+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7db6 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | human melatonin receptor MT1 - Gi1 complex | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Components Components |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Keywords Keywords | SIGNALING PROTEIN / gpcr / g-protein / complex | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationmelatonin receptor activity / Class A/1 (Rhodopsin-like receptors) / mating behavior / Activation of G protein gated Potassium channels / G-protein activation / G beta:gamma signalling through PI3Kgamma / Prostacyclin signalling through prostacyclin receptor / G beta:gamma signalling through PLC beta / ADP signalling through P2Y purinoceptor 1 / Thromboxane signalling through TP receptor ...melatonin receptor activity / Class A/1 (Rhodopsin-like receptors) / mating behavior / Activation of G protein gated Potassium channels / G-protein activation / G beta:gamma signalling through PI3Kgamma / Prostacyclin signalling through prostacyclin receptor / G beta:gamma signalling through PLC beta / ADP signalling through P2Y purinoceptor 1 / Thromboxane signalling through TP receptor / Presynaptic function of Kainate receptors / G beta:gamma signalling through CDC42 / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / G alpha (12/13) signalling events / Glucagon-type ligand receptors / G beta:gamma signalling through BTK / ADP signalling through P2Y purinoceptor 12 / Adrenaline,noradrenaline inhibits insulin secretion / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / Ca2+ pathway / G alpha (z) signalling events / Thrombin signalling through proteinase activated receptors (PARs) / Extra-nuclear estrogen signaling / G alpha (s) signalling events / G alpha (q) signalling events / G alpha (i) signalling events / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / High laminar flow shear stress activates signaling by PIEZO1 and PECAM1:CDH5:KDR in endothelial cells / Vasopressin regulates renal water homeostasis via Aquaporins / hormone binding / G protein-coupled receptor signaling pathway, coupled to cyclic nucleotide second messenger / adenylate cyclase inhibitor activity / positive regulation of protein localization to cell cortex / T cell migration / positive regulation of relaxation of smooth muscle / Adenylate cyclase inhibitory pathway / D2 dopamine receptor binding / adenylate cyclase-inhibiting serotonin receptor signaling pathway / G protein-coupled serotonin receptor binding / cellular response to forskolin / regulation of mitotic spindle organization / chemokine-mediated signaling pathway / Regulation of insulin secretion / neuropeptide signaling pathway / response to prostaglandin E / circadian rhythm / positive regulation of cholesterol biosynthetic process / negative regulation of insulin secretion / G protein-coupled receptor binding / response to peptide hormone / G protein-coupled receptor activity / G-protein beta/gamma-subunit complex binding / centriolar satellite / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / adenylate cyclase-inhibiting G protein-coupled receptor signaling pathway / ADP signalling through P2Y purinoceptor 12 / GDP binding / Adrenaline,noradrenaline inhibits insulin secretion / G alpha (z) signalling events / cellular response to catecholamine stimulus / ADORA2B mediated anti-inflammatory cytokines production / adenylate cyclase-activating dopamine receptor signaling pathway / GPER1 signaling / cellular response to prostaglandin E stimulus / heterotrimeric G-protein complex / G-protein beta-subunit binding / sperm principal piece / adenylate cyclase-activating G protein-coupled receptor signaling pathway / G protein activity / midbody / cell cortex / G alpha (i) signalling events / G alpha (s) signalling events / Hydrolases; Acting on acid anhydrides; Acting on GTP to facilitate cellular and subcellular movement / signaling receptor complex / Extra-nuclear estrogen signaling / ciliary basal body / G protein-coupled receptor signaling pathway / cell division / lysosomal membrane / GTPase activity / centrosome / GTP binding / nucleolus / magnesium ion binding / Golgi apparatus / extracellular exosome / nucleoplasm / membrane / plasma membrane / cytoplasm / cytosol Similarity search - Function | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human)   | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.3 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Authors Authors | Okamoto, H.H. / Kusakizako, T. / Shihioya, W. / Yamashita, K. / Nishizawa, T. / Nureki, O. