[English] 日本語
 Yorodumi
Yorodumi- PDB-7lxt: Structure of Plasmodium falciparum 20S proteasome with bound bort... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7lxt | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of Plasmodium falciparum 20S proteasome with bound bortezomib | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Components Components |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Keywords Keywords | HYDROLASE / proteasome / plasmodium falciparum / malaria / drug / bortezomib | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationER-Phagosome pathway / Cross-presentation of soluble exogenous antigens (endosomes) / Proteasome assembly / Antigen processing: Ub, ATP-independent proteasomal degradation / Orc1 removal from chromatin / CDK-mediated phosphorylation and removal of Cdc6 / FBXL7 down-regulates AURKA during mitotic entry and in early mitosis / KEAP1-NFE2L2 pathway / UCH proteinases / Ub-specific processing proteases ...ER-Phagosome pathway / Cross-presentation of soluble exogenous antigens (endosomes) / Proteasome assembly / Antigen processing: Ub, ATP-independent proteasomal degradation / Orc1 removal from chromatin / CDK-mediated phosphorylation and removal of Cdc6 / FBXL7 down-regulates AURKA during mitotic entry and in early mitosis / KEAP1-NFE2L2 pathway / UCH proteinases / Ub-specific processing proteases / Neddylation / Antigen processing: Ubiquitination & Proteasome degradation / MAPK6/MAPK4 signaling / ABC-family protein mediated transport / AUF1 (hnRNP D0) binds and destabilizes mRNA / Neutrophil degranulation / proteasome core complex / proteasome endopeptidase complex / proteasome core complex, beta-subunit complex / threonine-type endopeptidase activity / proteasome core complex, alpha-subunit complex / endopeptidase activity / ubiquitin-dependent protein catabolic process / proteasome-mediated ubiquitin-dependent protein catabolic process / hydrolase activity / nucleus / cytoplasm / cytosol Similarity search - Function | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological species |  | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.4 Å | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Authors Authors | Morton, C.J. / Metcalfe, R.D. / Liu, B. / Xie, S.C. / Hanssen, E. / Leis, A.P. / Tilley, L. / Griffin, M.D.W. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Funding support |  Japan, Japan,  Australia, 2items Australia, 2items
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Citation Citation |  Journal: Proc Natl Acad Sci U S A / Year: 2021 Journal: Proc Natl Acad Sci U S A / Year: 2021Title: Design of proteasome inhibitors with oral efficacy in vivo against and selectivity over the human proteasome. Authors: Stanley C Xie / Riley D Metcalfe / Hirotake Mizutani / Tanya Puhalovich / Eric Hanssen / Craig J Morton / Yawei Du / Con Dogovski / Shih-Chung Huang / Jeffrey Ciavarri / Paul Hales / Robert ...Authors: Stanley C Xie / Riley D Metcalfe / Hirotake Mizutani / Tanya Puhalovich / Eric Hanssen / Craig J Morton / Yawei Du / Con Dogovski / Shih-Chung Huang / Jeffrey Ciavarri / Paul Hales / Robert J Griffin / Lawrence H Cohen / Bei-Ching Chuang / Sergio Wittlin / Ioanna Deni / Tomas Yeo / Kurt E Ward / Daniel C Barry / Boyin Liu / David L Gillett / Benigno F Crespo-Fernandez / Sabine Ottilie / Nimisha Mittal / Alisje Churchyard / Daniel Ferguson / Anna Caroline C Aguiar / Rafael V C Guido / Jake Baum / Kirsten K Hanson / Elizabeth A Winzeler / Francisco-Javier Gamo / David A Fidock / Delphine Baud / Michael W Parker / Stephen Brand / Lawrence R Dick / Michael D W Griffin / Alexandra E Gould / Leann Tilley /       Abstract: The proteasome is a potential antimalarial drug target. We have identified a series of amino-amide boronates that are potent and specific inhibitors of the 20S proteasome (20S) β5 active site and ...The proteasome is a potential antimalarial drug target. We have identified a series of amino-amide boronates that are potent and specific inhibitors of the 20S proteasome (20S) β5 active site and that exhibit fast-acting antimalarial activity. They selectively inhibit the growth of compared with a human cell line and exhibit high potency against field isolates of and They have a low propensity for development of resistance and possess liver stage and transmission-blocking activity. Exemplar compounds, MPI-5 and MPI-13, show potent activity against infections in a SCID mouse model with an oral dosing regimen that is well tolerated. We show that MPI-5 binds more strongly to 20S than to human constitutive 20S (20Sc). Comparison of the cryo-electron microscopy (EM) structures of 20S and 20Sc in complex with MPI-5 and 20S in complex with the clinically used anti-cancer agent, bortezomib, reveal differences in binding modes that help to explain the selectivity. Together, this work provides insights into the 20S proteasome in , underpinning the design of potent and selective antimalarial proteasome inhibitors. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7lxt.cif.gz 7lxt.cif.gz | 1.1 MB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7lxt.ent.gz pdb7lxt.ent.gz | 902.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7lxt.json.gz 7lxt.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/lx/7lxt https://data.pdbj.org/pub/pdb/validation_reports/lx/7lxt ftp://data.pdbj.org/pub/pdb/validation_reports/lx/7lxt ftp://data.pdbj.org/pub/pdb/validation_reports/lx/7lxt | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  23574MC  7lxuC  7lxvC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-20S proteasome alpha- ... , 7 types, 14 molecules AOBPCQDRESFTGU
