[English] 日本語
 Yorodumi
Yorodumi- PDB-7oqa: Ternary complex of 14-3-3 sigma, Pin1pS72 phosphopeptide, and WQ162 -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7oqa | ||||||
|---|---|---|---|---|---|---|---|
| Title | Ternary complex of 14-3-3 sigma, Pin1pS72 phosphopeptide, and WQ162 | ||||||
 Components Components |
| ||||||
 Keywords Keywords | SIGNALING PROTEIN / protein-peptide complex small-molecule | ||||||
| Function / homology |  Function and homology information Function and homology informationcis-trans isomerase activity / phosphothreonine residue binding / negative regulation of cell motility / negative regulation of brown fat cell differentiation / regulation of protein localization to nucleus / mitogen-activated protein kinase kinase binding / GTPase activating protein binding / ubiquitin ligase activator activity / : / protein peptidyl-prolyl isomerization ...cis-trans isomerase activity / phosphothreonine residue binding / negative regulation of cell motility / negative regulation of brown fat cell differentiation / regulation of protein localization to nucleus / mitogen-activated protein kinase kinase binding / GTPase activating protein binding / ubiquitin ligase activator activity / : / protein peptidyl-prolyl isomerization / regulation of mitotic nuclear division / regulation of epidermal cell division / protein kinase C inhibitor activity / positive regulation of epidermal cell differentiation / keratinocyte development / keratinization / negative regulation of SMAD protein signal transduction / PI5P Regulates TP53 Acetylation / regulation of cell-cell adhesion / negative regulation of amyloid-beta formation / cytoskeletal motor activity / establishment of skin barrier / Regulation of localization of FOXO transcription factors / keratinocyte proliferation / Activation of BAD and translocation to mitochondria / phosphoserine residue binding / RHO GTPases Activate NADPH Oxidases / negative regulation of keratinocyte proliferation / cAMP/PKA signal transduction / negative regulation of protein localization to plasma membrane / SARS-CoV-2 targets host intracellular signalling and regulatory pathways / postsynaptic cytosol / negative regulation of protein kinase activity / negative regulation of stem cell proliferation / Rho protein signal transduction / SARS-CoV-1 targets host intracellular signalling and regulatory pathways / RHO GTPases activate PKNs / Chk1/Chk2(Cds1) mediated inactivation of Cyclin B:Cdk1 complex / positive regulation of protein localization / protein export from nucleus / TP53 Regulates Transcription of Genes Involved in G2 Cell Cycle Arrest / positive regulation of cell adhesion / negative regulation of innate immune response / release of cytochrome c from mitochondria / positive regulation of protein export from nucleus / stem cell proliferation / regulation of cytokinesis / TP53 Regulates Metabolic Genes / peptidylprolyl isomerase / peptidyl-prolyl cis-trans isomerase activity / Translocation of SLC2A4 (GLUT4) to the plasma membrane / Negative regulators of DDX58/IFIH1 signaling / negative regulation of transforming growth factor beta receptor signaling pathway / protein sequestering activity / regulation of protein stability / phosphoprotein binding / negative regulation of protein catabolic process / negative regulation of ERK1 and ERK2 cascade / positive regulation of protein phosphorylation / synapse organization / beta-catenin binding / protein destabilization / ISG15 antiviral mechanism / tau protein binding / intrinsic apoptotic signaling pathway in response to DNA damage / neuron differentiation / intracellular protein localization / positive regulation of canonical Wnt signaling pathway / regulation of protein localization / positive regulation of cell growth / regulation of gene expression / midbody / sperm midpiece / cellular response to hypoxia / Regulation of TP53 Activity through Phosphorylation / response to hypoxia / regulation of cell cycle / nuclear speck / protein stabilization / ciliary basal body / cadherin binding / protein kinase binding / glutamatergic synapse / negative regulation of transcription by RNA polymerase II / signal transduction / positive regulation of transcription by RNA polymerase II / : / extracellular exosome / nucleoplasm / identical protein binding / nucleus / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 1.8 Å MOLECULAR REPLACEMENT / Resolution: 1.8 Å | ||||||
 Authors Authors | Centorrino, F. / Ottmann, C. | ||||||
| Funding support | European Union, 1items
| ||||||
 Citation Citation |  Journal: To Be Published Journal: To Be PublishedTitle: A crystallography-based study of fragment extensions into the 14-3-3 binding groove Authors: Centorrino, F. / Wu, Q. / Cossar, P. / Brunsveld, L. / Ottmann, C. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7oqa.cif.gz 7oqa.cif.gz | 134.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7oqa.ent.gz pdb7oqa.ent.gz | 84.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7oqa.json.gz 7oqa.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/oq/7oqa https://data.pdbj.org/pub/pdb/validation_reports/oq/7oqa ftp://data.pdbj.org/pub/pdb/validation_reports/oq/7oqa ftp://data.pdbj.org/pub/pdb/validation_reports/oq/7oqa | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  7opwC  7oq7C  7oq8C 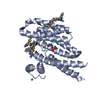 7oq9C  7oqgC  7oqjC  7oqsC  7oquC  7oqwC  7or3C  7or5C  7or7C  7or8C  7orgC  7orhC  7orsC  7ortC  4jc3S S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||||||
| Unit cell |
| ||||||||||||
| Components on special symmetry positions |
|
- Components
Components
| #1: Protein | Mass: 28226.518 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: SFN, HME1 / Production host: Homo sapiens (human) / Gene: SFN, HME1 / Production host:  | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| #2: Protein/peptide | Mass: 2195.378 Da / Num. of mol.: 1 / Source method: obtained synthetically / Source: (synth.)  Homo sapiens (human) / References: UniProt: Q13526, peptidylprolyl isomerase Homo sapiens (human) / References: UniProt: Q13526, peptidylprolyl isomerase | ||||||||
| #3: Chemical | | #4: Chemical | ChemComp-CL / | #5: Water | ChemComp-HOH / | Has ligand of interest | Y | Has protein modification | Y | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.36 Å3/Da / Density % sol: 47.98 % |
|---|---|
| Crystal grow | Temperature: 277.15 K / Method: vapor diffusion, sitting drop Details: 0.095 M Hepes pH7.1, 27%PEG 400, 0.19 M CaCl2, and 5 % Glycerol |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source: SEALED TUBE / Type: RIGAKU MICROMAX-003 / Wavelength: 1.54187 Å |
| Detector | Type: DECTRIS PILATUS 200K / Detector: PIXEL / Date: Nov 20, 2020 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.54187 Å / Relative weight: 1 |
| Reflection | Resolution: 1.8→34.48 Å / Num. obs: 26680 / % possible obs: 98.4 % / Redundancy: 6.1 % / Biso Wilson estimate: 12.25 Å2 / CC1/2: 0.998 / Rmerge(I) obs: 0.053 / Net I/σ(I): 20.2 |
| Reflection shell | Resolution: 1.8→1.83 Å / Rmerge(I) obs: 0.171 / Mean I/σ(I) obs: 6.3 / Num. unique obs: 1199 / CC1/2: 0.977 / % possible all: 88.9 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 4jc3 Resolution: 1.8→34.48 Å / SU ML: 0.1915 / Cross valid method: FREE R-VALUE / σ(F): 1.34 / Phase error: 20.6889 / Stereochemistry target values: GeoStd + Monomer Library
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 16.54 Å2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.8→34.48 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller


 PDBj
PDBj
















