refine-ID: X-RAY DIFFRACTION / Rfactor Rfree error: 0 / Total num. of bins used: 2 / % reflection obs: 97 %
| ||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Origin x: 2.038 Å / Origin y: 5.37 Å / Origin z: 23.3464 Å
| ||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller










 PDBj
PDBj








