| #1: Protein | Mass: 29531.656 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  Strain: isolate 3D7 References: UniProt: Q8IAR3, proteasome endopeptidase complex #2: Protein | Mass: 26556.391 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  Strain: isolate 3D7 References: UniProt: C6KST3, proteasome endopeptidase complex #3: Protein | Mass: 27977.664 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  Strain: isolate 3D7 References: UniProt: Q8IDG3, proteasome endopeptidase complex #4: Protein | Mass: 27263.285 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  Strain: isolate 3D7 References: UniProt: Q8IDG2, proteasome endopeptidase complex #5: Protein | Mass: 28417.367 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  Strain: isolate 3D7 References: UniProt: Q8IBI3, proteasome endopeptidase complex #6: Protein | Mass: 28871.697 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  Strain: isolate 3D7 References: UniProt: Q8IK90, proteasome endopeptidase complex #7: Protein | Mass: 29324.295 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  Strain: isolate 3D7 References: UniProt: O77396, proteasome endopeptidase complex |
|---|
-20S proteasome beta- ... , 7 types, 14 molecules HVIWJXKYLZMaNb
| #8: Protein | Mass: 29143.936 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  Strain: isolate 3D7 References: UniProt: Q8I0U7, proteasome endopeptidase complex #9: Protein | Mass: 25104.885 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  Strain: isolate 3D7 References: UniProt: Q8I6T3, proteasome endopeptidase complex #10: Protein | Mass: 24533.131 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  Strain: isolate 3D7 References: UniProt: Q8I261, proteasome endopeptidase complex #11: Protein | Mass: 22889.105 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  Strain: isolate 3D7 References: UniProt: Q8IKC9, proteasome endopeptidase complex #12: Protein | Mass: 23620.646 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  Strain: isolate 3D7 References: UniProt: Q8IJT1, proteasome endopeptidase complex #13: Protein | Mass: 27301.203 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  Strain: isolate 3D7 References: UniProt: A0A5K1K7U1, proteasome endopeptidase complex #14: Protein | Mass: 30909.893 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  Strain: isolate 3D7 References: UniProt: Q7K6A9, proteasome endopeptidase complex |
|---|
-Non-polymers , 1 types, 6 molecules 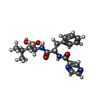
| #15: Chemical | ChemComp-BO2 / |
|---|
-Details
| Has ligand of interest | Y |
|---|---|
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Plasmodium falciparum 20S proteasome complexed with bortezomib Type: COMPLEX / Entity ID: #1-#7, #10-#11, #13-#14 / Source: NATURAL |
|---|---|
| Molecular weight | Value: 0.7 MDa / Experimental value: NO |
| Source (natural) | Organism:  |
| Buffer solution | pH: 7.4 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Specimen support | Grid material: GOLD / Grid mesh size: 300 divisions/in. / Grid type: Quantifoil R1.2/1.3 |
| Vitrification | Instrument: FEI VITROBOT MARK III / Cryogen name: ETHANE / Humidity: 100 % / Chamber temperature: 295 K |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TALOS ARCTICA |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 200 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 200 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Cs: 2.7 mm / C2 aperture diameter: 50 µm |
| Image recording | Electron dose: 50 e/Å2 / Detector mode: SUPER-RESOLUTION / Film or detector model: GATAN K2 SUMMIT (4k x 4k) |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.18.2_3874: / Classification: refinement | |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software |
| |||||||||||||||||||||||||
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | |||||||||||||||||||||||||
| Symmetry | Point symmetry: C2 (2 fold cyclic) | |||||||||||||||||||||||||
| 3D reconstruction | Resolution: 3.4 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 24536 / Symmetry type: POINT | |||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller
























 PDBj
PDBj




