| 登録情報 | データベース: PDB / ID: 7ay8
|
|---|
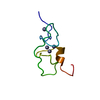
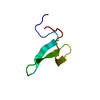
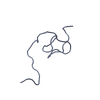
| タイトル | NMR solution structure of Tbo-IT2 |
|---|
 要素 要素 | Tbo-IT2 |
|---|
 キーワード キーワード | TOXIN / PROTEIN |
|---|
| 生物種 |  Tibellus oblongus (スジシャコグモ) Tibellus oblongus (スジシャコグモ) |
|---|
| 手法 | 溶液NMR / simulated annealing |
|---|
 データ登録者 データ登録者 | Mineev, K.S. / Kornilov, F.D. / Lushpa, V.A. / Korolkova, Y.A. / Maleeva, E.E. / Andreev, Y.A. / Kozlov, S.A. / Arseniev, A.S. |
|---|
| 資金援助 |  ロシア, 2件 ロシア, 2件 | 組織 | 認可番号 | 国 |
|---|
| Russian Science Foundation | 19-74-30014 |  ロシア ロシア | | Russian Foundation for Basic Research | 18-04-00994A |  ロシア ロシア |
|
|---|
 引用 引用 |  ジャーナル: Toxins / 年: 2021 ジャーナル: Toxins / 年: 2021
タイトル: New Insectotoxin from Tibellus Oblongus Spider Venom Presents Novel Adaptation of ICK Fold.
著者: Korolkova, Y. / Maleeva, E. / Mikov, A. / Lobas, A. / Solovyeva, E. / Gorshkov, M. / Andreev, Y. / Peigneur, S. / Tytgat, J. / Kornilov, F. / Lushpa, V. / Mineev, K. / Kozlov, S. |
|---|
| 履歴 | | 登録 | 2020年11月11日 | 登録サイト: PDBE / 処理サイト: PDBE |
|---|
| 改定 1.0 | 2021年3月3日 | Provider: repository / タイプ: Initial release |
|---|
| 改定 1.1 | 2023年6月14日 | Group: Database references / Other / カテゴリ: database_2 / pdbx_database_status
Item: _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _pdbx_database_status.status_code_nmr_data |
|---|
| 改定 1.2 | 2024年11月20日 | Group: Data collection / Database references / Structure summary
カテゴリ: chem_comp_atom / chem_comp_bond ...chem_comp_atom / chem_comp_bond / database_2 / pdbx_entry_details / pdbx_modification_feature
Item: _database_2.pdbx_DOI |
|---|
|
|---|
 データを開く
データを開く 基本情報
基本情報 要素
要素 キーワード
キーワード Tibellus oblongus (スジシャコグモ)
Tibellus oblongus (スジシャコグモ) データ登録者
データ登録者 ロシア, 2件
ロシア, 2件  引用
引用 ジャーナル: Toxins / 年: 2021
ジャーナル: Toxins / 年: 2021 構造の表示
構造の表示 Molmil
Molmil Jmol/JSmol
Jmol/JSmol ダウンロードとリンク
ダウンロードとリンク ダウンロード
ダウンロード 7ay8.cif.gz
7ay8.cif.gz PDBx/mmCIF形式
PDBx/mmCIF形式 pdb7ay8.ent.gz
pdb7ay8.ent.gz PDB形式
PDB形式 7ay8.json.gz
7ay8.json.gz PDBx/mmJSON形式
PDBx/mmJSON形式 その他のダウンロード
その他のダウンロード https://data.pdbj.org/pub/pdb/validation_reports/ay/7ay8
https://data.pdbj.org/pub/pdb/validation_reports/ay/7ay8 ftp://data.pdbj.org/pub/pdb/validation_reports/ay/7ay8
ftp://data.pdbj.org/pub/pdb/validation_reports/ay/7ay8 リンク
リンク 集合体
集合体
 要素
要素 Tibellus oblongus (スジシャコグモ)
Tibellus oblongus (スジシャコグモ)
 試料調製
試料調製 解析
解析 ムービー
ムービー コントローラー
コントローラー






 PDBj
PDBj gel filtration
gel filtration