[English] 日本語
 Yorodumi
Yorodumi- PDB-7t32: CryoEM structure of the adenosine 2A receptor-BRIL/Anti BRIL Fab ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7t32 | ||||||
|---|---|---|---|---|---|---|---|
| Title | CryoEM structure of the adenosine 2A receptor-BRIL/Anti BRIL Fab complex with ZM241385 | ||||||
 Components Components | Adenosine receptor A2a/Soluble cytochrome b562 Fusion Protein | ||||||
 Keywords Keywords | MEMBRANE PROTEIN / A2AAR / GPCR / ADENOSINE RECEPTOR | ||||||
| Function / homology |  Function and homology information Function and homology informationregulation of norepinephrine secretion / positive regulation of acetylcholine secretion, neurotransmission / negative regulation of alpha-beta T cell activation / positive regulation of circadian sleep/wake cycle, sleep / Adenosine P1 receptors / G protein-coupled adenosine receptor activity / response to purine-containing compound / G protein-coupled adenosine receptor signaling pathway / NGF-independant TRKA activation / Surfactant metabolism ...regulation of norepinephrine secretion / positive regulation of acetylcholine secretion, neurotransmission / negative regulation of alpha-beta T cell activation / positive regulation of circadian sleep/wake cycle, sleep / Adenosine P1 receptors / G protein-coupled adenosine receptor activity / response to purine-containing compound / G protein-coupled adenosine receptor signaling pathway / NGF-independant TRKA activation / Surfactant metabolism / synaptic transmission, dopaminergic / type 5 metabotropic glutamate receptor binding / negative regulation of vascular permeability / sensory perception / synaptic transmission, cholinergic / intermediate filament / presynaptic active zone / positive regulation of urine volume / positive regulation of glutamate secretion / response to caffeine / blood circulation / eating behavior / inhibitory postsynaptic potential / regulation of calcium ion transport / alpha-actinin binding / asymmetric synapse / axolemma / membrane depolarization / prepulse inhibition / cellular defense response / phagocytosis / positive regulation of synaptic transmission, glutamatergic / neuron projection morphogenesis / astrocyte activation / presynaptic modulation of chemical synaptic transmission / positive regulation of synaptic transmission, GABAergic / positive regulation of long-term synaptic potentiation / central nervous system development / positive regulation of protein secretion / response to amphetamine / regulation of mitochondrial membrane potential / positive regulation of apoptotic signaling pathway / excitatory postsynaptic potential / synaptic transmission, glutamatergic / apoptotic signaling pathway / locomotory behavior / electron transport chain / negative regulation of inflammatory response / vasodilation / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / blood coagulation / cell-cell signaling / adenylate cyclase-activating G protein-coupled receptor signaling pathway / presynaptic membrane / G alpha (s) signalling events / phospholipase C-activating G protein-coupled receptor signaling pathway / negative regulation of neuron apoptotic process / postsynaptic membrane / calmodulin binding / periplasmic space / electron transfer activity / positive regulation of ERK1 and ERK2 cascade / iron ion binding / response to xenobiotic stimulus / inflammatory response / negative regulation of cell population proliferation / neuronal cell body / apoptotic process / heme binding / regulation of DNA-templated transcription / dendrite / lipid binding / protein-containing complex binding / glutamatergic synapse / enzyme binding / membrane / identical protein binding / plasma membrane Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.4 Å | ||||||
 Authors Authors | Zhang, K.H. / Wu, H. / Hoppe, N. / Manglik, A. / Cheng, Y.F. | ||||||
| Funding support |  United States, 1items United States, 1items
| ||||||
 Citation Citation |  Journal: Nat Commun / Year: 2022 Journal: Nat Commun / Year: 2022Title: Fusion protein strategies for cryo-EM study of G protein-coupled receptors. Authors: Kaihua Zhang / Hao Wu / Nicholas Hoppe / Aashish Manglik / Yifan Cheng /  Abstract: Single particle cryogenic-electron microscopy (cryo-EM) is used extensively to determine structures of activated G protein-coupled receptors (GPCRs) in complex with G proteins or arrestins. However, ...Single particle cryogenic-electron microscopy (cryo-EM) is used extensively to determine structures of activated G protein-coupled receptors (GPCRs) in complex with G proteins or arrestins. However, applying it to GPCRs without signaling proteins remains challenging because most receptors lack structural features in their soluble domains to facilitate image alignment. In GPCR crystallography, inserting a fusion protein between transmembrane helices 5 and 6 is a highly successful strategy for crystallization. Although a similar strategy has the potential to broadly facilitate cryo-EM structure determination of GPCRs alone without signaling protein, the critical determinants that make this approach successful are not yet clear. Here, we address this shortcoming by exploring different fusion protein designs, which lead to structures of antagonist bound A adenosine receptor at 3.4 Å resolution and unliganded Smoothened at 3.7 Å resolution. The fusion strategies explored here are likely applicable to cryo-EM interrogation of other GPCRs and small integral membrane proteins. #1:  Journal: Science / Year: 2012 Journal: Science / Year: 2012Title: Structural Basis for Allosteric Regulation of GPCRs by Sodium Ions Authors: Liu, W. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7t32.cif.gz 7t32.cif.gz | 100.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7t32.ent.gz pdb7t32.ent.gz | 70.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7t32.json.gz 7t32.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/t3/7t32 https://data.pdbj.org/pub/pdb/validation_reports/t3/7t32 ftp://data.pdbj.org/pub/pdb/validation_reports/t3/7t32 ftp://data.pdbj.org/pub/pdb/validation_reports/t3/7t32 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  25648MC  8cxoC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
| #1: Protein | Mass: 43415.855 Da / Num. of mol.: 1 / Mutation: YES Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human), (gene. exp.) Homo sapiens (human), (gene. exp.)  Gene: ADORA2A, ADORA2, cybC / Production host:  Homo sapiens (human) / References: UniProt: P29274, UniProt: P0ABE7 Homo sapiens (human) / References: UniProt: P29274, UniProt: P0ABE7 |
|---|---|
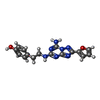
| #2: Chemical | ChemComp-ZMA / |
| Has ligand of interest | Y |
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: CELL / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: A2A adenosine receptor-BRIL/Anti BRIL Fab complex / Type: COMPLEX / Entity ID: #1 / Source: RECOMBINANT |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Source (recombinant) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Buffer solution | pH: 7.5 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Vitrification | Cryogen name: OTHER |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: SPOT SCAN FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: SPOT SCAN |
| Electron lens | Mode: BRIGHT FIELD / Nominal defocus max: 2000 nm / Nominal defocus min: 1000 nm |
| Image recording | Electron dose: 67 e/Å2 / Film or detector model: GATAN K3 (6k x 4k) |
- Processing
Processing
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION |
|---|---|
| 3D reconstruction | Resolution: 3.4 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 215946 / Symmetry type: POINT |
 Movie
Movie Controller
Controller




 PDBj
PDBj