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2021 Journal: Nat Struct Mol Biol / Year: 2021Title: Cryo-EM structure of the human MT-G signaling complex. Authors: Hiroyuki H Okamoto / Hirotake Miyauchi / Asuka Inoue / Francesco Raimondi / Hirokazu Tsujimoto / Tsukasa Kusakizako / Wataru Shihoya / Keitaro Yamashita / Ryoji Suno / Norimichi Nomura / ...Authors: Hiroyuki H Okamoto / Hirotake Miyauchi / Asuka Inoue / Francesco Raimondi / Hirokazu Tsujimoto / Tsukasa Kusakizako / Wataru Shihoya / Keitaro Yamashita / Ryoji Suno / Norimichi Nomura / Takuya Kobayashi / So Iwata / Tomohiro Nishizawa / Osamu Nureki /    Abstract: Melatonin receptors (MT and MT) transduce inhibitory signaling by melatonin (N-acetyl-5-methoxytryptamine), which is associated with sleep induction and circadian rhythm modulation. Although recently ...Melatonin receptors (MT and MT) transduce inhibitory signaling by melatonin (N-acetyl-5-methoxytryptamine), which is associated with sleep induction and circadian rhythm modulation. Although recently reported crystal structures of ligand-bound MT and MT elucidated the basis of ligand entry and recognition, the ligand-induced MT rearrangement leading to G-coupling remains unclear. Here we report a cryo-EM structure of the human MT-G signaling complex at 3.3 Å resolution, revealing melatonin-induced conformational changes propagated to the G-protein-coupling interface during activation. In contrast to other G-coupled receptors, MT exhibits a large outward movement of TM6, which is considered a specific feature of G-coupled receptors. Structural comparison of G and G complexes demonstrated conformational diversity of the C-terminal entry of the G protein, suggesting loose and variable interactions at the end of the α5 helix of G protein. These notions, together with our biochemical and computational analyses, highlight variable binding modes of Gα and provide the basis for the selectivity of G-protein signaling. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7db6.cif.gz 7db6.cif.gz | 212.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7db6.ent.gz pdb7db6.ent.gz | 163.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7db6.json.gz 7db6.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/db/7db6 https://data.pdbj.org/pub/pdb/validation_reports/db/7db6 ftp://data.pdbj.org/pub/pdb/validation_reports/db/7db6 ftp://data.pdbj.org/pub/pdb/validation_reports/db/7db6 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  30627MC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | |
| EM raw data |  EMPIAR-10714 (Title: human melatonin receptor MT1 - Gi1 complex / Data size: 878.0 EMPIAR-10714 (Title: human melatonin receptor MT1 - Gi1 complex / Data size: 878.0 Data #1: Unaligned movies for MT1-Gi complex [micrographs - multiframe]) |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Guanine nucleotide-binding protein ... , 3 types, 3 molecules ABC
| #1: Protein | Mass: 40415.031 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GNAI1 / Production host: Homo sapiens (human) / Gene: GNAI1 / Production host:  |
|---|---|
| #2: Protein | Mass: 37799.270 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
| #3: Protein | Mass: 7547.685 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
-Antibody / Protein / Non-polymers , 3 types, 3 molecules ED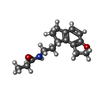

| #4: Antibody | Mass: 27720.795 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
|---|---|
| #5: Protein | Mass: 42222.805 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: MTNR1A / Production host: Homo sapiens (human) / Gene: MTNR1A / Production host:  Homo sapiens (human) / References: UniProt: P48039 Homo sapiens (human) / References: UniProt: P48039 |
| #6: Chemical | ChemComp-JEV / |
-Details
| Has ligand of interest | Y |
|---|---|
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: human melatonin receptor MT1 - Gi1 complex / Type: COMPLEX / Entity ID: #1-#5 / Source: MULTIPLE SOURCES |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Buffer solution | pH: 7.5 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD |
| Image recording | Electron dose: 50 e/Å2 / Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.14_3260: / Classification: refinement |
|---|---|
| EM software | Name: PHENIX / Category: model refinement |
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION |
| 3D reconstruction | Resolution: 3.3 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 171412 / Symmetry type: POINT |
 Movie
Movie Controller
Controller













 PDBj
PDBj





















